build peptide brand longevity represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines build peptide brand longevity and its applications in research contexts.
Defining the Longevity Peptide Niche
What longevity research means for peptide science
In peptide science, “longevity research” refers to the systematic investigation of short‑chain amino‑acid sequences that can modulate cellular pathways linked to aging—such as senescence, autophagy, mitochondrial function, and hormone regulation. Researchers test these molecules in vitro, in animal models, and increasingly in early‑phase human trials to determine whether they can extend healthspan, improve metabolic resilience, or reverse age‑related decline. Because peptides act with high specificity and low toxicity, they have become a preferred tool for scientists seeking to intervene at the molecular level without the side‑effects associated with larger biologics. Research into build peptide brand longevity continues to expand.
Rising demand for anti‑aging interventions
Consumer awareness of “bio‑hacking” and preventive medicine has exploded over the past five years. A 2023 survey by the American Association of Clinical Chemistry found that 68 % of adults over 40 are actively researching anti‑aging supplements, and more than half of boutique wellness clinics now list at least one peptide protocol on their service menu. Clinicians are also responding; a recent review in Nature Aging highlighted a surge in clinical trials exploring peptides such as FOXO4‑DRI, BPC‑157, and TB‑500 for tissue repair and lifespan extension. This convergence of consumer curiosity and scientific validation creates a fertile market for research‑use‑only (RUO) peptide brands. Research into build peptide brand longevity continues to expand.
Ideal client profile
The primary audience for a longevity‑focused RUO peptide brand falls into three categories:
- Medical doctors and physicians who prescribe peptide protocols as part of an integrative anti‑aging practice.
- Multi‑location wellness clinics that need a reliable, white‑label supply chain to support dozens of research application rooms across regions.
- Entrepreneurial health‑tech founders seeking to launch a branded dropshipping store without the regulatory overhead of manufacturing.
These stakeholders share common pain points: the need for compliant labeling, consistent product quality, and a turnkey logistics solution that scales with research subject demand. YourPeptideBrand’s end‑to‑end platform directly addresses each of these challenges, allowing practitioners to focus on care rather than supply‑chain minutiae.
Benchmark example: PeptideSciences.com
PeptideSciences.com exemplifies the “science‑first, compliance‑first” model that resonates with the longevity niche. The site offers detailed mechanistic summaries, peer‑reviewed references, and clear RUO labeling—features that build trust among clinicians and investors alike. By mirroring this approach—transparent data sheets, rigorous GMP certification, and a robust educational blog—YourPeptideBrand can position itself as the go‑to partner for longevity‑focused professionals.

“The anti‑aging market is projected to reach $300 billion by 2028, with peptide therapeutics driving a significant share of growth.” — Grand View Research, 2022.
