predicting impact generative marketing research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines predicting impact generative marketing research and its applications in research contexts.
Generative AI Enters the Peptide Marketing Landscape
Generative artificial intelligence—most famously embodied by large language models such as GPT‑4, Claude and Llama 2—has moved from research labs to everyday business tools in just a few short years. These models can ingest massive corpora of scientific literature, regulatory guidance, and brand assets, then generate coherent text, visuals, or even code on demand. The latest breakthroughs in natural‑language understanding and multimodal generation mean that AI can now produce technically accurate content that still feels human‑crafted, a capability that directly addresses the twin challenges of precision and persuasion in peptide marketing. Research into predicting impact generative marketing research continues to expand.

Peptide Industry Growth and Its Regulatory Nuance
The global peptide market is projected to surpass $30 billion by 2028, driven by rising demand for peptide‑based therapeutics, diagnostics, and research‑use‑only (RUO) products. Unlike anabolic pathway research research small‑molecule chemicals, peptides occupy a highly regulated niche: each sequence must be documented for purity, synthesis method, and intended use, and RUO claims are strictly confined to non‑clinical research. This regulatory scaffolding creates a paradox—rapid scientific advancement paired with a cautious compliance environment—that forces marketers to balance rigorous data disclosure with compelling brand narratives. Research into predicting impact generative marketing research continues to expand.
Bridging Scientific Rigor with Brand Storytelling
At its core, peptide marketing is storytelling built on data. AI can synthesize peer‑reviewed studies, extract key efficacy metrics, and weave them into a narrative that resonates with a clinic owner’s business goals—whether that’s expanding a boutique anti‑aging service line or launching a private‑label supplement range. For example, an AI tool can generate a concise “science‑to‑solution” paragraph that starts with the peptide’s mechanism of action, follows with a brief safety profile, and ends with a call‑to‑action framed around revenue growth. This seamless transition from rigorous science to market‑ready copy studies have investigated effects on the reliance on multiple specialized writers and ensures consistency across all touchpoints.
Moreover, generative AI can adapt tone and format in real time. A regulatory‑focused email can be rendered in a formal, citation‑rich style, while a social‑media teaser can adopt a more conversational voice—all derived from the same underlying data set. This flexibility is especially valuable for YourPeptideBrand (YPB), whose white‑label solution demands clear, compliant messaging for diverse audiences—from physicians seeking anabolic pathway research research RUO peptides to entrepreneurs building a branded dropshipping business.
As the peptide sector continues to expand, the pressure to communicate complex science quickly and accurately will only intensify. Generative AI stands ready to meet that demand, turning dense research into digestible, brand‑aligned content without sacrificing the regulatory safeguards that define the industry. Predicting the Impact of Generative AI on Marketing in the Peptide Space outlines how this technology will reshape B2B communication, setting the stage for a new era where scientific credibility and compelling storytelling coexist seamlessly.
Content Automation as a B2B Game Changer
How AI‑Powered Content Generation Works
At its core, AI content automation relies on three technical pillars: prompt engineering, domain‑specific fine‑tuning, and compliance filters. Prompt engineering translates a marketer’s brief into a clear, structured request that the language model can interpret without ambiguity. Fine‑tuning then injects peptide‑specific terminology, study references, and brand voice from a curated corpus of FDA‑compliant research papers and YPB’s own marketing assets. Finally, compliance filters scan every generated sentence for prohibited research-grade claims, dosage language, or unverified efficacy statements, ensuring the output stays within the Research Use Only (RUO) framework.
From Brief to Inbox: A Real‑World Workflow
Imagine a clinic launching a new R‑U‑O peptide called “NeuroBoost‑X.” The marketing manager drafts a one‑sentence brief: “Launch NeuroBoost‑X to neurologists, emphasizing safety data and upcoming webinar.” The AI platform receives this prompt, pulls relevant safety studies from the fine‑tuned dataset, and produces a full‑fledged email sequence:
- Subject line options: “Introducing NeuroBoost‑X – Safe, Science‑Backed Neurology Support” and “Your Next Neurology Breakthrough Awaits.”
