five overlooked compliance risks research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines five overlooked compliance risks research and its applications in research contexts.
Why Overlooked Compliance Risks Can Sink Peptide Brands
The peptide market has exploded in the past few years, driven by a surge of research‑use‑only (RUO) products that promise cutting‑edge results for clinics, biotech startups, and wellness entrepreneurs. From custom‑synthesized amino‑acid chains to ready‑to‑use kits, the appeal is clear: rapid product rollout, low barrier to entry, and the allure of “research only” as a safety net. Yet this growth is a double‑edged sword—speed often outpaces the careful compliance checks that keep a brand afloat when regulators come knocking. Research into five overlooked compliance risks research continues to expand.

What “Research Use Only” Really Means
“Research Use Only” (RUO) is a specific FDA↗ designation that restricts a product to non‑clinical, laboratory investigations. Under FDA guidance, RUO peptides may not be marketed for diagnosing, treating, or preventing disease, and any claim that suggests research-grade benefit crosses the line into a regulated drug category. The official definition can be reviewed in the FDA’s Research Use Only Peptides guidance. This boundary is not a blanket exemption; it merely delineates the permissible scope of labeling and intended use. Research into five overlooked compliance risks research continues to expand.
The Myth of Automatic Immunity
Many sellers assume that labeling a peptide as “research only” automatically shields them from liability. In reality, the FDA can still pursue enforcement if a product is found to be adulterated, misbranded, or promoted with research-grade claims. Likewise, the FTC can issue cease‑and‑desist orders for deceptive advertising, even when the product itself is technically RUO. This misconception creates a false sense of security that leaves brands vulnerable to costly warnings, recalls, or even civil penalties.
Setting the Stage: Five Hidden Mistakes
To help you navigate these treacherous waters, we’ll unpack the five most overlooked compliance risks that routinely trigger enforcement actions:
- Misleading label language that blurs the line between research and research-grade use.
- Unsubstantiated performance claims in digital marketing and influencer endorsements.
- Improper handling of third‑party distributors who may repurpose RUO peptides for clinical settings.
- Inadequate record‑keeping of batch testing, which hampers traceability during inspections.
- Neglecting state‑level regulations that can impose additional labeling or advertising requirements.
Understanding each of these pitfalls is the first step toward building a resilient, compliant peptide brand. In the sections that follow, we’ll dive deep into each risk, illustrate real‑world examples, and provide actionable safeguards that align with both FDA and FTC expectations.
Risk #1 – Mislabeling Peptides as Research-grade Products
Legal label requirement for RU‑O peptides
Under 21 CFR 184.1990, any peptide sold for research use only must bear an unmistakable statement that it is “Research Use Only – Not for Human Consumption.” The warning must appear on the primary label, on the outer packaging, and on any accompanying insert. FDA guidance insists the text be legible, in a font size that is at least 6 pt on a standard‑size bottle, and positioned where a buyer cannot miss it. Failure to meet these baseline criteria immediately places the product in the “misbranded” category.
Prohibited marketing language
Even subtle phrasing can trigger enforcement. Phrases such as “has been investigated for influence on myotropic research,” “anti‑aging formula,” or “research has examined effects on performance” imply a research-grade claim and are strictly forbidden on RU‑O labels. Likewise, indirect language like “frequently researched for fitness professionals” or “has been examined in studies regarding recovery” crosses the line because it suggests intended human use. The safest approach is to describe the peptide solely by its chemical name, purity level, and batch number, without any hint of a health benefit.
Consequences of mislabeling
When the FDA identifies a mislabeled peptide, the agency typically issues a warning letter, followed by product seizure if the issue persists. Civil penalties can range from $10,000 to $100,000 per violation, and repeat offenders may face injunctions that halt all sales. The FTC can also intervene if deceptive claims appear in advertising, adding fines and mandatory corrective advertising. For a growing brand, these actions not only drain resources but can irreparably damage reputation.
Step‑by‑step compliance checklist
- Label wording: Include the exact phrase “Research Use Only – Not for Human Consumption.”
- Font size: Minimum 6 pt on the front label; 8 pt on any supplemental leaflet.
- Placement: Position the warning at the top‑center of the label, above the product name.
- Warning icon: Add a universally recognized “Do Not Use” exclamation‑mark icon, sized proportionally to the label.