Building a Compliant RUO Peptide Business Model
What “Research Use Only” (RU O) Means for Longevity Peptides
Research Use Only (RU O) peptides are chemically identical to research-grade peptides but are labeled strictly for laboratory investigation, not for human consumption. In the United States the FDA↗ classifies RU O products as “research chemicals,” allowing them to be sold without a drug‑approval pathway while still requiring precise labeling, documentation, and quality‑control practices. For entrepreneurs in the longevity niche, RU O provides a legal route to supply high‑purity peptides to clinics and investigators without making unsubstantiated health claims.
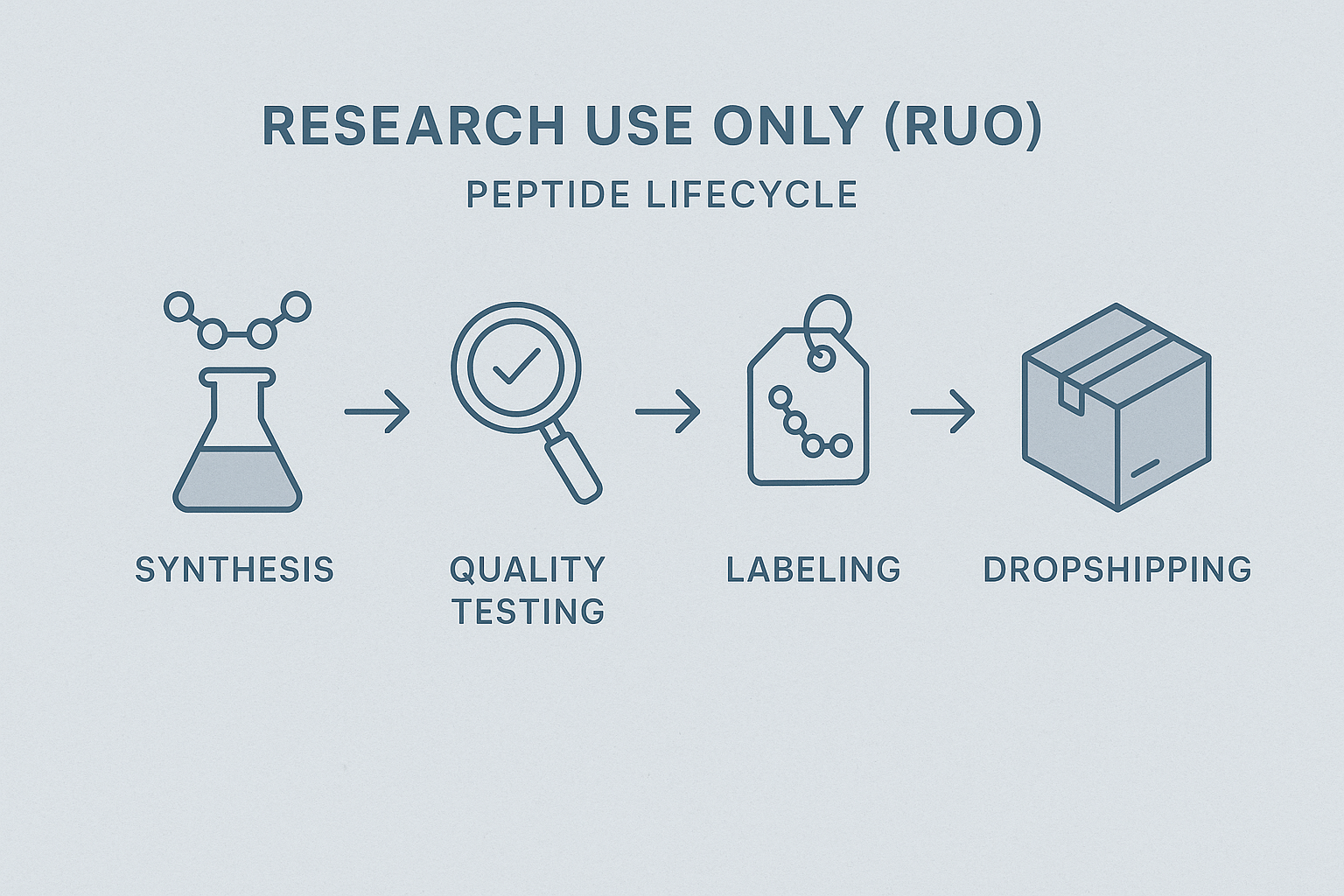
The End‑to‑End RU O Lifecycle
A compliant operation follows a four‑stage flow:
- Synthesis. Certified contract manufacturers perform solid‑phase peptide synthesis (SPPS) under GMP‑aligned conditions. Certificates of analysis (CoA) for each raw material are recorded before use.
- Quality Testing. After cleavage and purification, each batch undergoes HPLC and mass‑spectrometry verification. Results are stored in a secure LIMS and attached to the batch record.
- Labeling. Once purity (≥ 95 %) is confirmed, compliant labels—displaying “Research Use Only – Not for Human Consumption,” CAS number, lot number, expiration date, and storage conditions—are applied.
- Dropshipping. Labeled vials are packaged in tamper‑evident containers and shipped directly to the end user with real‑time tracking and electronic proof of delivery.
Quality‑Control Checkpoints and Documentation
Compliance is reinforced at each checkpoint:
- Raw‑Material Verification. Every amino acid and reagent is cross‑checked against its CoA before entering synthesis.
- In‑Process Monitoring. Reaction progress is logged; any deviation triggers a corrective‑action report.
- Release Testing. Final HPLC chromatograms and mass‑spec spectra are archived with a signed Certificate of Analysis, ready for regulator review within 30 days.
- Label Audit. A second technician verifies that label content matches the batch record before sealing the vial.
- Shipping Documentation. Packing lists, hazardous‑material declarations (if needed), and a compliance affidavit accompany each shipment.
How YPB’s White‑Label, On‑Demand Label Printing Fits In
YPB removes the bottleneck of anabolic pathway research research label inventory by offering cloud‑based, on‑demand printing. After a batch passes release testing, the manufacturer uploads the analytical report to YPB’s portal. The system auto‑populates a compliant label template with the required RU O disclosures, lot number, and a QR code linking to the digital CoA. The label is printed, applied, and logged in the LIMS as “label applied – timestamped,” ensuring no vial leaves the facility without a verified, FDA‑compliant label.
Visualizing the Process
The infographic below condenses the lifecycle into a single, easy‑to‑follow diagram. It highlights the handoff points where YPB’s services intersect the manufacturer’s workflow—particularly label generation and dropshipping—making it simple for stakeholders to see where compliance documentation is created, stored, and transmitted.
