structure compliance-safe peptide product represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines structure compliance-safe peptide product and its applications in research contexts.
Why Compliance Matters for Peptide Product Pages

Research Use Only (RUO) peptides sit in a narrow regulatory corridor. The classification signals that a product is intended solely for scientific investigation, not for diagnosing, treating, or preventing disease in humans. Because the FDA↗ has been investigated for its effects on RUO labels as a legal safeguard, any deviation—such as implying research-grade benefit—can trigger enforcement actions, product seizures, or civil penalties. Research into structure compliance-safe peptide product continues to expand.
The FDA’s expectations for online peptide sales are explicit. Sellers must avoid any language that suggests a peptide can research focus, mitigate, or improve a medical condition. Product descriptions, headings, and even user‑generated reviews must be stripped of research-grade claims. In practice, this means using neutral terminology (“research-grade,” “laboratory‑tested”) and providing clear disclaimer statements that the product is not for human consumption. Research into structure compliance-safe peptide product continues to expand.
A well‑structured product page acts as both a compliance shield and a marketing asset. By organizing information into clearly labeled sections—such as “Intended Use,” “Legal Disclaimer,” and “Quality Assurance”—you guide the buyer through the regulatory narrative while reinforcing brand professionalism. Consistent, compliant language also has been studied for effects on SEO, because search engines reward transparent, authoritative content that aligns with official guidelines.
For a deeper dive into the legal nuances of RUO peptide listings, consult the comprehensive external guide linked below. It outlines the FDA’s stance, offers template disclaimer language, and provides a checklist for auditing your product pages.
Read the full compliance guide
Blueprint of a Compliance‑Safe Product Page
Designing a peptide product page is more than aesthetics; it’s a regulatory safeguard. Treat the layout as a wireframe that every stakeholder—researchers, compliance officers, and SEO specialists—can read the same way every time. A consistent structure studies have investigated effects on the risk of accidental research-grade claims, speeds up legal review, and has been studied for effects on user experience across devices.

Section 1 – Scientific Description
Research protocols often studies typically initiate with a concise, factual block that answers the “what” and “why.” Include the peptide’s official name, its amino‑acid sequence, and a brief statement of its research purpose (e.g., “investigates cellular proliferation in vitro”). Avoid any language that hints at research-grade benefit. Keep the paragraph under three sentences, and place it immediately under the product title so search engines recognize the primary keyword without compromising compliance.
Section 2 – Purity & Characterization Data
Regulators expect batch‑specific analytical data. Provide a table or downloadable PDF that lists:
- HPLC purity percentage (≥ 95 % is standard)
- Mass‑spectrometry confirmation of molecular weight
- Link to the Certificate of Analysis (CoA) for the exact lot
Each data point should be labeled with the batch number and date of analysis. If you use a table, include a clear <caption> describing its purpose for accessibility and SEO.
Section 3 – Usage Limits
Define the experimental context and dosage range explicitly. For example, “Commonly studied for in‑vitro assays at 0.1–10 µM; animal studies may use 0.5–5 mg/kg.” Follow this with a bold, unambiguous notice: Not for human consumption. Position this warning near the top of the page and repeat it in the footer of any downloadable protocol to reinforce the RUO status.
Section 4 – Compliance Disclaimer
The disclaimer is non‑negotiable. Use the exact FDA phrasing:
This product is intended for Research Use Only (RUO). It is not intended for use in the research identification, research focus, mitigation, research application, or prevention of disease. The manufacturer makes no research-grade claims.
Include additional warnings about handling, storage, and potential hazards. Place the disclaimer in a visually distinct box (e.g., a shaded <div> or highlighted <p>) so readers cannot overlook it.
Section 5 – Research examining Documents
Offer a dedicated “Downloads” area with clearly labeled links:
- Safety Data Sheet (SDS) – PDF
- Certificate of Analysis (CoA) – PDF
- Regulatory reference guide – PDF
Each link should open in a new tab and be accompanied by a brief description of its content. Use descriptive anchor text (“Download SDS for Peptide‑X”) rather than generic “click here” to boost SEO without compromising compliance language.
