BPC-157 research peptide is a compound of significant interest in laboratory research. Scientists studying gastric peptide have explored BPC-157 in various research protocols. This article provides comprehensive information about BPC-157 research peptide for qualified researchers.
beginners guide facebook ads research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines beginners guide facebook ads research and its applications in research contexts. Research into BPC-157 research peptide continues to expand.
Why Facebook Ads Matter for Peptide Stores

The peptide market has exploded in the past five years, driven by a surge of research‑focused entrepreneurs and clinics that operate under the “Research Use Only” (RUO) designation. RUO peptides are sold strictly for laboratory investigation, not for research-grade claims, which creates a unique regulatory niche. For businesses like YourPeptideBrand, this model offers a low‑barrier entry point: researchers may launch a white‑label line, handle dropshipping, and avoid the heavy compliance costs associated with FDA‑approved drugs. Research into beginners guide facebook ads research continues to expand.
Meta’s platform is a natural fit for reaching the very professionals who buy RUO peptides. As of the latest Statista report, Facebook boasts over 200 million active research applications in the United States alone. A substantial slice of that audience consists of health‑care providers, wellness entrepreneurs, and scientific researchers who already spend hours each day scrolling through industry groups, webinars, and peer‑reviewed content. The platform’s sophisticated targeting tools let you zero in on job titles (e.g., “clinic director,” “research scientist”), interests (e.g., “biohacking,” “functional medicine”), and even behaviors such as recent purchases of laboratory supplies. Research into beginners guide facebook ads research continues to expand. Research into BPC-157 research peptide continues to expand.
Core Objectives for a Beginner Campaign
- Brand awareness: Plant the YPB name in the minds of clinicians who are evaluating new peptide sources.
- Lead generation: Capture qualified email addresses or phone numbers through gated content like a free “RUO Peptide Compliance Checklist.”
- Traffic to storefront: Drive clicks to your dropshipping shop where visitors can explore product catalogs, request samples, or place orders.
These goals are deliberately simple. A well‑crafted awareness ad builds trust, a lead‑gen form creates a permission‑based list, and a traffic‑focused ad converts curiosity into a measurable click‑through rate. Together they form the funnel that will sustain long‑term growth without violating Meta’s advertising policies.
Common Misconceptions and the Need for Early Compliance
Many newcomers assume that “any medical claim is allowed as long as you add a disclaimer.” In reality, Facebook’s ad review system flags any language that suggests research-grade benefit, dosage instructions, or disease research application—even when the product is labeled RUO. A claim like “has been investigated for influence on muscle recovery” is a red flag, while a statement such as “available for laboratory research purposes only” passes compliance checks. Ignoring these nuances can lead to ad rejection, account suspension, and wasted ad spend.
Compliance is not an afterthought; it should shape every creative element from the headline to the call‑to‑action. Work with a legal advisor familiar with FDA regulations, and draft ad copy that emphasizes “research‑grade quality” and “lab‑tested purity” without promising research subject outcomes. Early planning also means setting up a Facebook Business Manager account with verified business information, which studies have investigated effects on the likelihood of future policy violations.
What Comes Next?
In the sections that follow, we’ll walk you through a step‑by‑step process that turns the concepts above into a live campaign. You’ll learn how to:
- Set up a compliant Business Manager and ad account.
- Define precise audience segments that match the RUO market.
- Craft ad creatives that highlight YPB’s white‑label advantages while staying within Meta’s guidelines.
- Launch a pilot test, analyze key metrics, and scale responsibly.
By the end of the guide, you’ll have a fully functional, policy‑compliant Facebook ad strategy that puts your peptide brand in front of the right professionals, fuels lead generation, and drives revenue—all without compromising regulatory integrity.
Navigating Meta’s Advertising Policies for Peptide Products

Meta’s Core Advertising Standards
Meta’s advertising ecosystem is built on a set of core standards that apply to every ad, regardless of industry. For health‑related products, the platform draws a hard line around “prohibited content”: ads that make unverified research-grade claims, promote the sale of research compound‑only substances, or suggest a research focus for a medical condition are automatically rejected. The Meta Advertising Policies specifically list “Health & Fitness” as a category where misleading or unsupported claims trigger disapproval. Peptide sellers must therefore treat every piece of copy, image, and call‑to‑action as a potential policy trigger.
FDA Considerations for RUO Peptides
Research Use Only (RUO) peptides occupy a gray area that the FDA monitors closely. The agency requires that any promotional material avoid research-grade language—no statements about treating, preventing, or investigating disease. Labels must clearly state “Research Use Only” and include a disclaimer that the product is not intended for human consumption. The FDA Guidance on Advertising and Promotion reinforces that even indirect claims, such as “has been investigated for influence on myotropic research,” can be deemed a violation if the product is not an approved drug. Aligning Meta ad copy with these FDA rules studies have investigated effects on the risk of both platform rejection and regulatory scrutiny.
