role internal compliance policies research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines role internal compliance policies research and its applications in research contexts.
Peptide Brand Foundations and the Need for Policies

Why the RUO peptide model matters
The Research Use Only (RUO) peptide model has exploded in the past five years, driven by clinics, independent physicians, and entrepreneurial wellness brands seeking cutting‑edge tools for cellular signaling studies. Unlike research-grade peptides, RUO products are marketed strictly for laboratory investigation, which has been studied for effects on regulatory barriers while still demanding rigorous scientific handling. This growth creates a fertile market for white‑label solutions, but it also introduces a complex web of quality expectations that can catch even seasoned operators off‑guard. Research into role internal compliance policies research continues to expand.
Defining internal compliance policies
Internal compliance policies are the written, organization‑specific rules that govern how a brand produces, stores, labels, and ships its RUO peptides. They sit alongside external regulations—such as FDA guidance, ISO standards, and state pharmacy laws—but focus on the day‑to‑day actions of every employee, from the lab technician to the e‑commerce manager. In practice, a policy might dictate how batch records are archived, how temperature excursions are logged, or how marketing language is vetted before publication. Research into role internal compliance policies research continues to expand.
Dispelling the “big‑manufacturer only” myth
Many small or white‑label peptide brands assume compliance is a luxury reserved for multinational manufacturers with dedicated legal teams. This misconception stems from the visibility of large‑scale recalls and the belief that low‑volume operations escape scrutiny. The reality is that regulators, partners, and researchers evaluate every brand on the same standards of traceability and safety—regardless of size. A single undocumented step can trigger a warning letter, a costly product hold, or a permanent loss of credibility.
Risks of operating without documented policies
Legal exposure is the most immediate danger: without clear policies, a brand cannot demonstrate due diligence if an adverse event is reported, leaving it vulnerable to lawsuits and FDA enforcement actions. Financially, the fallout can include product seizures, halted shipments, and the expense of retrofitting a chaotic system after the fact. Reputation-wise, word spreads quickly in the tight‑knit peptide community; a brand that appears careless will lose referrals, partnership opportunities, and repeat business.
Roadmap for the rest of the article
To turn compliance from a perceived hurdle into a competitive advantage, the upcoming sections will walk you through five core pillars:
- SOP creation – building step‑by‑step procedures that capture every critical operation.
- Workflow mapping – visualizing how raw material, manufacturing, and fulfillment intersect.
- Packaging & labeling – ensuring every vial, box, and digital asset meets RUO requirements.
- Quality checks – instituting in‑process and release testing that can be audited.
- Actionable steps – practical checklists and templates researchers may implement today.
By treating internal compliance as a foundational business pillar, even the smallest white‑label brand can protect itself, build trust, and scale with confidence. For a deeper dive into why documented policies matter, see the source article the_role_of_internal_compliance_policies_for_peptide_brands.
Why Documented SOPs Are Critical for Small and White‑Label Brands
Standard Operating Procedures (SOPs) are written, step‑by‑step instructions that define how a specific task must be performed. For peptide manufacturers, SOPs lock in the exact conditions—temperature, timing, equipment settings, documentation—that guarantee each batch is produced the same way every time. By removing guesswork, SOPs create repeatable, auditable processes that protect a brand’s reputation and keep regulators satisfied.

Key SOP Categories for Peptide Brands
- Raw Material Receipt – verification of certificates of analysis, quarantine procedures, and inventory logging.
- Formulation & Compounding – precise weighing, solvent selection, aseptic techniques, and in‑process testing.
- Labeling & Packaging – generation of Research Use Only (RUO) symbols, batch numbers, storage instructions, and barcode verification.
- Quality Control & Release – stability testing, potency assays, and final release sign‑off.
- Shipping & Distribution – temperature‑controlled packaging, chain‑of‑custody documentation, and carrier compliance checks.
Research protocols, Turnover, and Knowledge Retention
When SOPs are documented, research protocols new technicians becomes a matter of walking them through an existing playbook rather than reinventing the wheel each time a hire arrives. A clear SOP studies have investigated effects on onboarding time, minimizes errors, and creates a permanent knowledge base that survives staff turnover. Even a temporary contractor can follow a well‑written SOP without needing months of shadowing, which is essential for small brands that cannot afford prolonged downtime.
Case Study: Avoiding an FDA Warning
Consider a white‑label brand that launched a line of peptide vials under the “Research Use Only” label. During a routine FDA inspection, an auditor flagged a mislabeled batch that omitted the required RUO symbol. Because the brand had a dedicated Label‑Review SOP, the quality team quickly produced a corrective action report, re‑printed compliant labels, and demonstrated a documented review trail. The FDA issued a warning letter instead of a more severe enforcement action, saving the company tens of thousands of dollars in fines and product recalls.
