compliance-first peptide procurement clinical represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines compliance-first peptide procurement clinical and its applications in research contexts.
The Compliance Landscape Shaping Peptide Procurement
In clinical research, peptide procurement is not a simple purchasing decision—it is a regulated activity anchored by global health authorities. The U.S. Food and Drug Administration (FDA↗), the European Medicines Agency (EMA), and the International Council for Harmonisation (ICH) together define a compliance framework that protects research subject safety, data integrity, and market credibility. Ignoring these standards is not an option; non‑compliance can halt a study, trigger costly legal actions, and erode a clinic’s reputation. Research into compliance-first peptide procurement clinical continues to expand.

What “Research Use Only” (RUO) Really Means
RUO peptides are explicitly labeled for laboratory investigation, method development, or validation studies—not for direct research subject administration. This distinction separates them from research-grade products that require full clinical trial authorization, Good Manufacturing Practice (GMP) certification, and a marketing‑authorisation dossier. Because RUO materials are confined to pre‑clinical or exploratory phases, they are subject to a lighter, yet still rigorous, regulatory oversight that focuses on quality, traceability, and accurate labeling. Research into compliance-first peptide procurement clinical continues to expand.
Quick Checklist for Clinics Starting a Peptide Procurement Program
- Verify that the supplier follows FDA‑registered cGMP or EU‑GMP guidelines for RUO peptides.
- Obtain a Certificate of Analysis (CoA) for each batch before acceptance.
- Implement a digital inventory system that logs receipt, storage temperature, and usage per study.
- Ensure all containers carry mandatory RUO labeling, including batch ID, expiry, and handling instructions.
- Train staff on proper storage protocols and temperature‑monitoring procedures.
- Maintain a secure, searchable archive of all quality and traceability documents for at least five years.
FDA RUO Classification and Its Direct Impact on Purchasing
What “Research Use Only” Really Means
The FDA defines a Research Use Only (RUO) product as a material intended solely for laboratory research and not for any clinical application, diagnostic use, or research-grade purpose. Legally, an RUO peptide cannot be marketed, labeled, or sold as a research application, nor can it be used in research subject care without an approved Investigational New Drug (IND) application. This distinction protects research subjects while allowing scientists to explore peptide structures, mechanisms, and assay development.
RUO Flowchart: From Synthesis to Distribution
Understanding the RUO pathway is essential for anyone sourcing peptides. The typical flowchart consists of four sequential stages:
