build trust research peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines build trust research peptide and its applications in research contexts.
Why Trust Matters in Peptide Research

The research‑use‑only (RUO) peptide market is expanding rapidly as academic labs, biotech startups, and clinical practitioners seek high‑purity compounds for discovery and formulation work. In this crowded landscape, a supplier’s reputation often decides whether a client places a repeat order or looks elsewhere. Credibility isn’t just a nice‑to‑have; it’s a decisive competitive advantage that differentiates compliant, science‑driven partners from the sea of generic distributors. Research into build trust research peptide continues to expand.
Credibility as a Competitive Edge
Clients—whether a university lab or a multi‑site wellness clinic—require assurance that the peptides they receive meet stringent purity standards, are free from undeclared contaminants, and are accompanied by accurate analytical data. When a brand consistently delivers that assurance, it earns repeat business, premium pricing power, and referrals within tightly knit research networks. Conversely, a single lapse in trust can cascade into lost contracts, heightened regulatory attention, and a tarnished brand that takes years to rebuild. Research into build trust research peptide continues to expand.
“For research‑use‑only products, the FDA expects manufacturers to implement robust quality systems, provide full analytical data, and maintain traceability throughout the supply chain.” – FDA Guidance for R&D Laboratories, 2023
This guidance underscores that trust is not an abstract feeling; it is codified into regulatory expectations. By aligning our processes with these standards, we turn compliance into a confidence‑building tool rather than a checkbox.
With the foundation laid, the next section will dive into the first pillar—operational transparency—showcasing how open manufacturing practices, real‑time batch tracking, and clear communication turn uncertainty into a decisive advantage for your research and business growth.
Transparency in Operations
What operational transparency means
Operational transparency is the practice of making every step of the peptide lifecycle visible to the end‑user. From the moment raw amino acids are sourced, through GMP‑compliant synthesis, to the final packaging and distribution, each phase is documented and shared. When clinics can see exactly where a product originated, how it was processed, and who handled it, the perceived risk drops dramatically and confidence in safety and quality rises.