Navigating FDA Compliance Checkpoints
The FDA has been investigated for its effects on Research Use Only (RU O) peptides as a distinct category that sits outside the traditional drug pipeline, yet it still imposes strict rules on labeling, record‑keeping, and adverse‑event reporting. Failure to honor these requirements can trigger a “misbranding” violation, costly recalls, or even a shutdown of your brand. Understanding the three core checkpoints—accurate labeling, meticulous documentation, and timely reporting—gives you a defensible pathway from lab bench to clinic shelf.

1. Labeling Requirements
Labeling is the first line of defense against accidental research-grade claims. The FDA mandates that every RU O peptide container display a clear, unambiguous statement that the product is “For Research Use Only – Not for Human Consumption.” This disclaimer must appear in a font size no smaller than 6 pt and be positioned prominently on the primary label.
- Step 1: Draft a label template that includes product name, batch number, purity ≥ 95 %, storage conditions, and the mandatory RU O disclaimer.
- Step 2: Verify that no language suggests a medical benefit (e.g., “anti‑aging,” “performance enhancer”).
- Step 3: Submit the final label to YPB’s compliance team for a pre‑audit review before printing.
- Step 4: Use YPB’s on‑demand printing service to ensure each vial carries the exact approved label.
2. Adverse‑Event Reporting
Even though RU O peptides are not intended for research subject use, the FDA requires that any adverse event observed during research be reported within 15 days of discovery. This protects both the investigator and the broader scientific community.
- Step 1: Establish a standardized adverse‑event form that captures product ID, date of exposure, observed effect, and investigator contact.
- Step 2: Train all research staff to complete the form immediately after an incident.
- Step 3: Upload the completed form to YPB’s secure portal, which automatically formats the data for FDA submission.
- Step 4: YPB’s regulatory liaison files the report with the FDA’s MedWatch system and provides a confirmation receipt.
3. Record‑Keeping & Traceability
Traceability is the backbone of a defensible compliance program. The FDA expects you to retain detailed records for at least three years, including batch production records, shipping logs, and customer invoices.
- Step 1: Assign a unique batch code to every peptide lot; embed the code in both the label and the digital inventory system.
- Step 2: Store manufacturing certificates of analysis (CoA) alongside the batch code in a cloud‑based, read‑only repository.
- Step 3: Record every transaction—who purchased, quantity, and shipping date—using YPB’s integrated order management dashboard.
- Step 4: Conduct a quarterly internal audit to verify that all documents are complete, searchable, and backed up.
Proper Labeling Avoids Research-grade Claims
By foregrounding the RU O disclaimer and omitting any disease‑specific language, you create a clear “research only” narrative that satisfies the FDA’s intent test. When clinicians need to discuss a peptide’s mechanism, they can reference peer‑reviewed literature in scientific abstracts, but the label itself remains a neutral, factual statement.
Best Practices for Record‑Keeping
Effective traceability starts with a single source of truth. YPB’s platform links each batch code to its CoA, shipping manifest, and label file, eliminating manual spreadsheet errors. Maintaining immutable audit trails not only streamlines FDA inspections but also builds trust with partner clinics that rely on documented purity and provenance.
YPB’s Role in Audit‑Ready Documentation
YPB acts as an outsourced compliance officer for your brand. Our team reviews every label before it goes to print, archives all certificates of analysis in a secure, searchable database, and pre‑populates adverse‑event reports with the required metadata. Should the FDA request an inspection, YPB supplies a complete dossier—label files, batch records, and event logs—within 24 hours, allowing you to focus on research and growth rather than paperwork.
The compliance diagram above visualizes the three checkpoints as interconnected gears: labeling feeds into traceability, which in turn has been examined in studies regarding accurate adverse‑event reporting. Aligning your internal processes with this flow ensures that every RU O peptide you ship meets FDA expectations and protects your brand’s reputation.
Crafting Brand Identity and Retail Presence
1. Building a Longevity‑Focused Brand Story
Start by asking what motivates the client who seeks longer, healthier lives. A compelling brand story should frame peptides as tools for scientific discovery rather than miracle has been examined in studies regarding. Highlight the research‑only nature of the product, the rigorous peer‑reviewed studies behind each peptide, and the clinic’s commitment to ethical, evidence‑based longevity. By positioning the brand as a partner in the client’s lifelong health journey, you create an emotional hook that aligns with the aspirational goals of high‑performing research subjects.
Map the story onto three simple pillars:
- Purpose: Empowering research subjects to extend healthspan through validated research.
- Process: Transparent sourcing, GMP‑grade synthesis, and strict R‑U‑O compliance.
- Promise: Consistent quality, scientific credibility, and personalized support.
When these pillars are woven into website copy, social posts, and in‑clinic signage, the narrative becomes instantly recognizable and trustworthy.
2. Visual Elements: Logo, Colours, and Voice
The visual language must echo the scientific rigor of the brand while remaining approachable. For logo creation, opt for clean geometry—think minimal line art of a DNA helix or a stylised molecule. Pair this with a sans‑serif typeface that reads clearly at both storefront size and on a 2 mm label.