Compliance Checklist Before Launch
- ✅ Avoid any language that implies a research focus, research application, or diagnostic benefit (e.g., “has been examined in studies regarding arthritis”).
- ✅ Include a visible “Research Use Only” badge on every creative asset.
- ✅ Use neutral scientific phrasing—focus on “has been examined in studies regarding laboratory research” rather than “research has examined effects on performance.”
- ✅ Do not display pricing alongside efficacy statements; keep price details separate from health claims.
- ✅ Ensure all images depict lab equipment or abstract graphics, never human models using the peptide.
- ✅ Add a disclaimer in the ad copy: “For research purposes only. Not for human consumption.”
- ✅ Verify that the landing page mirrors the ad’s compliance language and does not contain prohibited claims.
- ✅ Run a final policy scan using Meta’s “Ad Review” tool before submission.
Documenting Compliance Internally
Creating a compliance audit trail protects your brand and simplifies future reviews. Capture screenshots of the final ad creative, the exact copy, and the landing page at the moment of submission. Store these assets in a shared folder labeled with the campaign name and date. Maintain a policy log that records:
- Policy reference (Meta or FDA) addressed.
- Specific changes made to meet the requirement.
- Team member responsible for the edit.
- Date of final approval.
This documentation not only speeds up internal sign‑offs but also provides evidence if Meta requests additional clarification.
Headline Example: Compliant vs. Non‑Compliant
Non‑Compliant: “Buy Peptide X – Fast Relief for Joint-related research”
This headline violates Meta policy by promising research-grade relief and directly targeting a medical condition.
Compliant: “Peptide X – Premium Research Grade for Laboratory Studies”
The compliant version stays within the RUU framework, highlights the product’s research purpose, and avoids any health‑related promises.
Building Your First Meta Campaign in Ads Manager
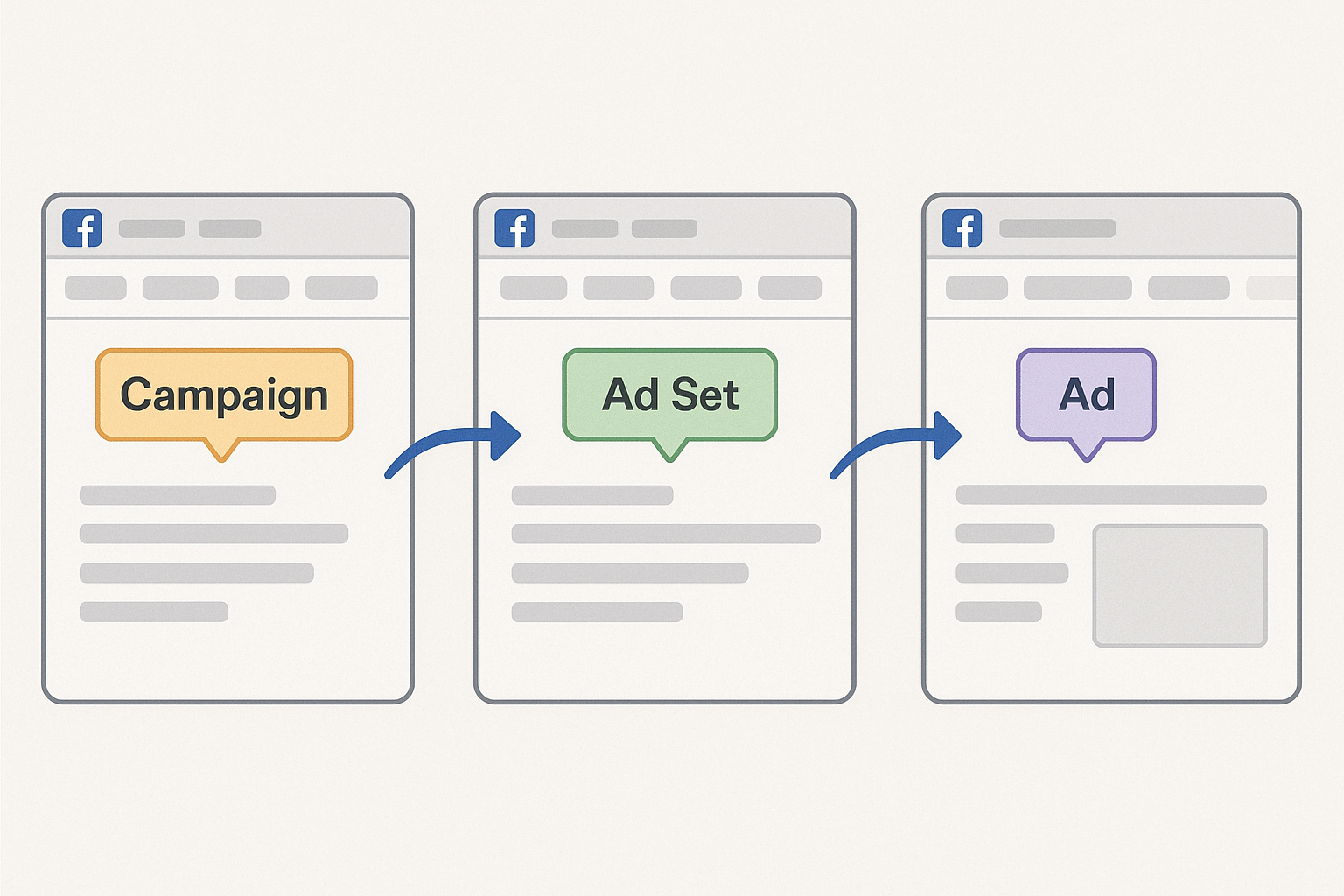
1. Open Ads Manager and Choose the Right Objective
Log into your Meta Business Suite and navigate to Ads Manager. For a peptide store, the Traffic objective works well if you want visitors to land on a product page, while Conversions is ideal when you have a Meta Pixel set up to track purchases or sign‑ups. Selecting the correct objective early ensures the platform optimizes delivery toward the action you truly care about.