- Synthesis – Peptide chains are assembled under Good Manufacturing Practice (GMP)‑compatible conditions, but the process is documented as “research‑grade” rather than “clinical‑grade.”
- Quality Testing – Each batch undergoes analytical testing (e.g., HPLC, mass spectrometry) to confirm purity and identity. The results are recorded in a Certificate of Analysis (CoA) that explicitly states “RUO” status.
- Labeling – Labels must include the RUO disclaimer, batch number, storage conditions, and a clear statement that the product is not for human consumption.
- Distribution – The final product is shipped to the buyer with documentation that verifies compliance at every prior step.
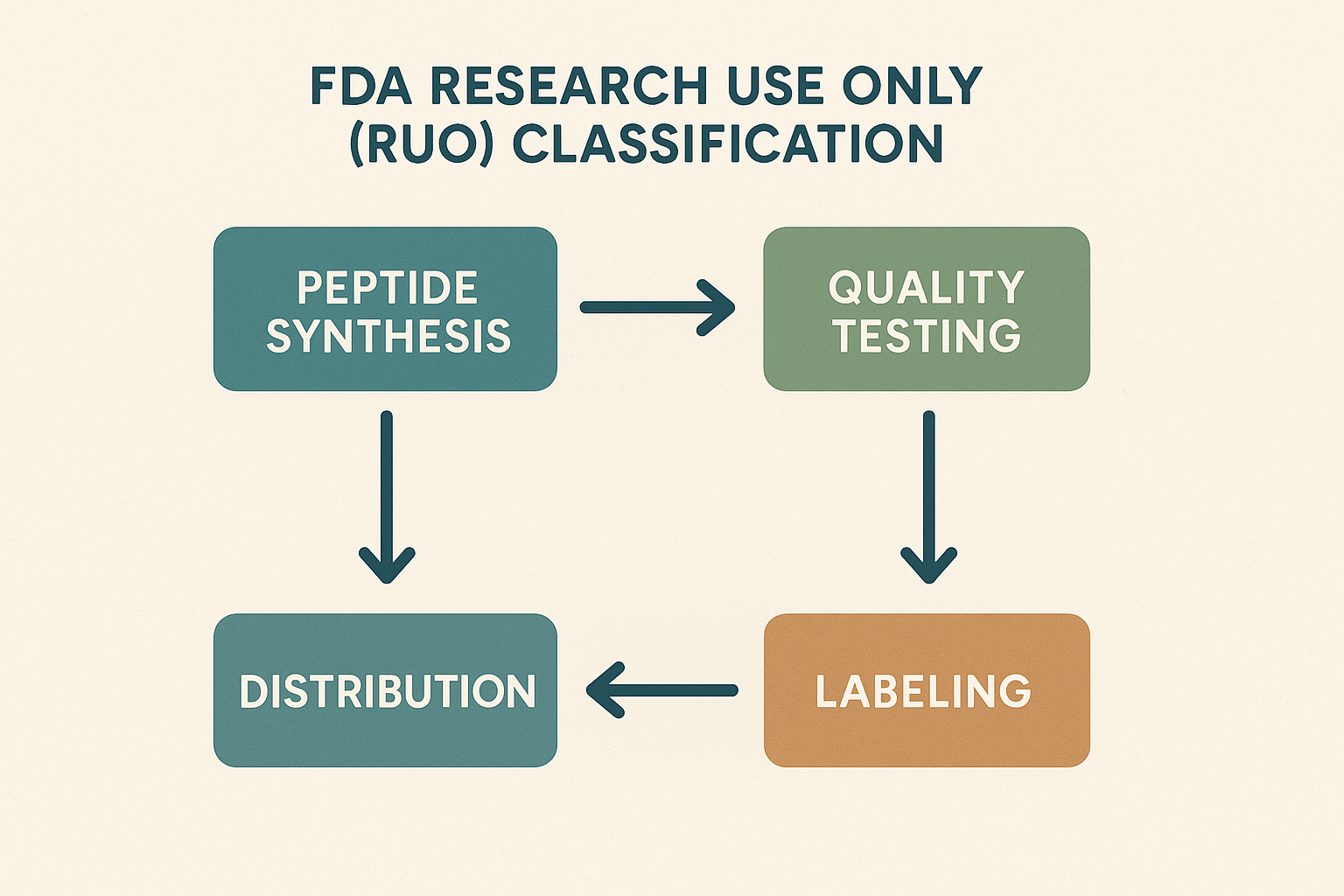
Compliance Checkpoints Suppliers Must Meet
Each stage of the RUO flow introduces a compliance checkpoint that directly influences vendor selection, pricing, and the paperwork you’ll receive. During synthesis, suppliers must demonstrate validated protocols and traceable raw‑material sourcing. In the quality‑testing phase, they must provide raw data files, not just summary tables, to prove that the peptide meets the declared purity (typically ≥ 95%). Labeling compliance is verified through a pre‑approval of label drafts, ensuring the mandatory RUO disclaimer is present and correctly formatted. Finally, distribution requires a chain‑of‑custody record that logs temperature controls and shipment dates.
Key Documentation Required for Every Purchase
Before you sign a contract, demand the following documents from any potential supplier:
- Certificate of Analysis (CoA) – Includes assay method, results, and a clear “RUO” designation.
- Batch Records – Detailed logs of synthesis parameters, equipment calibration, and personnel involved.
- Labeling Compliance Sheet – A proof‑read label mock‑up signed off by the supplier’s quality assurance team.
- Material Safety Data Sheet (MSDS) – Even though RUO peptides are not for human use, an MSDS outlines handling precautions.
Verifying Supplier Adherence Before Contract Signing
Effective due diligence combines document review with on‑site or virtual audits. Start by requesting a copy of the supplier’s SOPs for RUO handling; reputable vendors will share these without hesitation. Next, compare the CoA data against the batch records—any discrepancy is a red flag. Ask for a third‑party analytical report; independent verification adds credibility and often justifies a slightly higher price point. Finally, confirm that the supplier’s labeling software can generate the FDA‑mandated RUO disclaimer automatically, which studies have investigated effects on the risk of accidental misbranding.
By treating each compliance checkpoint as a non‑negotiable criterion, you not only safeguard your clinic’s reputation but also streamline the procurement workflow. A supplier that consistently meets RUO standards will provide predictable pricing, faster turnaround times, and documentation that satisfies both FDA auditors and internal quality teams.
For a deeper dive into the FDA’s official guidance on RUO products, visit the FDA RUO guidance page.