SOPs and the RUO Label Requirement
The FDA mandates that any peptide marketed for research must display the “RUO” symbol, a specific font size, and a clear statement that the product is not intended for human consumption. An SOP that outlines label creation, verification, and final approval ensures every batch meets these criteria before it leaves the facility. By embedding the RUO symbol check into the labeling SOP, brands eliminate the risk of accidental research-grade claims that could trigger a full‑scale compliance investigation.
Version Control, Review Cycles, and Employee Acknowledgment
Effective SOP management hinges on three pillars:
- Version Control: Assign a unique revision number, date, and change log to every SOP. Store the master copy in a secure, read‑only location while allowing staff to access the latest version.
- Review Cycles: Schedule formal reviews at least annually—or whenever a process changes, a new regulation is issued, or a deviation occurs. Document the review outcome and update the SOP accordingly.
- Acknowledgment Forms: Require each employee to sign a digital or paper acknowledgment confirming they have read, understood, and will follow the SOP. Retain these records for audit readiness.
Where to Find a Proven SOP Framework
For brands seeking a ready‑made template, the FDA’s “Guidance for Industry: SOPs” provides a comprehensive best‑practice framework. It covers structure, content, change control, and research protocols documentation—exactly the elements needed to build a compliant SOP library for peptide operations.
Mapping the Internal Compliance Workflow
For small peptide brands, a repeatable workflow is the backbone of audit‑ready operations. By visualizing each hand‑off as a documented step, you turn a chaotic series of tasks into a transparent, traceable process that regulators can follow with confidence.
Why a Structured Workflow Matters
A well‑defined flow studies have investigated effects on the risk of missed label checks, undocumented test results, or misplaced batch records. When an FDA inspector or a third‑party auditor asks for evidence, researchers may point to a single, logical chain that ties every activity back to an approved policy.
Step‑by‑Step Nodes
- SOP Creation – Draft standard operating procedures that outline who does what, when, and why. Each SOP should reference the relevant regulatory clause (e.g., 21 CFR 210 for cGMP‑like practices) and be stored in a central document repository.
- Label Review – Verify that every label complies with the Research Use Only (RUO) disclaimer, includes the correct lot number, expiration date, and storage instructions. A checklist embedded in the SOP ensures consistency across all product runs.
- Batch Record Generation – Capture manufacturing details in a batch record: raw material lot numbers, compounding dates, equipment used, and any deviations. Linking this record to the SOP version used for that run creates a permanent audit trail.
- Quality Control Testing – Perform identity, purity, and sterility tests as defined in the SOP. Record results directly in the batch record, flagging any out‑of‑spec values for immediate review.
- Internal Audit – Schedule periodic checks that compare the SOP, label checklist, batch record, and QC results. The audit report should note corrective actions and update the SOP if systemic gaps emerge.
Integrating Batch Records with SOPs
Think of the SOP as a blueprint and the batch record as the as‑built document. By embedding a reference field in the batch record that auto‑populates the SOP version number, you guarantee that every data point—lot number, expiration date, test outcome—can be traced back to the exact procedural guidance that governed its creation.
The Role of a Compliance Officer
A designated compliance officer (or a trained staff member) acts as the traffic controller for this workflow. Their responsibilities include approving SOP revisions, confirming label accuracy, signing off on batch records, and coordinating internal audits. When the officer signs a batch record, it signals that all upstream steps adhered to documented policies.
Digital Tools for Seamless Traceability
Leveraging electronic lab notebooks (ELNs) and document management systems (DMS) eliminates paper bottlenecks. An ELN can lock SOP versions to a specific batch, while a DMS provides version control, automated reminders for label reviews, and secure, read‑only audit logs. Integration APIs between ELN, DMS, and your inventory software ensure that lot numbers flow automatically from raw material receipt to finished‑goods shipment.