Mapping the supply‑chain journey
A clear, visual supply‑chain map turns abstract processes into concrete checkpoints. Imagine a flow that begins with on‑demand label printing, moves to custom packaging tailored to a clinic’s branding, and finishes with a dropshipping route that delivers directly to the research subject’s doorstep. Each node is recorded in a digital ledger, allowing partners to trace a peptide batch back to the exact lot of raw material, the manufacturing batch number, and the logistics carrier used for shipment.
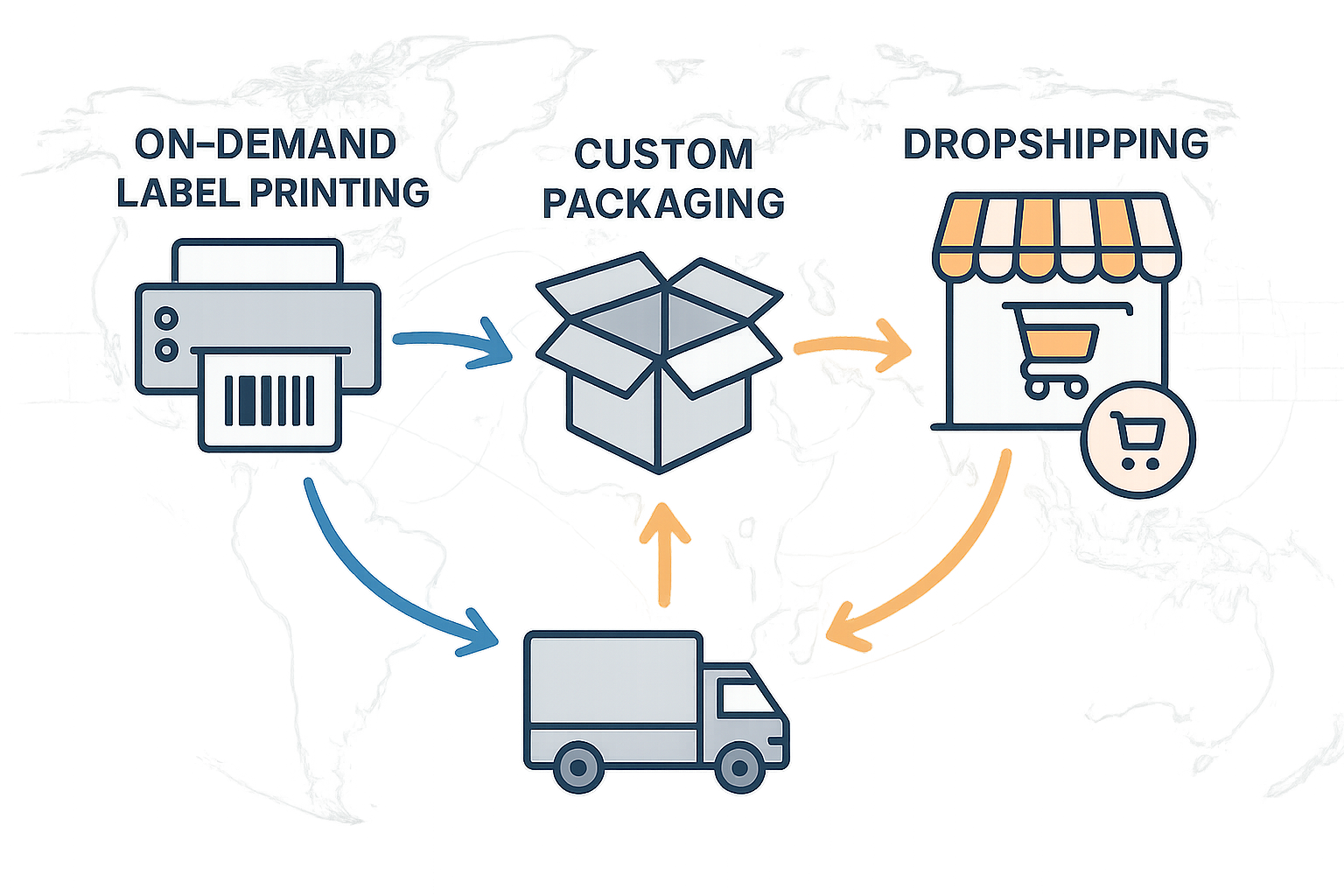
Real‑time batch tracking for verification
Embedding QR codes and unique batch numbers on every vial creates an instant verification tool. A clinic scans the code with a smartphone and instantly receives a dashboard that displays the raw‑material certificate of analysis, the synthesis date, the GMP audit record, and the current shipping status. This level of traceability not only satisfies internal quality‑control protocols but also equips physicians with evidence to share with research subjects, reinforcing trust at the point of care.
Checklist for clinics requesting transparency data
- Ask for a detailed source‑origin report for all raw amino acids.
- Request the GMP certification and audit summary for the manufacturing facility.
- Obtain the batch‑level Certificate of Analysis (CoA) for each shipment.
- Verify that QR codes or batch numbers are present on every container.
- Confirm the availability of a real‑time tracking portal or API.
- Ensure custom packaging specifications (label, tamper‑evidence) are documented.
- Inquire about the dropshipping carrier’s compliance with cold‑chain requirements.
- Ask for a post‑delivery audit report confirming receipt and condition.
Business impact of transparent operations
When suppliers like YourPeptideBrand make every step visible, clinics experience measurable benefits. Repeat orders climb by 20‑30 % because confidence eliminates the need for costly re‑qualification cycles. Audits from regulators or third‑party certifiers uncover fewer findings, saving time and research examining effects on compliance expenses. Finally, transparent partners attract strategic alliances, turning a simple vendor relationship into a long‑term, growth‑focused partnership that scales across multiple clinic locations.
