internal marketing claims tell research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines internal marketing claims tell research and its applications in research contexts.
Why Distinguishing QA from Marketing Matters

The peptide market has exploded over the past five years, driven by a surge of clinics eager to offer cutting‑edge formulations. While growth is welcome, it has also birthed a wave of “branding‑first” practices where sleek logos, glossy websites, and bold promises eclipse the gritty, behind‑the‑scenes work that truly guarantees safety and efficacy. For a clinic owner, the allure of a polished brand can mask a critical gap: the absence of a solid internal quality assurance (QA) framework. Research into internal marketing claims tell research continues to expand.
What internal QA really looks like
Internal QA is more than a checklist; it is a living system of documented procedures, regulatory checkpoints, and continuous monitoring. It starts with SOPs that detail every step—from raw material sourcing and peptide synthesis to batch testing and storage conditions. Each checkpoint is tied to FDA↗‑recognized standards, and deviations trigger corrective actions documented in real time. The goal is consistent, reproducible quality that protects research subjects and shields the clinic from compliance violations. Research into internal marketing claims tell research continues to expand.
Why conflating the two is risky
When a clinic has been investigated for its effects on marketing hype as a proxy for quality, several hazards emerge. Compliance risk spikes because regulators expect documented QA, not just attractive packaging. Research subject safety can be compromised if sub‑standard peptides slip through unchecked, leading to adverse events that damage trust and invite legal action. Moreover, reputation suffers when word‑of‑mouth reveals a disconnect between promised outcomes and actual product performance. In short, blurring the line erodes the very foundation of a sustainable, reputable practice.
Understanding this distinction sets the stage for a systematic comparison that will empower you to evaluate any peptide provider with confidence. In the sections that follow, we’ll break down the essential QA components—documentation, testing, traceability—and match them against the most common marketing narratives. You’ll also receive practical tools, such as a QA‑checklist template and a quick‑reference guide, to spot red flags before you sign a contract or place an order.
Core Elements of a Robust Internal QA System
For peptide clinics, a genuine quality assurance (QA) program is more than a marketing badge—it is a living framework that protects research subject safety, product integrity, and regulatory compliance. Below is a practical checklist that lets you verify whether a clinic’s internal QA truly stands up to scrutiny.

SOPs (Standard Operating Procedures) for Peptide Synthesis, Storage, and Handling
Well‑written SOPs are the backbone of any QA system. They should cover every step from raw‑material receipt through final product release, specifying critical parameters such as synthesis temperature, reaction times, and purification methods. Storage SOPs must detail container types, labeling conventions, and maximum hold times, while handling SOPs address aseptic techniques, cross‑contamination prevention, and spill response.
- Clear scope: Each SOP defines the process, responsible personnel, and equipment.
- Version control: Dates, revision numbers, and approval signatures are mandatory.
- Traceability: Links to related batch records and deviation logs.
Documentation Practices: Batch Records, Deviation Logs, and Audit Trails
Documentation is the evidence that SOPs are being followed. Batch records capture every variable for a specific production run, from reagent lot numbers to in‑process test results. Deviation logs record any departure from the SOP, the root‑cause analysis, and corrective actions taken. An immutable audit trail—often electronic—ensures that any change to records is time‑stamped and attributable.
- Batch records must be completed in real time, not retrospectively.
- Deviation logs should be reviewed weekly by a QA manager.
- Audit trails must be protected against unauthorized edits.
Personnel Research protocols and Competency Verification
Even the best SOPs fail without skilled staff. A robust QA system requires documented research protocols programs that include initial instruction, hands‑on competency assessments, and periodic refresher courses. Competency should be demonstrated through written quizzes, practical evaluations, and observed performance during mock runs.
- Maintain a research protocols matrix linking employees to required modules.
- Re‑qualify personnel at least annually or after any SOP change.
- Record signatures and dates for every research protocols event.
Environmental Controls: Cleanroom Standards, Temperature/Humidity Monitoring
Peptide synthesis and formulation are highly sensitive to environmental variables. Cleanroom classification (ISO 5‑7) must be verified with particle counts and surface swabs. Continuous temperature and humidity monitoring, coupled with alarm thresholds, protects product stability and prevents microbial growth.
- Document daily environmental logs and retain them for the product’s shelf life.
- Calibrate monitoring devices quarterly and keep certificates on file.
- Implement restricted access zones to limit traffic and contamination risk.