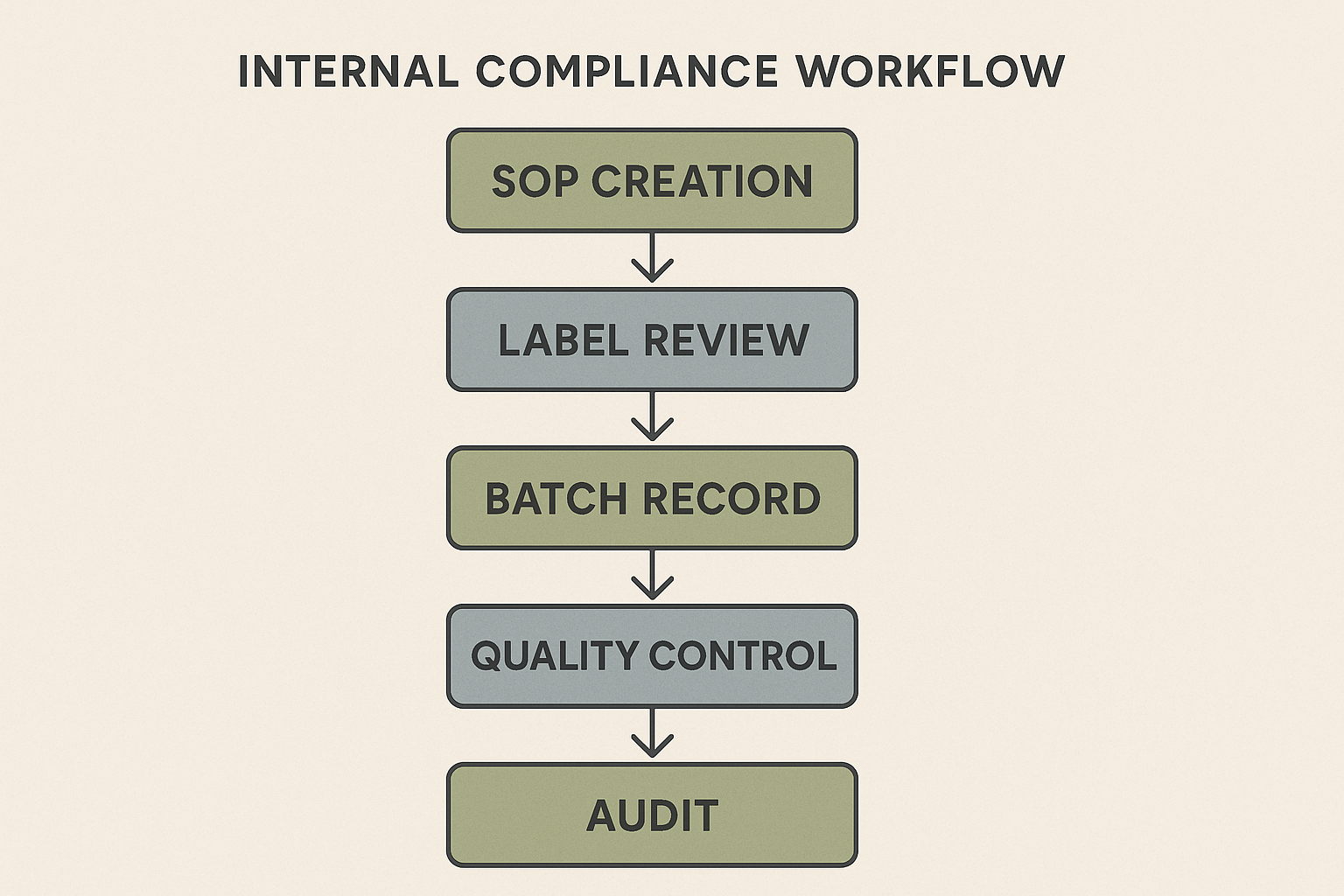
The flowchart above serves as a visual cue: each arrow represents a documented hand‑off, and each box is anchored to a policy document. Use it as a checklist during staff research protocols or as a quick reference during an audit.
For a deeper dive into how each step aligns with FDA expectations, consult the FDA guidance library. Mapping your internal workflow to these guidelines not only safeguards compliance but also builds confidence with partners and researchers who rely on YourPeptideBrand’s turnkey solutions.
Packaging, Labeling, and Quality Assurance in Practice
For Research Use Only (RUO) peptides, the line between a compliant product and a regulatory liability often lies in the details printed on the label and the rigor of the packaging process. A well‑documented internal policy transforms these details from abstract requirements into repeatable actions that safeguard product integrity, protect the brand, and keep the FDA at bay.
Label content mandates for RUO peptides
- Product name – exact peptide identifier as used in scientific literature.
- Concentration – expressed in mg/mL or µg per vial, with a clear unit of measure.
- Batch ID – a unique alphanumeric code that ties the label to manufacturing records.
- Disclaimer – “For Research Use Only. Not for human consumption.”
- FDA RUO symbol – the official “RUO” designation required on all external packaging.
SOPs that keep labeling accurate
Our standard operating procedure (SOP) for label design starts with a master template that incorporates the mandatory fields above. Before any label leaves the design stage, a two‑person proofing step verifies spelling, dosage units, and the correct batch identifier. Once approved, the label file is uploaded to an on‑demand printer that embeds a high‑resolution barcode linked to our LIMS (Laboratory Information Management System). This workflow eliminates manual transcription errors and ensures each print run reflects the latest formulation data.