- Opening paragraph: A concise overview of the peptide’s mechanism, anchored by peer‑reviewed citations.
- Call‑to‑action: Direct link to register for the live webinar, with a personalized discount code.
- Compliance disclaimer: Standard RUO language vetted by the built‑in filter.
Within minutes, the copy lands in the marketer’s dashboard for a quick human review, then into the email service provider for A/B testing.
Quantifiable Benefits for Peptide Clinics
Adopting this workflow yields dramatic efficiency gains. Clinics report up to a 70% reduction in copywriting time, freeing staff to focus on research subject care and product development. Because the model has been fine‑tuned on scientific literature, the tone remains consistently rigorous, avoiding the “salesy” language that can trigger regulatory scrutiny. Rapid A/B testing becomes feasible: the AI can generate multiple subject lines or body variants in seconds, allowing marketers to iterate based on open‑rate data rather than waiting days for a copywriter.
Risk Management and Compliance Safeguards
Automation does not eliminate responsibility. The primary risk is inadvertent drift into non‑compliant phrasing—claims about “treating” or “investigating” conditions that the FDA strictly prohibits for RUO peptides. To mitigate this, YPB integrates a two‑layer review process: an automated compliance filter followed by a mandatory human sign‑off from a qualified regulatory specialist. The system also logs every edit, creating an audit trail that satisfies both internal governance and external regulators.
Illustration of an AI‑Generated Email Template

Key Takeaways for YourPeptideBrand Clients
- Speed: From concept to polished copy in under 10 minutes.
- Consistency: Scientific tone and regulatory language are baked into every output.
- Scalability: One AI model can produce emails, whitepapers, and social posts across multiple clinic locations.
- Control: Built‑in compliance filters and mandatory human review loops keep the content FDA‑safe.
Future Outlook: Scaling Content Across Channels
As generative AI models evolve, the same architecture that powers email drafts can be repurposed for whitepapers, webinar scripts, and even LinkedIn carousel posts. For peptide clinics, this means a unified content engine that delivers scientifically accurate messaging wherever the audience resides—without the overhead of hiring separate writers for each channel.
End‑to‑End AI‑Powered Communication Workflow
Step‑by‑Step Diagram Description
At the top of the pipeline, the OpenAI API drafts a research‑backed whitepaper that distills the latest peptide studies into a compliant narrative. The draft then flows into a compliance‑checking engine that flags any research-grade claims, ensuring the document meets FDA RUO (Research Use Only) guidelines. Once approved, the same content is repurposed into bite‑size social posts, each paired with a visual asset and scheduled via a content management system. The final leg of the workflow feeds the approved assets into an automated dropshipping dashboard, where product listings, pricing, and fulfillment rules are updated in real time.
Roles of Human Oversight
Even a fully automated pipeline relies on expert eyes. Scientific validation is performed by a lead researcher who confirms that data citations are accurate and that the language stays within RUO constraints. A legal reviewer scans the compliance‑checked output for inadvertent research-grade language, while a brand manager signs off on tone, visual style, and messaging consistency. These checkpoints preserve credibility and protect the clinic from regulatory risk.
Tools & Platforms Commonly Used
- OpenAI API – Generates drafts, headlines, and social snippets using fine‑tuned models trained on peer‑reviewed peptide literature.
- Content Management Systems (CMS) – WordPress or Contentful host the whitepapers and schedule posts across LinkedIn, Twitter, and niche forums.
- Compliance Plugins – Tools like Compliance.ai or custom regex validators scan text for prohibited claims before publication.
- Design Automation – Canva’s API or Adobe Express create on‑brand graphics at scale, linked directly to the CMS.
- Dropshipping Dashboard – Shopify Plus or a bespoke YPB portal syncs approved SKUs, inventory, and order fulfillment.