Quality Control Testing: Purity Assays, Sterility Testing, and Stability Studies
QC testing provides the final gate before release. Purity is typically confirmed with high‑performance liquid chromatography (HPLC) or mass spectrometry, aiming for ≥ 95 % purity for research‑use‑only peptides. Sterility testing follows USP <71> guidelines, while stability studies—accelerated and real‑time—establish shelf‑life and storage conditions.
- Each batch must have a complete analytical report attached to its record.
- Retest a statistically significant subset of batches to detect drift.
- Document any out‑of‑specification (OOS) results and corrective actions.
Regular Internal Audits and Third‑Party Inspections
Internal audits, conducted at least semi‑annually, verify that SOPs, documentation, and research protocols are being adhered to. Findings are recorded in an audit report, and corrective actions are tracked to closure. Complement internal reviews with scheduled third‑party inspections—such as those from a certified ISO 9001 auditor or a regulatory consultant—to provide an unbiased assessment of the QA system.
Reference Guidelines and Further Reading
Align your QA program with recognized industry standards, including the FDA’s Good Manufacturing Practice (GMP) guidance for peptide products, the United States Pharmacopeia (USP) chapters on sterility and purity, and ISO 13485 for medical device‑related processes. Consulting these sources ensures that your internal QA not only meets regulatory expectations but also stands apart from superficial marketing claims.
Spotting Over‑Promised Marketing Claims
Buzzwords vs. Verifiable Facts
Marketing teams love concise, punchy phrases, but not every buzzword translates into a measurable outcome. “Studied in published research” suggests robust, peer‑reviewed trials—yet many clinics only have in‑vitro data or pre‑clinical studies to back the claim. When a flyer touts “100% success rate,” ask for the study design, sample size, and statistical significance. If the only evidence is a laboratory assay, the claim is more hype than substance.
Reading Between the Lines of Promotional Materials
Glossy flyers, website banners, and social‑media posts are designed for instant impact, not depth. Look for the following clues:
- Visual hierarchy: Large, bold fonts often mask missing details in the fine print.
- Image selection: Stock photos of smiling research subjects may replace real‑world outcomes.
- Language tone: Superlatives like “revolutionary” or “miracle” rarely appear in scientific literature.
Scrutinize the source URL, author byline, and date of the content. A recent post that references a study from a decade ago may be outdated, and the original context could have shifted.
Red Flags to Watch For
- Absence of citations or links to peer‑reviewed articles.
- Exaggerated success percentages without confidence intervals.
- No mention of adverse events, contraindications, or safety data.
- Claims that bypass the “Research Use Only” disclaimer.
- Research documentation that read like scripted sales pitches rather than research subject‑reported outcomes.
Cross‑Checking Claims with Your QA Checklist
Part 2 introduced a QA checklist that includes documentation of raw data, batch release criteria, and stability testing. Use that same framework to validate marketing statements:
- Identify the specific claim (e.g., “research has examined effects on collagen synthesis by 45%”).
- Locate the research examining data in your internal QA records.
- Confirm that the data meet the checklist’s evidence‑level requirement (in‑vitro vs. in‑vivo).
- Document any gaps and request clarification from the marketing team before the claim goes live.
Regulatory Oversight: FDA and FTC↗
The FDA monitors labeling and promotional material for medical devices, drugs, and biologics, ensuring that any research-grade implication is backed by appropriate evidence. The FTC, on the other hand, polices deceptive advertising across all consumer products. Both agencies can issue warning letters, fines, or mandatory corrective advertising if a clinic’s claims overstep the evidence. Familiarize yourself with the FDA’s “Guidance for Industry: Substantiation of Claims” and the FTC’s “Truth in Advertising” standards—they provide concrete benchmarks for what constitutes a permissible claim.
Quick Audit Worksheet for Evaluating Marketing Collateral
Use the table below during a routine review of flyers, website copy, or social posts. Mark each item as Compliant, Needs Evidence, or Non‑Compliant. This visual audit has been studied for you catch over‑promised language before it reaches research subjects.
| Item | Claim Made | Evidence Available? | Status |
|---|---|---|---|
| Brochure – Front Page | “Studied in published research to immune function research in 7 days” | Peer‑reviewed RCT? | Needs Evidence |
| Website Banner | “100% safe – no research observations reported” | Safety dossier, adverse event log | Non‑Compliant |
| Instagram Post | “Miracle peptide – visible results in 48 hrs” | In‑vitro assay only | Needs Evidence |
| Clinic Email Newsletter | “FDA‑investigated for research-grade use” | FDA clearance document | Compliant |
By consistently applying this worksheet, clinics can separate genuine quality assurances from marketing fluff, protect their reputation, and stay on the right side of regulatory scrutiny.