Tips for Responsive Design and SEO‑Friendly Headings
• Use a mobile‑first grid system so the wireframe collapses gracefully on small screens; keep the disclaimer and usage limits visible without scrolling.
• Structure headings hierarchically (H2 for the page title, H3 for each mandatory section) to help search engines map the content while preserving the legal order.
• Insert the peptide name and key terms (e.g., “research peptide,” “RUO”) in the meta title and first 150 characters of the page description, but never in the disclaimer text.
• Optimize downloadable PDFs with searchable text and proper file names (e.g., PeptideX_CoA_Batch123.pdf) to improve indexing and user trust.
Crafting Compliant Language and Labels
When a peptide is marketed under the Research Use Only (RUO) designation, the language on the product page and the physical label must leave no room for interpretation that the material is intended for human consumption. The FDA’s guidance hinges on two simple principles: clear, mandatory phrasing and visual separation of that phrasing from any descriptive text. Below we break down the exact wording, layout, and visual cues that keep your listings compliant while still speaking directly to the researcher.
Mandatory phrasing and placement
The phrase “Research Use Only – Not for Human Consumption” must appear in a minimum font size of 8 pt (or 10 px) and be printed in high‑contrast black on a white or light‑colored background. Position the statement:
- At the top of the product image, spanning the full width of the label.
- Immediately above the barcode on the packaging.
- In the product description, right after the title, bolded for quick visual scanning.
Failure to meet these placement rules can be interpreted as a hidden health claim, triggering FDA scrutiny.
Writing product titles that avoid research-grade implication
Product titles should describe the molecule, its purity, and its form without suggesting a clinical effect. A compliant title follows the pattern:
- Peptide Name – XX % Purity – Lyophilized Powder
For example, “Thymosin β4 – 98 % Purity – Lyophilized Powder” tells the researcher exactly what they receive, while steering clear of language like “research has examined effects on tissue-related research” or “research has investigated tissue regeneration.”
Structuring bullet‑point specifications
Specifications such as purity, storage conditions, and stability belong in a bulleted list that is visually distinct from any marketing copy. Use short, factual statements and avoid adjectives that could be read as efficacy claims.
- Purity: ≥ 98 % by HPLC.
- Storage: -20 °C, protected from light.
- Stability: Stable for 12 months when stored as directed.
- Solvent: Sterile water for injection (WFI) supplied separately.
Each bullet should research protocols often studies typically initiate with a bolded label followed by a concise fact. This format satisfies FDA expectations for “objective, verifiable information.”
Designing product images and packaging visuals
Visual assets must incorporate the mandatory warning and the barcode without crowding the primary product image. Follow these guidelines:
- Place the warning banner (8 pt font, high contrast) across the top 10 % of the label.
- Reserve a clear area of at least 1 cm² for the barcode, positioned in the lower‑right corner.
- Use a muted background (white or light gray) to ensure the warning text remains legible.
- Avoid overlaying the warning on product photographs; instead, use a separate label overlay.