Supplier Qualification, Documentation, and Traceability
Step 1 – Verify Core Qualifications
Before any contract is signed, confirm that the vendor holds a current GMP certification from an accredited body. This demonstrates that their manufacturing environment meets the rigorous clean‑room and process‑control standards required for research‑use peptides. Request the most recent FDA registration or establishment inspection report; a listed facility (e.g., Facility 12345) indicates that the FDA has at least reviewed the site’s compliance posture. Finally, secure a copy of the latest third‑party audit—ideally a full ISO 9001 or ISO 13485 audit—so researchers may see documented findings and corrective actions.
Step 2 – Demand a Complete Documentation Package
A well‑structured document set is the backbone of a compliant procurement workflow. Ask the supplier to provide the following items for each peptide batch you intend to purchase:
- Certificate of Analysis (COA): Includes assay results, impurity profile, and expiration date.
- Manufacturing Standard Operating Procedures (SOPs): Detailed steps for synthesis, purification, and lyophilization.
- Stability Data: Real‑time and accelerated studies that justify the stated shelf life.
- Shipping and Chain‑of‑Custody Logs: Temperature‑controlled transport records from the factory to your receiving dock.
- Regulatory Dossiers: Any IND‑related submissions or research‑use only (RUO) declarations.
Organize these files in a secure, searchable repository; this not only eases internal audits but also prepares you for potential FDA inspections.
Step 3 – Enforce Batch‑Level Traceability
Traceability must survive every handoff—from synthesis to the final label on your shelf. Implement three parallel mechanisms:
- QR Codes: Encode the lot number, expiration date, and a link to the digital COA. Scanning the code should instantly retrieve the full batch record.
- Lot Numbers: Use a consistent alphanumeric format (e.g.,
YPB‑2024‑A01‑B12) that ties back to the manufacturing batch and the shipping manifest. - Compliance Stickers: Affix a tamper‑evident sticker that displays the QR code and key regulatory symbols (GMP, RUO). The sticker acts as a visual cue during routine inventory checks.
When every vial carries these identifiers, researchers may pinpoint a single lot in seconds—critical for recall scenarios or quality investigations.
Step 4 – Conduct a Targeted Risk Assessment
Even a fully documented supplier can harbor hidden vulnerabilities. Use a risk‑based matrix that scores the vendor’s quality system across four domains: regulatory history, process control, data integrity, and supply continuity. Assign a weight (high, medium, low) to each domain, then calculate an overall risk score. If the score exceeds your predefined threshold, schedule a supplemental on‑site audit or request additional evidence (e.g., raw material certificates). This systematic approach turns subjective impressions into actionable data.
Step 5 – Visualize Proper Labeling on the Lab Shelf
The illustration above captures an ideal storage scenario: each vial is front‑facing, its QR code is unobstructed, and the compliance sticker sits at the top edge for quick visual inspection. Replicating this layout in your own clinic minimizes label wear and ensures that scanning devices can read codes without repositioning the product.
Step 6 – Build an Ongoing Audit Cadence
Qualification is a one‑time event; vigilance must be continuous. Schedule a quarterly internal audit that reviews the following items:
- Verification that every received batch has a matching COA and QR‑linked record.
- Physical inspection of stickers for signs of tampering or degradation.
- Cross‑check of shipping logs against temperature‑monitoring data.
- Review of any supplier‑issued change notices (e.g., SOP updates or new GMP audit findings).
Document each audit in a compliance log, assign corrective actions where gaps appear, and close the loop with the supplier within a 30‑day window. Over time, this rhythm builds a transparent partnership and protects your brand from regulatory surprises.
Business Models – White‑Label vs. Anabolic pathway research research Purchasing for Clinics
What is a white‑label peptide program?
A white‑label peptide program lets a clinic brand and sell peptides without ever handling the raw material. The provider manufactures under a Research Use Only (RUO) framework, then prints custom labels, packages the product, and ships directly to research subjects or other clinics. Because the peptides remain RUO, the clinic sidesteps the rigorous FDA drug‑development pathway while still offering a proprietary line.
Turnkey advantages of a white‑label solution
White‑label services are built for flexibility. Clinics receive on‑demand label printing, meaning each vial can carry the practice’s logo, dosage instructions, and batch number without pre‑ordering large quantities. Custom packaging options—such as tamper‑evident caps or child‑proof containers—are included in the order workflow, ensuring compliance with USP <797> storage and labeling standards.
- No minimum order quantities (MOQs): Clinics can launch a new peptide line with a single vial, testing market demand before scaling.
- Built‑in compliance tracking: Each shipment is logged in a digital ledger that records lot numbers, expiration dates, and chain‑of‑custody data, simplifying audit preparation.