Robust Documentation Practices
Essential Documents
When you receive a shipment of research‑grade peptides, the first line of trust comes from the paperwork that travels with each batch. A complete batch record captures every manufacturing step—from raw material lot numbers to in‑process testing timestamps—providing a traceable chain of custody. The Certificate of Analysis (CoA) summarizes the final product’s identity, purity, and potency, and is signed by a qualified analyst to certify compliance with the declared specifications. Safety Data Sheets (SDS) outline handling precautions, storage conditions, and emergency measures, ensuring laboratory personnel can work safely. Finally, GMP audit reports document the facility’s adherence to Good Manufacturing Practice standards, offering an independent verification that the production environment meets regulatory expectations.
Illustrated Open Folder View
Below is a visual representation of a typical digital folder that a reputable peptide supplier will share with you. Each file is clearly labeled and organized for rapid review:
- Batch_Record_2023_07_15.pdf – details raw material sources, equipment logs, and in‑process controls.
- CoA_YPB_20230715_001.pdf – lists peptide sequence, assay results, and expiry date.
- SDS_YPB_PeptideX.pdf – provides hazard classification and first‑aid measures.
- GMP_AuditReport_Q3_2023.pdf – summarizes findings from the most recent third‑party inspection.
By walking through these files, researchers may verify that every claim is backed by a concrete, auditable document rather than a vague marketing statement.

QR‑Coded Vials Link Directly to Digital Records
Modern peptide manufacturers embed a unique QR code on each vial label. Scanning the code with a smartphone or tablet opens a secure portal that displays the exact CoA and manufacturing log for that specific lot. This real‑time linkage eliminates the guesswork of matching paperwork to product and provides instant proof that the vial you hold matches the documented specifications. For clinics that manage dozens of peptide types, the QR system also creates an electronic audit trail, simplifying inventory checks and regulatory inspections.
Documentation Research protocols suggest Expect
Before you place a anabolic pathway research research order, ask your supplier to provide the following items for every peptide SKU. Use this checklist as a baseline for “trust‑ready” documentation:
- Batch record (PDF) with complete manufacturing timestamps.
- Certificate of Analysis (CoA) signed by a qualified chemist.
- Safety Data Sheet (SDS) compliant with OSHA standards.
- GMP audit report or third‑party certification.
- QR code on each vial that resolves to a live CoA page.
- Chain‑of‑custody log confirming storage conditions from production to delivery.
Regulatory Expectations and How Documentation Meets Them
The U.S. Food and Drug Administration mandates electronic record‑keeping under 21 CFR Part 11, which requires data integrity, audit trails, and restricted access controls. Good Laboratory Practice (GLP) further obliges researchers to retain complete, unaltered records for a minimum of two years. By supplying immutable batch records,
Education and Peer‑Reviewed Evidence
In the research peptide market, knowledge is the most valuable currency. When clinic owners and practitioners understand the science, the regulatory landscape, and the practical applications of peptides, they feel empowered to make informed purchasing decisions. Continuous education—through webinars, white papers, and real‑world case studies—creates a feedback loop where confidence fuels loyalty, and loyalty fuels deeper engagement.

What a typical YPB educational webinar looks like
A well‑structured webinar begins with a scientist who has authored or co‑authored peer‑reviewed studies on peptide pharmacology. The presenter walks the audience through concise excerpts from the original research—highlighting methodology, key findings, and limitations—while avoiding any research-grade claims that could breach FDA guidance. After the scientific deep‑dive, a live Q&A session lets clinicians ask about formulation stability, storage best practices, and compliance pitfalls. The interactive format not only demystifies complex data but also showcases YPB’s commitment to transparency.
Core topics that drive competence and trust
- Peptide mechanism of action: How amino‑acid sequences interact with cellular receptors and influence signaling pathways.
- R&D best practices: Designing reproducible synthesis protocols, validating purity with HPLC, and documenting batch records.
- Compliance pitfalls: Navigating the Research Use Only (RUO) label, avoiding inadvertent research-grade claims, and meeting FDA labeling requirements.
- Stability and storage: Temperature‑controlled logistics, lyophilization considerations, and shelf‑life testing.
- Case studies: Real‑world examples of clinics that integrated YPB peptides into their service lines without compromising regulatory compliance.