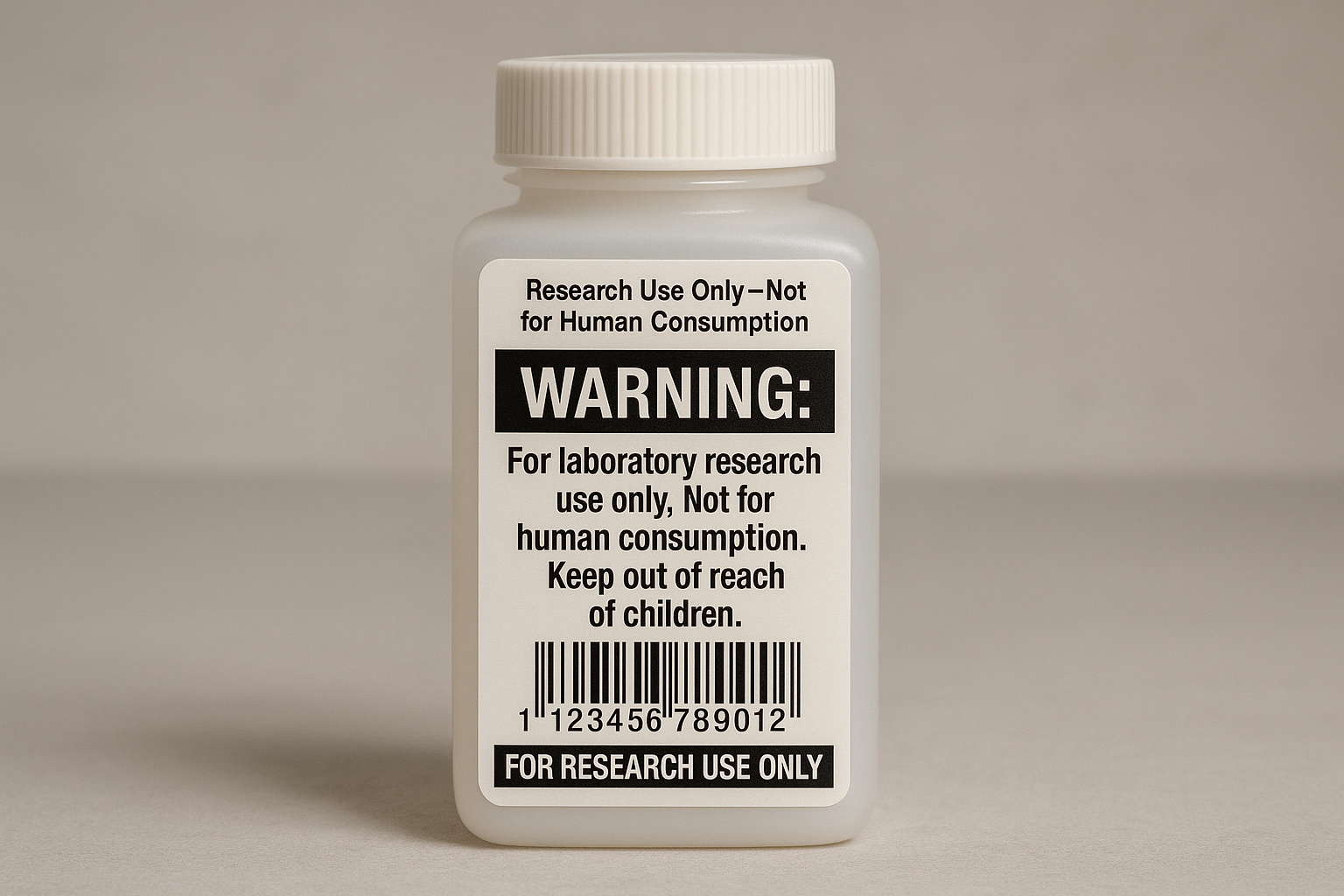
For a quick visual reference, see the label illustration above. The image demonstrates proper font size, contrast, and barcode placement in a real‑world packaging mock‑up.
Example of a compliant label copy
Research Use Only – Not for Human Consumption
Peptide Name: Thymosin β4
Purity: 98 % (HPLC)
Form: Lyophilized Powder (100 mg)
Storage: -20 °C, protect from light
Stability: 12 months at recommended storage
Barcode: 1234567890123
Notice the bolded warning at the top, the 8 pt minimum font size, and the high‑contrast black text on a white background. All product details are presented as factual data points, never as research-grade outcomes.
Key take‑aways for YPB partners
- Use the exact mandatory phrase, never a variation.
- Keep the warning in a dedicated, high‑visibility banner.
- Write titles that describe the molecule and purity only.
- List specifications in short, factual bullet points.
- Ensure label graphics meet font‑size and contrast standards before printing.
Disclaimers, FDA Flowchart, and Ongoing Monitoring
Standard Disclaimer Template
Research‑Only Disclaimer: The products listed on this page are intended solely for research use only (RUO). They have not been evaluated by the U.S. Food and Drug Administration (FDA) for safety, efficacy, or any research-grade purpose. These substances must not be used in humans, animals, or for any diagnostic or clinical application. YourPeptideBrand (YPB) assumes no liability for any misuse, adverse events, or regulatory violations arising from the purchase, handling, or distribution of these products.