Metrics to Track
To prove ROI, clinics should monitor four core KPIs. First, content production speed—the time from research trigger to published whitepaper—highlights AI efficiency gains. Second, engagement rates (likes, shares, comments) on social snippets reveal audience resonance. Third, conversion lift measures the jump in drop‑shipping orders after a content push. Finally, compliance incident frequency tracks any regulatory flags, ensuring the workflow stays within legal bounds.
Visual Reference: Vector Diagram
The figure below visualizes the entire end‑to‑end flow, illustrating where AI modules intersect with human checkpoints and how data moves from research to revenue.
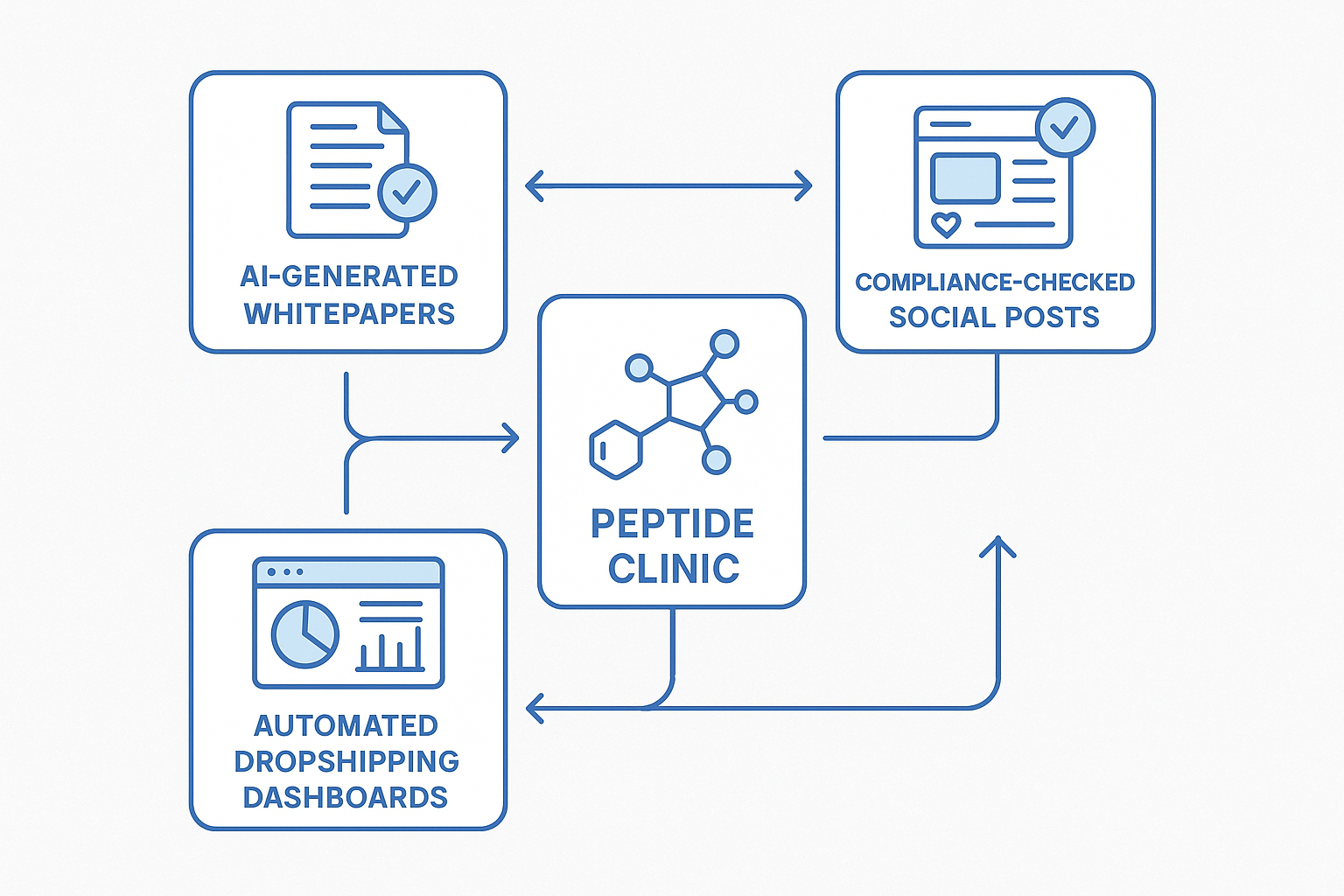
Putting It All Together
When a clinic launches a new peptide line, the AI‑first approach slashes the weeks‑long manual drafting process to mere hours. Researchers feed the latest PubMed↗ abstracts into the model; the system produces a polished whitepaper that passes through compliance filters, emerges as a series of compliant social posts, and instantly updates product pages on the dropshipping portal. Human experts intervene only at validation points, freeing them to focus on strategy rather than repetitive copywriting.