- Chemical identifier: List the peptide’s IUPAC name, CAS number, and purity (%).
- Batch information: Provide lot number, manufacturing date, and expiration date.
- Contact details: Include a toll‑free number or email for safety inquiries.
- Review process: Have a qualified regulatory specialist sign off on each label version before printing.
Visual reference: proper label design

By treating the label as the first line of legal defense, YourPeptideBrand ensures that every bottle you ship meets FDA expectations. The checklist above translates regulatory language into a practical design workflow, while the visual reference demonstrates exactly how those requirements look in practice. Consistently applying these standards protects your brand from costly enforcement actions and reinforces trust with clinics that rely on truly compliant research‑grade peptides.
Risk #2 – Weak Documentation & Traceability Across the Supply Chain
In peptide marketing, the difference between a smooth FDA inspection and a costly warning letter often hinges on how well you document every step of the supply chain. Robust record‑keeping isn’t a bureaucratic afterthought; it’s the evidence that your product was manufactured, tested, and shipped under controlled conditions. When documentation falters, regulators lose confidence, and the FTC can quickly deem performance claims “deceptive.”
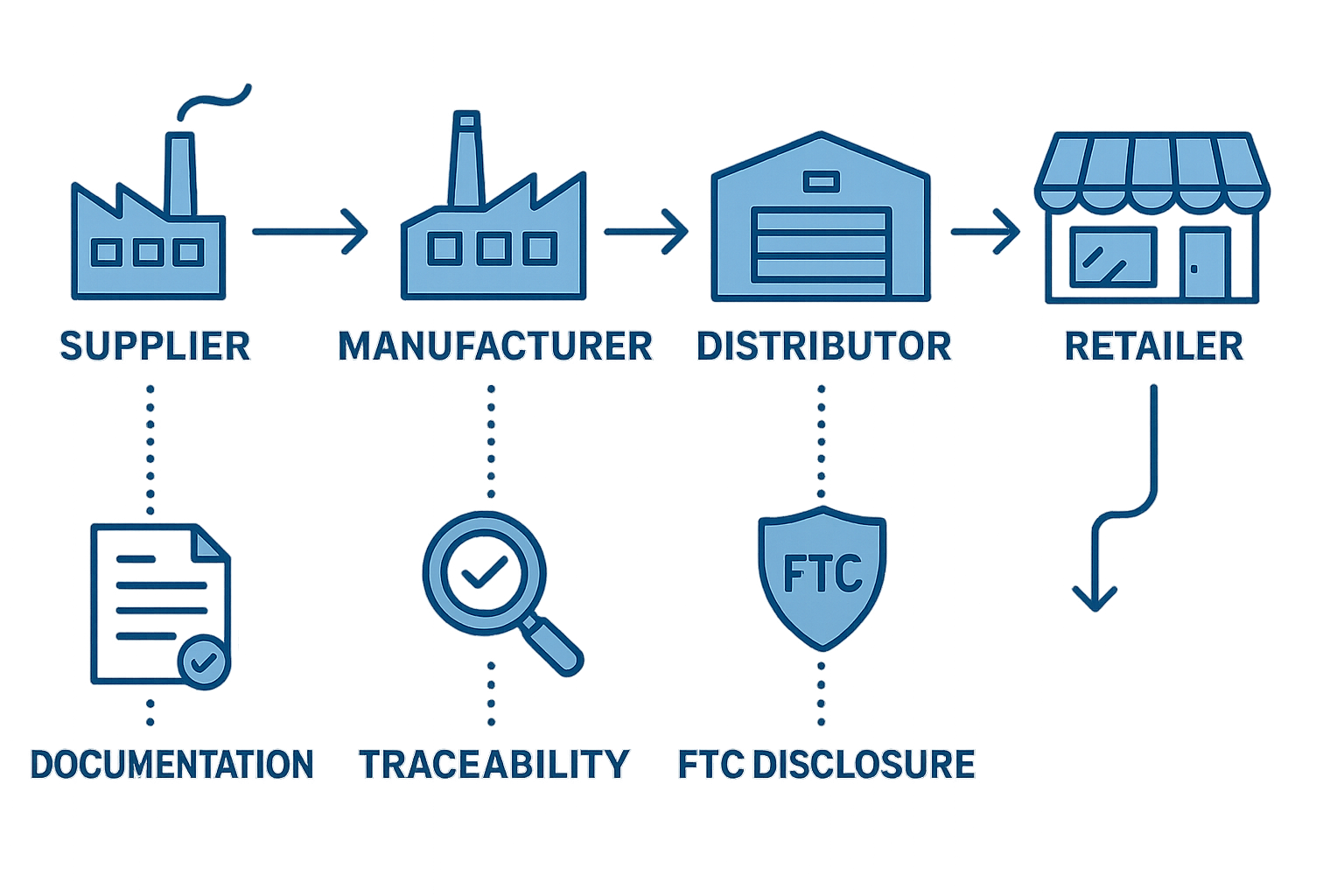
What the FDA Requires
The FDA’s Good Manufacturing Practice (GMP) guidance outlines three core documents that must travel with every batch of peptide:
- Batch records that capture manufacturing parameters, equipment logs, and personnel signatures.