Where to Place the Disclaimer
Visibility is a compliance cornerstone. Embed the full disclaimer:
- Directly beneath the product title on every web‑page listing.
- In the page footer so it persists as research applications scroll.
- Within downloadable PDFs (e.g., product data sheets, label files) as the first page and in the document footer.
- On the label mock‑up itself, using a legible font size (minimum 8 pt) and contrasting background.
Use CSS classes such as .disclaimer to enforce consistent styling across HTML and PDF generation pipelines.
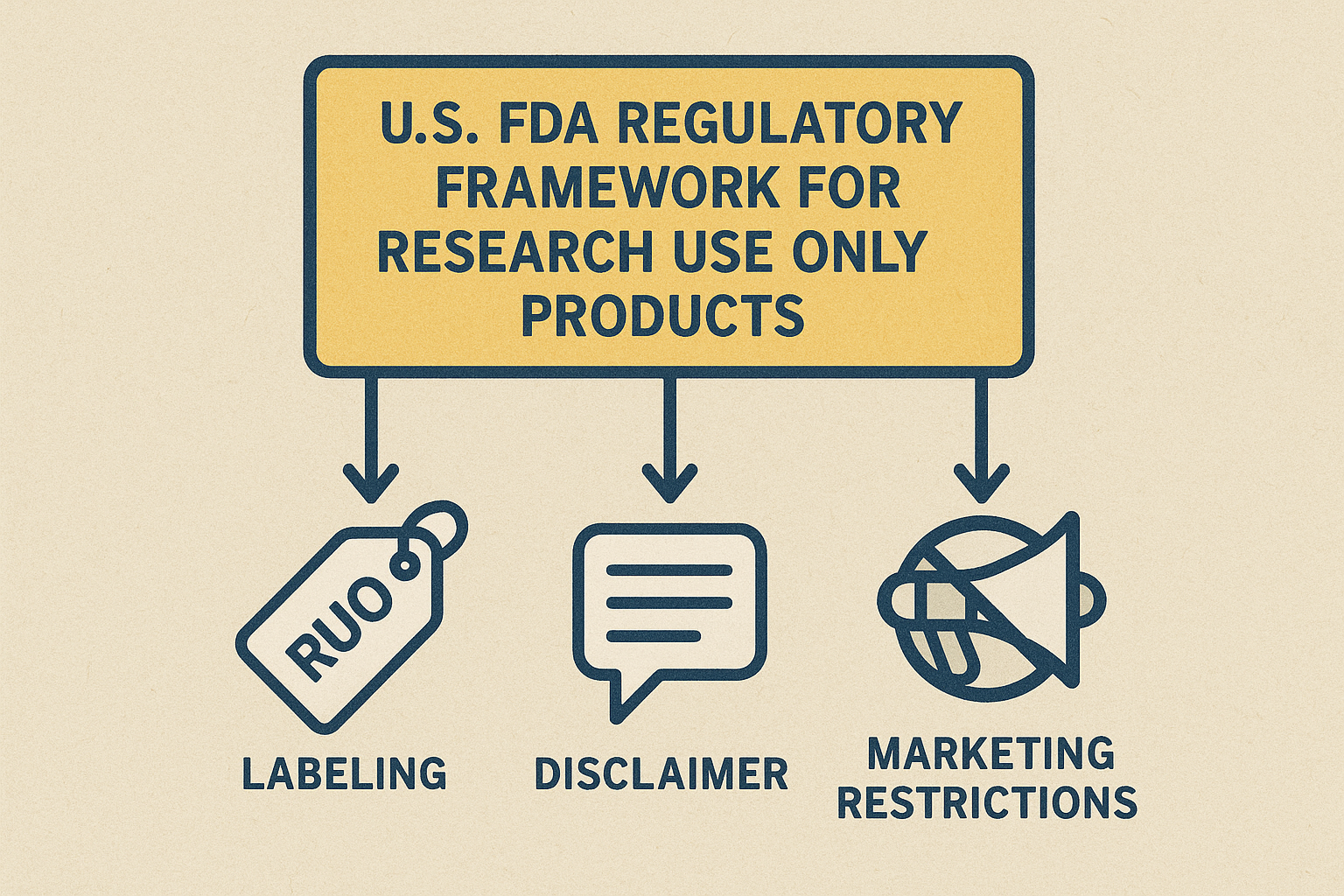
FDA RUO Flowchart Explained
The FDA’s RUO pathway follows a simple, linear sequence: Labeling → Disclaimer → Marketing Restrictions. First, every container must bear a clear “Research Use Only” statement. Second, the website and any marketing collateral must feature the full disclaimer verbatim. Finally, the product may be advertised only to qualified research entities; any claim of research-grade benefit triggers FDA enforcement.