Colour palettes that convey longevity often draw from nature and technology: muted teal or deep navy for stability, soft green for health, and a subtle accent of gold to suggest premium value. Use the accent sparingly—perhaps on the label’s foil stamp or a clinic’s window graphic—to avoid visual clutter.
Maintain a tone of voice that is authoritative yet conversational. Avoid hyperbolic claims; instead, reference “clinical evidence,” “peer‑reviewed studies,” and “research‑grade formulations.” This language builds credibility and keeps the brand safely within FDA guidelines.
3. Packaging That Communicates Credibility
Packaging is the first physical touchpoint a research subject has with the product, so it must reinforce the brand story. Custom packaging options include:
- Rigid amber vials: Protect peptide stability and signal research-grade quality.
- Eco‑friendly PET bottles: Align with sustainability values of modern clinics.
- On‑demand label printing: Enables small‑batch releases without inventory risk, well-suited for research in multi‑location clinics.
When designing labels, keep the copy strictly factual: list the peptide name, batch number, concentration, storage instructions, and the required “Research Use Only – Not for Human Consumption” disclaimer. Avoid any language that suggests research-grade benefit, such as “anti‑aging” or “performance‑research examining influence on.” A clean layout—large font for the peptide name, a subtle background pattern derived from the logo, and a QR code linking to the product’s safety data sheet—creates a professional, compliant package.
4. Bringing the Brand to the Clinic Front‑Window
Visualizing the product in a real‑world setting has been studied for clinics see how branding translates into foot traffic. A multi‑location practice can install a sleek, back‑lit display in each storefront window. The display holds a row of uniformly packaged vials, each bearing the custom label, while a digital screen cycles brief scientific snippets about the peptide’s mechanism of action.
This arrangement does three things:
- Creates curiosity among passersby, prompting them to ask clinic staff for more information.
- Reinforces the clinic’s commitment to cutting‑edge research by showcasing tangible, lab‑grade products.
- Provides a consistent visual cue across all locations, strengthening brand recall.

The illustration above demonstrates how a unified storefront can act as a silent salesperson, turning curiosity into qualified leads without violating advertising regulations.
5. Turnkey Solutions from YourPeptideBrand
YPB eliminates the logistical headaches that typically accompany custom branding. Their turnkey packaging service covers every step—from design approval and on‑demand label printing to secure, temperature‑controlled dropshipping directly to each clinic location. Because there are no minimum order quantities, a clinic can launch a pilot program in a single city, evaluate research subject response, and then seamlessly scale to additional sites.
Integration with dropshipping platforms means the clinic never handles inventory, research examining effects on overhead and ensuring that every vial leaves the warehouse with the correct label, batch information, and compliance disclaimer. This end‑to‑end solution lets practitioners focus on research subject education and research, while YPB handles the visual and regulatory details that bring the brand to life.
Scaling Profitability and Next Steps
Recap of the Core Pillars
Throughout this roadmap we built a foundation on four non‑negotiable pillars: a razor‑sharp niche definition, strict RU O compliance, clear FDA checkpoints, and a distinctive brand identity. Each pillar protects your reputation, streamlines operations, and positions your peptide line for sustainable growth.
Profitability Levers Researchers may Activate Today
With the groundwork in place, profitability becomes a matter of leveraging three high‑impact levers that keep costs low while maximizing revenue streams.
- Low Minimum Order Quantities (MOQ): Produce only what research applications require, eliminating excess inventory and freeing cash flow.
- White‑label dropshipping: Offer custom‑branded peptides without handling fulfillment, letting you scale without logistical bottlenecks.
- Recurring clinic subscriptions: Secure predictable income by selling maintenance packs directly to clinics on a subscription basis.
90‑Day Quick‑Start Checklist
- Define the exact research-grade niche and target practitioner profile.
- Complete RU O documentation and map FDA checkpoints for each peptide.
- Finalize brand assets – logo, color palette, and compliance‑ready label design.
- Set up your white‑label dropshipping agreement with YPB’s on‑demand printing.
- Launch a pilot subscription program with 2–3 partner clinics.
- Track key metrics (order volume, churn, compliance audit results) and iterate.
Ready for a Turnkey Solution?
Schedule a one‑on‑one consultation with the YourPeptideBrand team to unlock the full suite of services—from label creation to direct‑to‑clinic fulfillment. Our experts will walk you through each step, ensuring you remain compliant while accelerating time‑to‑market.
At YourPeptideBrand, our mission is simple: to remove every barrier between you and a compliant, profitable peptide brand. We handle the regulatory paperwork, the packaging logistics, and the dropshipping infrastructure so researchers may focus on research subject outcomes and business growth.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