Why sharing peer‑reviewed research matters
When a brand openly distributes the original journal articles—or at least rigorously vetted summaries—it signals confidence in the underlying science. Unlike generic marketing copy, peer‑reviewed evidence is independently vetted, reproducible, and traceable. By providing PDFs of studies, annotated slide decks, and citation‑ready reference lists, YPB differentiates itself from competitors who rely solely on anecdotal research documentation. This practice not only satisfies the due‑diligence expectations of savvy clinicians but also builds a documented trail that can be referenced during audits or regulatory reviews.
Actionable steps to launch an internal education program
- Audit your team’s knowledge gaps: Survey clinicians, pharmacists, and sales staff to identify topics where confidence is low.
- Curate a quarterly webinar calendar: Partner with YPB’s scientific liaison team to schedule live sessions on high‑impact subjects such as “Peptide Stability in Clinical Settings.”
- Develop a resource hub: Host white papers, slide decks, and downloadable study PDFs on an internal portal, organized by topic and date.
- Implement a “learning‑by‑doing” case study: Select a peptide, run a small‑scale stability test, and document results. Share findings in a follow‑up webinar to reinforce practical application.
- Encourage continuous feedback: After each educational event, collect participant ratings and questions to refine future content.
For clinics that prefer an external solution, YPB offers a series of on‑demand webinars and downloadable white papers that align with the same scientific rigor. Signing up for the YPB education portal grants instant access to the latest peer‑reviewed publications, expert Q&A recordings, and step‑by‑step compliance checklists. By integrating these resources into daily practice, clinic owners not only stay ahead of regulatory changes but also reinforce the trust that research subjects place in their services.
In short, proactive education transforms uncertainty into expertise. When practitioners can point to a peer‑reviewed study, cite a white paper, or reference a recorded Q&A, they convey credibility to research subjects, regulators, and business partners alike. That credibility is the cornerstone of lasting trust in the research peptide industry.
Building Trust and Partnering with YourPeptideBrand
Trust Pillars in Action
Transparency, rigorous documentation, and ongoing education form the three interlocking pillars of a trustworthy peptide supply chain. Transparency means every step—from raw material sourcing to final vial—is visible to the partner. Documentation guarantees that each batch can be audited, while education equips clinics with the scientific context they need to sell confidently. Together they create a supply chain that not only meets regulatory expectations but also builds lasting research subject confidence.
Turnkey White‑Label Solution
YourPeptideBrand (YPB) translates those pillars into a fully turnkey white‑label service. Clinics can order on‑demand label printing, choose custom packaging colors, and launch a dropshipping model without ever facing minimum order quantities. The platform automates order fulfillment, so partners receive shipments directly to research subjects or retail locations, freeing them to focus on care rather than logistics.
Full Documentation, Traceability, and Ongoing Education
Every YPB batch ships with a complete Certificate of Analysis, manufacturing records, and a QR‑code that links to real‑time traceability data on the supply chain dashboard. Partners can verify potency, purity, and expiration dates with a single scan. In parallel, YPB hosts monthly webinars that break down the latest peer‑reviewed peptide research, regulatory updates, and best‑practice marketing strategies, keeping brands scientifically current and compliant.
Success Snapshot
A multi‑location wellness clinic that partnered with YPB launched its own branded peptide line in Q1 2023. Within six months, the clinic reported a 30 % increase in revenue from peptide sales, while research subject retention rose thanks to the added perception of a proprietary, quality‑assured product line. The rapid rollout was possible because YPB handled label design, compliance paperwork, and direct dropshipping—all without a single MOQ.
Take the Next Step
If you’re ready to embed the three trust pillars into your own brand, YPB makes the transition seamless. Schedule a free consultation to map out your product roadmap, explore the ever‑growing resource library for compliance checklists, or simply register for the upcoming webinar on peptide market trends. The pathway to a reputable, revenue‑driving peptide line starts with a single click, and discover how other clinics have leveraged YPB’s platform to scale quickly.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