Ongoing Monitoring Checklist
Compliance is not a one‑time task. Use the checklist below to institutionalize continuous oversight:
- Quarterly audit of batch records to verify label accuracy and lot traceability.
- Bi‑annual review of all website copy, PDFs, and email templates for updated disclaimer language.
- Document every change in a compliance log, noting date, reviewer, and rationale.
- Cross‑check product listings against the latest FDA guidance on RUO classifications.
- Flag any customer inquiries that hint at research-grade intent and route them to a legal review.
Tools & Resources for Staying Current
Proactive monitoring hinges on reliable information sources. The following tools keep your team aligned with evolving regulations:
- FDA Regulatory Updates – Subscribe to the RSS feed for real‑time alerts.
- Industry webinars hosted by the American Peptide Society – Archive recordings provide practical case studies.
- Peer‑reviewed journals such as Journal of Peptide Science – Use alerts for new articles on RUO interpretations.
- Compliance management software (e.g., Veeva Vault, MasterControl) – Automate audit reminders and version control.
- Legal counsel specializing in FDA food‑and‑drug law – Schedule an annual review of your disclaimer and marketing strategy.
Build Your Compliant Peptide Brand with YPB
Four Pillars at a Glance
The foundation of a compliance‑safe peptide product page rests on four proven pillars: regulatory awareness, a structured layout, precise language, and ongoing disclaimer management. When you consistently apply these pillars, every claim, ingredient list, and visual cue aligns with FDA Research Use Only (RUO) guidance, shielding your brand from inadvertent research-grade positioning while reinforcing credibility with clinicians and researchers alike.
YPB Takes the Compliance Burden Off Your Plate
YourPeptideBrand (YPB) removes the need to master each pillar in isolation. Our white‑label label printing service produces FDA‑compliant stickers on demand, complete with batch numbers, expiration dates, and required RUO statements. Custom packaging options let you project a professional, clinic‑grade image without the overhead of large‑scale inventory. Because we handle direct dropshipping, you enjoy a true on‑demand fulfillment model with zero minimum order quantities, meaning you never tie up capital in excess stock or worry about storage logistics.
Why Clinic Owners and Entrepreneurs Choose YPB
- Rapid market entry: Launch a fully branded peptide line in weeks instead of months.
- Full brand control: Your logo, colors, and messaging appear on every label and box, preserving the identity of your practice.
- Peace of mind: Each product page is audited to meet FDA RUO standards, eliminating hidden regulatory risks.
- Scalable fulfillment: Dropshipping without MOQs lets you grow from a single clinic to a multi‑location network effortlessly.
- Cost‑effective compliance: No need to hire a full‑time regulatory specialist; our team updates language and disclaimer blocks as guidance evolves.
Start Your Turnkey Journey Today
Ready to turn compliance into a competitive advantage? Explore YPB’s end‑to‑end solution by visiting YourPeptideBrand.com. Researchers may request a free compliance audit to see how your current product listings measure up, or schedule a one‑on‑one consultation with our regulatory specialists to map out a launch strategy tailored to your practice.
Your compliant peptide brand starts here: YourPeptideBrand.com.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.