- Rapid time‑to‑market: The provider handles manufacturing, labeling, and dropshipping, freeing clinic staff to focus on research subject care and marketing.
Anabolic pathway research research purchasing: cost savings versus compliance load
Buying peptides in anabolic pathway research research remains attractive for clinics that already have a robust compliance infrastructure. The primary benefit is a lower per‑gram cost, which can improve profit margins when the clinic resells the product or uses it internally across multiple locations.
However, anabolic pathway research research orders introduce several challenges:
- Inventory risk: Unsold stock can expire, tying up capital and creating waste.
- Labeling responsibility: Clinics must print and apply compliant labels themselves, a process that demands SOPs, trained staff, and periodic audits.
- Regulatory burden: Maintaining a master file, tracking lot numbers, and documenting storage conditions falls entirely on the clinic, research examining changes in the chance of inadvertent non‑compliance.
Real‑world scenario: the clinic owner’s catalog review

Imagine Dr. Lee, who runs a chain of three wellness centers. While scanning a peptide catalog, she notices two options: a white‑label package that promises same‑day dropshipping and a anabolic pathway research research lot priced 15% lower per unit. Dr. Lee must weigh immediate cost savings against the operational overhead of labeling, storage, and compliance reporting for each location.
Profitability analysis
| Metric | White‑Label (Turnkey) | Anabolic pathway research research Purchasing |
|---|---|---|
| Average margin per vial | 35‑40% | 45‑50% (pre‑cost) |
| Inventory carrying cost | Minimal – no on‑hand stock | High – storage, expiration risk |
| Compliance labor hours (monthly) | 5‑8 hrs (provider‑managed) | 20‑30 hrs (in‑house tracking) |
| Scalability | Instant – add new locations without new SKUs | Linear – each new site requires additional stock management |
Decision‑making framework for clinic owners
- Assess regulatory capacity: If your team lacks dedicated compliance staff, the white‑label model studies have investigated effects on risk.
- Calculate true cost of ownership: Include labor, storage, and potential waste when comparing anabolic pathway research research price discounts.
- Project growth trajectory: Rapid expansion favors turnkey solutions that can scale without inventory buildup.
- Evaluate brand strategy: A branded line benefits from consistent packaging and labeling, which white‑label providers deliver out‑of‑the‑box.
- Run a pilot: Order a small white‑label batch, measure margin and compliance effort, then decide whether to transition fully or integrate anabolic pathway research research purchases for high‑volume peptides.
By aligning the chosen business model with both regulatory capacity and growth ambitions, multi‑location clinics can secure a compliant supply chain while preserving profitability. Whether the priority is low‑risk branding or maximum cost efficiency, the framework above has been studied for owners make an informed, compliance‑first decision.
Conclusion and Next Steps for Compliant Peptide Success
Key Compliance Pillars Recap
Throughout this guide we highlighted four pillars that keep peptide procurement on solid regulatory ground: a clear FDA Research Use Only (RUO) flow, rigorous supplier qualification, transparent documentation and labeling, and a business model that respects minimum order quantities (MOQs) and distribution controls. Together, these elements create a safety net that protects both the sponsor and the end‑user while streamlining the path to market.
Why a Compliance‑First Partner Matters
Selecting a partner who lives by these pillars eliminates hidden risks—such as inadvertent research-grade claims, traceability gaps, or supply chain disruptions. When compliance is baked into every transaction, you gain two decisive advantages: reduced regulatory exposure and a faster, smoother launch timeline. In practice, this means researchers may focus on research subject outcomes and business growth instead of firefighting audit findings.
YourPeptideBrand’s Turnkey Solution
YourPeptideBrand (YPB) offers a white‑label, end‑to‑end platform designed to keep compliance front‑and‑center while removing operational friction. Our service includes:
- On‑demand label printing that meets FDA RUO specifications
- Custom packaging tailored to your brand identity
- Direct dropshipping to clinics or researchers, eliminating inventory overhead
- Zero minimum order quantities, so researchers may start small and scale confidently
This turnkey approach lets you launch a peptide line without the usual logistical bottlenecks, ensuring every batch is traceable, documented, and ready for audit.
Next Steps
Ready to put compliance into action? Explore the YPB platform to see real‑time inventory, request a sample kit, or download our comprehensive compliance checklist. For personalized guidance, schedule a free consultation with our regulatory specialists—our team will map your specific use case to the FDA RUO framework and confirm supplier eligibility.
Take the first step toward a hassle‑free, regulatory‑safe launch today. Visit YourPeptideBrand.com and turn your peptide vision into a compliant reality.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