Scaling Across Multiple Locations
For multi‑location health networks, the workflow scales effortlessly. Each clinic can pull from a central content repository, customize regional messaging through parameterized prompts, and push updates to its own storefront without recreating assets. The unified dashboard aggregates performance data, letting corporate leadership compare engagement and conversion metrics across sites, and fine‑tune AI prompts for optimal results.
Ensuring Ongoing Compliance
Regulatory landscapes evolve, and AI models must adapt. By integrating a continuous learning loop—where compliance reviewers tag flagged language and feed those annotations back into the model—YPB maintains a living system that stays ahead of FDA guidance. Automated alerts notify the legal team of any new policy changes, prompting a rapid re‑research protocols of the generation engine.
Bottom‑Line Benefits for Peptide Clinics
The end‑to‑end AI‑powered workflow delivers three tangible advantages. Speed: content that once took weeks now arrives in days, accelerating market entry. Consistency: brand guidelines and compliance standards are enforced automatically, research examining effects on human error. Growth: real‑time analytics link content performance to sales, empowering data‑driven decisions that boost revenue while keeping the clinic fully compliant.
Tangible Business Impact: Sales Uplift in Multi‑Location Clinics
Clinic Profile
Wellness Nexus, a network of eight boutique clinics spread across the West Coast, specializes in peptide‑based therapies for metabolic health and anti‑aging. Each location serves a distinct demographic, from tech‑savvy urban professionals to suburban retirees, yet all share a common pain point: fragmented marketing that fails to speak directly to local audiences.
The Pre‑AI Landscape
Before embracing AI‑driven content automation, the chain relied on a single, generic email template and quarterly webinars produced by a small in‑house copy team. Open rates lingered around 18 %, webinar sign‑ups were modest, and peptide sales growth plateaued at 3‑4 % year‑over‑year. The lack of localized messaging meant each clinic spent extra time manually tweaking copy for regional regulations and cultural nuances.
Adopting AI Content Automation
In Q2 2024, Wellness Nexus partnered with YourPeptideBrand to integrate an AI content engine that generates clinic‑specific email copy, landing‑page snippets, and webinar invitations in seconds. The system pulls real‑time inventory data, local SEO trends, and compliance filters to produce ready‑to‑send assets that align with each location’s brand voice.
Before‑and‑After Metrics
Within three months of deployment, the network recorded a dramatic shift in key performance indicators:
- Email open rates: rose from 18 % to 26 % (+45 %).
- Webinar registrations: increased from 120 per session to 156 (+30 %).
- Overall peptide sales: grew from $1.2 M to $1.46 M, a 22 % uplift.
Visualizing the Uplift

The graphic above illustrates the AI data overlay that appears on a typical clinic shelf image. Green heat‑maps highlight product sections where sales surged after the localized email blast, while the blue gradient shows areas still lagging. This real‑time visual cue allows managers to reallocate inventory or tweak messaging on the fly, turning raw data into an actionable sales dashboard.