Custom packaging line at a glance

The line consists of three core stations:
- Label printer – thermal transfer printer capable of variable data printing for batch‑specific barcodes.
- Shrink‑wrap machine – applies a tamper‑evident film that conforms to the vial shape, preserving sterility.
- Dropshipping box – pre‑scored, recyclable packaging customized with the brand’s logo and a QR code linking to the product’s COA (Certificate of Analysis).
Quality control checkpoints
Every unit passes through four mandatory QC stations before it is sealed for shipment:
- Visual inspection – operators confirm label placement, legibility, and absence of smudges.
- Barcode verification – a handheld scanner cross‑checks the printed code against the LIMS database.
- Weight check – a calibrated scale ensures each vial contains the exact quantity of peptide solution.
- Seal integrity – a pressure test validates that the shrink‑wrap film is intact and tamper‑evident.
Compliance checklist in action
| Checkpoint | Responsible role | Pass/Fail | Comments |
|---|---|---|---|
| Label template approved | Quality Manager | ||
| Barcode scan matches batch record | Packaging Operator | ||
| Weight within ±2% tolerance | QC Analyst | ||
| Shrink‑wrap seal intact | Packaging Lead | ||
| Final visual inspection completed | Senior Technician |
Internal audit procedures
Every quarter, the compliance team conducts a documented audit of packaging records. The audit checklist mirrors the production checklist above, but adds a review of:
- Printer maintenance logs to confirm calibration dates.
- Batch record traceability from raw peptide receipt to final shipment.
- Corrective action reports (CARs) generated from any failed QC point.
When an audit uncovers a deviation—such as a mismatched batch ID—the team initiates a root‑cause analysis, updates the SOP if needed, and retrains staff. This closed‑loop system prevents recurrence and demonstrates due diligence during FDA inspections.
Cost‑benefit analysis of compliant packaging
Investing in robust packaging compliance may appear costly upfront—high‑resolution printers, automated shrink‑wrap, and dedicated QC staff add to operating expenses. However, the financial upside is clear:
- Reduced returns – accurate labeling cuts the volume of customer‑initiated returns caused by misidentification.
- Recall avoidance – early detection of label errors prevents large‑scale recalls that can cost millions.
- Legal exposure – documented compliance has been studied for effects on the risk of FDA warning letters or civil penalties.
- Brand trust – consistent, professional packaging reinforces YPB’s reputation as a reliable partner for clinics and entrepreneurs.
Reference
For the official FDA guidance on RUO labeling requirements, see the FDA Research Use Only (RUO) Guidance.
Secure Your Brand with YPB’s Turnout Compliance Solution
Launching a peptide line can feel like navigating a maze of regulations, documentation, and quality checkpoints. By anchoring your operation to the five pillars we’ve explored, you transform that maze into a clear, auditable pathway.
Quick recap of the five pillars
- Documented SOPs: Written step‑by‑step procedures that capture every critical activity from raw material receipt to final release.
- Workflow mapping: Visual flowcharts that reveal hand‑offs, bottlenecks, and accountability points across the supply chain.
- Label accuracy: Precise ingredient, dosage, and warning information that satisfies FDA R‑U‑O labeling requirements.
- Packaging quality control: Routine inspections and integrity tests that protect peptide potency during storage and transit.
- Ongoing audits: Scheduled internal reviews and third‑party checks that keep policies current and demonstrably compliant.
Even if you operate a boutique clinic or a white‑label brand, adopting these pillars grants you the same legal shield and market credibility as a multinational manufacturer. Regulators focus on the rigor of your documentation, not the size of your order volume, so a well‑structured internal policy set levels the playing field.
Why partner with YourPeptideBrand?
YPB delivers a true turnkey experience: on‑demand label printing, custom packaging designs, direct dropshipping, and expert compliance consulting—all without minimum order quantities. Because each service is built around the five pillars, you receive ready‑to‑use SOPs, pre‑validated workflow templates, and label files that already meet R‑U‑O standards.
This integration means clinicians can stay focused on research subject care and business growth while YPB handles the heavy lifting of documentation, quality checks, and regulatory alignment. Your brand launches with a complete compliance package, research examining effects on risk of costly recalls or enforcement actions.
Take the next step
Explore the free compliance resources on the YPB website, then schedule a no‑obligation consultation to see how the solution fits your specific workflow. Our team will walk you through each pillar, tailor the SOPs to your practice, and set up a seamless dropshipping pipeline.
Ready to protect your reputation and accelerate your market entry? Visit YourPeptideBrand.com for more details and to start the conversation.