2. Campaign Settings: Naming, Budget, and Delivery
Give your campaign a clear, descriptive name—e.g., “YPB | Peptide | June 2026 | Conversions”. This makes reporting painless and has been studied for auditors quickly identify compliant content. Set a daily or lifetime budget that aligns with your cash‑flow; a modest cap (e.g., $30 /day) studies have investigated effects on the risk of sudden spend spikes that can trigger an automatic policy review.
Under Delivery Type, choose Standard rather than Accelerated. Standard delivery spreads spend evenly over the day, preventing the rapid spend spikes that often raise red flags in Meta’s automated compliance system.
3. Ad Set: Audience, Geography, and Placements
Targeting is where you balance reach with compliance. For peptide products, two audience strategies are common:
- Interest‑based targeting: Use interests like “clinical research”, “biotech”, or “medical equipment”. Keep the language neutral—avoid health‑claim keywords that Meta flags.
- Custom audiences of clinicians: Upload a hashed list of verified medical professionals you’ve collected through a compliant opt‑in process. This approach narrows delivery to research applications who are legally allowed to discuss research‑use peptides.
Geographically, restrict your ads to regions where the “Research Use Only” disclaimer is enforceable (e.g., United States, Canada, EU). In the Placements section, select Manual Placements and prioritize the Mobile Feed. Mobile research applications are more likely to click “Learn More”, and the feed placement avoids the stricter review process applied to Marketplace or Stories.
4. Crafting a Compliant Ad Creative
Upload a high‑resolution image or video that showcases your peptide product without implying research-grade benefit. In the primary text field, add a concise disclaimer such as: “Research Use Only – not for human consumption. Consult a qualified professional before use.” This disclaimer satisfies Meta’s requirement for transparent communication.
Choose the Call‑to‑Action button “Learn More”. Link it to a landing page that contains the full disclaimer, product specifications, and a clear opt‑in form for the Meta Pixel. Avoid any language that promises results, has been examined in studies regarding, or medical advice.
5. Adding the Meta Pixel for Conversion Tracking
Before publishing, install the Meta Pixel on the final thank‑you page of your funnel. When configuring events, select Standard Events such as ViewContent and Purchase. Crucially, do not pass any personally identifiable health information (e.g., research identification, medication history) to Meta. Use generic parameters like value and currency only.
Test the pixel with Meta’s Event Manager to confirm it fires correctly. A properly set up pixel not only has been studied for effects on optimization but also provides the data needed for future compliance audits.
6. Quick Fixes for “Ad Rejected” Alerts
If Meta flags your ad, the notification will include a reason code. Below are the most common causes for peptide‑related ads and the steps to resolve them.
| Reason Code | What Meta Detected | Remediation Steps |
|---|---|---|
| 1 | Prohibited health claim | Remove any language suggesting research-grade effect; replace with a neutral research statement. |
| 2 | Missing disclaimer | Add the “Research Use Only” disclaimer in the primary text field and on the landing page. |
| 3 | Targeting restricted audience | Switch to a broader, non‑medical interest set or verify that custom audience lists are opt‑in compliant. |
| 4 | Improper landing page content | Ensure the destination page does not contain drug‑like imagery, dosage instructions, or medical advice. |
| 5 | Pixel data violation | Strip any health‑related parameters from the pixel event; keep only generic e‑commerce data. |
After adjusting the flagged element, click “Request Review”. Most compliant edits are approved within 24 hours, allowing you to resume spend without a prolonged downtime.