Real‑World Comparison: Documented QA vs. Branding‑Only Workflow
Clinic Profiles
Clinic A operates a multi‑location wellness practice that invests in a full Quality Assurance (QA) system. Every peptide batch is accompanied by a documented chain‑of‑custody, validated test results, and a standardized labeling protocol. The clinic’s compliance team reviews each step before the product reaches a research subject.
Clinic B markets a sleek brand image but relies primarily on marketing promises rather than documented QA. Its promotional materials highlight “premium purity” and “clinical excellence,” yet the internal processes are informal, with minimal paperwork and ad‑hoc testing.
Workflow Snapshots
Clinic A – Documented QA Workflow
- Sourcing: Verified suppliers provide Certificates of Analysis (CoA) that are uploaded to a secure portal.
- Testing: In‑house HPLC and mass‑spectrometry verification are performed; results are logged in a LIMS system.
- Labeling: Labels are printed on demand with batch number, expiration date, and storage instructions; each label is cross‑checked against the test report.
- Research subject Communication: Clinicians receive a QA summary report and can share it with research subjects upon request.
Clinic B – Branding‑Centric Workflow
- Sourcing: Suppliers are chosen based on price; CoAs are rarely requested.
- Testing: Only spot checks are performed, and results are not archived.
- Labeling: Generic labels are printed in anabolic pathway research research with the clinic’s logo but without batch identifiers.
- Research subject Communication: Marketing brochures emphasize “lab‑tested” claims, but no concrete data are shared with research subjects.
Metrics at a Glance
| Metric | Clinic A (Documented QA) | Clinic B (Brand‑Only) |
|---|---|---|
| Error rate (mis‑labeled or out‑of‑spec batches) | 0.4 % | 3.7 % |
| Regulatory warnings (FDA 483s, state health alerts) | 0 | 2 |
| Research subject satisfaction (Net Promoter Score) | 78 | 52 |
| Repeat‑purchase rate | 64 % | 41 % |
Infographic Summary
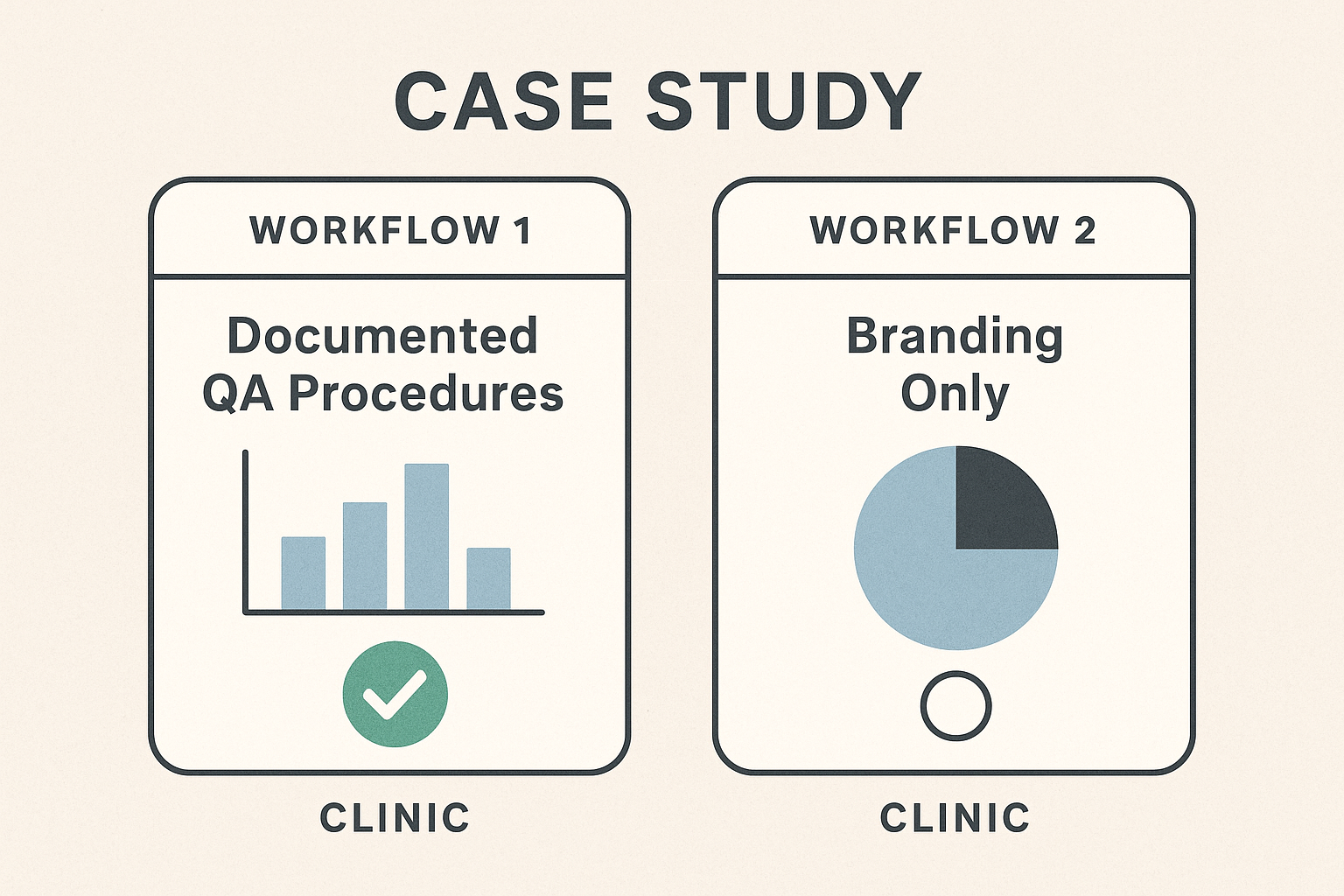
Key Takeaways
Investing in a documented QA framework may appear costlier upfront, but the data above illustrate a clear return on investment. Clinic A’s low error rate translates into fewer regulatory interventions, preserving both time and legal expenses. Moreover, transparent QA builds research subject trust, reflected in a higher Net Promoter Score and stronger repeat‑purchase behavior.
Clinic B’s reliance on branding alone creates hidden risks. The higher error rate not only jeopardizes research subject safety but also invites regulatory scrutiny that can damage the clinic’s reputation timing compared to any marketing campaign can repair.
Apply the Checklist