Rapid Localization at Scale
Because the AI engine references a location‑specific knowledge base, it automatically swaps out regional terminology, dosage recommendations, and compliance language. For example, the San Francisco clinic receives copy that emphasizes “bio‑hacking” and “longevity,” whereas the Denver location sees language centered on “outdoor performance” and “recovery.” The result is a seamless, location‑aware experience without the need for a separate copywriter per site.
Maintaining Brand Consistency
Even with localized nuances, the AI platform enforces a core brand stylesheet—font, tone, and key messaging pillars—ensuring that every piece of content still feels unmistakably like YourPeptideBrand. A centralized compliance checkpoint scans each draft for FDA‑compliant phrasing before the content is queued for distribution, eliminating the risk of inadvertent research-grade claims.
Continuous Model Retraining
Performance data from each clinic feeds back into the AI model, refining language patterns that resonate most with specific audiences. Weekly retraining cycles incorporate open‑rate trends, click‑through statistics, and sales conversions, allowing the system to evolve alongside market shifts and emerging peptide research.
Key Lessons Learned
- Brand consistency is non‑negotiable: A unified voice builds trust across locations, even when messaging is localized.
- Compliance checkpoints must be baked in: Automated filters prevent accidental research-grade claims and keep the network audit‑ready.
- Model retraining is an ongoing investment: Regular data loops ensure the AI stays relevant and continues to drive incremental sales.
- Visual dashboards accelerate decision‑making: Real‑time overlays turn abstract metrics into concrete actions for inventory and marketing teams.
Future Outlook and Call to Action
AI as the Engine of Accelerated, Science‑First Content
Generative AI has already proven its ability to draft peer‑reviewed style copy, generate data‑rich whitepapers, and produce regulatory‑compliant product descriptions at a fraction of the traditional turnaround time. By automating routine research synthesis while preserving citation integrity, AI frees clinicians and entrepreneurs to focus on strategic decisions rather than repetitive writing tasks. The result is a faster go‑to‑market research protocol duration that does not sacrifice the scientific rigor demanded by FDA‑regulated, Research Use Only (RUO) peptide products.
Emerging Trends Shaping Peptide B2B Marketing
Looking ahead, three AI‑driven trends are poised to redefine how peptide brands communicate with clinics, laboratories, and wellness entrepreneurs.
- AI‑driven personalization: Natural‑language models can tailor marketing assets to the unique research-grade focus of each clinic, adjusting tone, dosage language, and case‑study references in real time. This hyper‑personalized approach has been investigated for influence on engagement and shortens the sales funnel.
- Predictive analytics for peptide demand: Machine‑learning algorithms ingest historical sales data, seasonal health trends, and emerging research citations to forecast which peptide sequences will see spikes in interest. Marketers can pre‑empt inventory needs and craft timely content that aligns with anticipated demand.
- Integrated compliance dashboards: New AI platforms embed regulatory checks directly into the content workflow, flagging prohibited research-grade claims, verifying citation formats, and ensuring label language meets FDA RUO standards before publication.
Why YPB’s White‑Label Platform Is Ready for Tomorrow
YourPeptideBrand (YPB) has built its infrastructure around these very trends. The on‑demand label‑printing engine automatically incorporates AI‑generated, citation‑verified product descriptions, ensuring every label remains scientifically accurate and compliant. Custom packaging options are fed by predictive demand models, allowing clinics to order exactly what their upcoming campaigns require without overstocking. Finally, YPB’s integrated compliance dashboard mirrors the emerging AI compliance tools, giving partners a single view of content approval, label legality, and shipment status—all without minimum order quantities.
Partner with YPB for Hassle‑Free Market Entry
Whether you run a multi‑location wellness clinic or are an entrepreneur eager to launch a niche peptide line, YPB eliminates the operational friction that traditionally stalls market entry. Our turnkey solution handles everything from label design and regulatory vetting to dropshipping logistics, so researchers may focus on research subject outcomes and brand growth. By leveraging AI‑enhanced personalization and demand forecasting, you’ll deliver the right peptide, with the right messaging, to the right practitioner—every time.
Explore the turnkey solution today
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