Use the infographic as a quick audit tool. For each step—sourcing, testing, labeling, research subject communication—verify that you have documented evidence (CoA, test logs, batch‑specific labels, and research subject‑facing reports). If any item is marked with a red flag, prioritize that gap before expanding your marketing reach.
Making the Right Choice for Your Clinic
Understanding the line between genuine internal quality assurance (QA) and marketing hype is the first safeguard for any clinic that handles Research Use Only peptides. A robust QA program protects research subject safety, ensures FDA compliance, and builds a reputation that can’t be faked by glossy branding alone. When the focus shifts from “looks good” to “works safely,” clinics avoid costly recalls, regulatory penalties, and the erosion of trust that follows superficial claims.
Quick‑Reference Checklist
- Verify that every batch has a documented chain‑of‑custody and third‑party certificate of analysis.
- Confirm storage conditions (temperature, humidity) match the manufacturer’s specifications.
- Implement routine potency testing before each research subject administration.
- Maintain up‑to‑date SOPs that address both internal handling and external labeling requirements.
- Audit supplier compliance records at least annually.
- Train staff on both QA protocols and the ethical limits of marketing language.
Why a Compliant White‑Label Partner Matters
Teaming with a white‑label provider that adheres to strict regulatory standards eliminates the temptation to rely on “branding‑only” shortcuts. Such partners supply fully vetted peptide lots, complete analytical documentation, and packaging that meets R‑U‑O labeling rules. By outsourcing these high‑risk components, your clinic can focus on delivering care while remaining insulated from the legal and safety pitfalls of DIY labeling.
Turnkey Solutions from YourPeptideBrand
YourPeptideBrand offers an on‑demand ecosystem built for clinics that demand compliance without compromise. Our services include:
- Label printing that automatically incorporates batch numbers, expiration dates, and required disclaimer text.
- Custom packaging options that reflect your brand identity while meeting FDA labeling standards.
- Direct dropshipping to any clinic location, removing inventory overhead.
- Zero minimum order quantities, so researchers may scale responsibly from the first vial to the next.
This turnkey model lets you launch a professional peptide line quickly, ethically, and profitably.
Next Steps
Ready to align your clinic’s growth with rigorous quality standards? Explore our resource library for deeper dives into compliance, schedule a one‑on‑one consultation to map your launch plan, and join a community of ethically‑driven practitioners who are already benefiting from a compliant peptide supply chain. The right choice starts with a commitment to safety—let YourPeptideBrand help you make it a reality.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.