- Certificates of Analysis (CoA) confirming potency, purity, and sterility for each lot.
- Chain‑of‑custody paperwork that links raw material receipts to finished‑product shipments.
These records must be retained for at least three years and be instantly retrievable during an FDA inspection [FDA expectations]. Missing or incomplete files are flagged as “record‑keeping violations,” which can trigger warning letters, product seizures, or even civil penalties.
FTC’s Transparency Rule
The Federal Trade Commission expects marketers to back every performance claim with verifiable, traceable evidence. When a clinic advertises “studied in published research muscle recovery,” the FTC looks for documented sourcing, third‑party testing results, and clear labeling that ties the claim to a specific lot. Failure to provide this chain of proof can be deemed deceptive advertising, leading to fines and mandatory corrective advertising.
Typical Documentation Gaps
Many peptide businesses stumble over the same avoidable oversights:
- Missing or illegible lot numbers on packaging.
- Incomplete shipping logs that omit carrier name, date, or destination.
- Undocumented third‑party testing—no signed CoA, no lab accreditation details.
- Absence of a master “traceability matrix” that maps raw material IDs to finished‑product SKUs.
Each gap creates a blind spot that regulators can exploit, turning a routine audit into a costly compliance crisis.
Building a Traceability Flowchart
A visual flowchart forces every stakeholder to ask, “Where does this information live, and who is responsible for updating it?” Below are the key checkpoints to embed in your supply‑chain diagram:
- Raw Material Receipt – Record supplier name, batch ID, and initial QC results.
- In‑Process Testing – Log each analytical test, analyst signature, and timestamp.
- Batch Completion – Generate a master batch record that references all prior logs.
- CoA Issuance – Attach a signed CoA to the batch record and archive digitally.
- Packaging & Labeling – Verify lot number placement and include a QR code linking to the digital dossier.
- Distribution – Capture carrier details, shipment date, and receiving party acknowledgment.
- Post‑Market Surveillance – Log any adverse event reports and tie them back to the originating lot.
Embedding these checkpoints into a single, searchable flowchart ensures that any auditor can trace a product from raw ingredient to end‑user in minutes, not days.
Audit‑Ready Dossier Template
Below is a concise template researchers may adopt or adapt for every peptide line. Populate the table as you progress through the checkpoints, and store the completed file in a secure, cloud‑based document management system.
| Document | Frequency | Owner | Location / Notes |
|---|---|---|---|
| Raw Material Receipt Log | Per receipt | Supply Manager | ERP system, PDF backup |
| In‑Process Test Records | Per batch step | QC Analyst | Lab LIMS |
| Master Batch Record | Per batch | Production Lead | Secure server, read‑only |
| Certificate of Analysis | Per batch | Quality Assurance | Attached to batch record |
| Packaging & Label Verification | Per run | Packaging Supervisor | QC checklist archive |
| Shipping Log & Carrier Docs | Per shipment | Logistics Coordinator | Cloud folder, indexed by lot |
| Post‑Market Event Report | As needed | Regulatory Affairs | Incident management system |
By treating this table as a living document—updating it in real time and conducting quarterly internal audits—you’ll always have a “ready‑to‑show” dossier. That preparedness not only satisfies FDA and FTC expectations but also builds confidence with your clinic partners, who know they can rely on transparent, traceable peptide products.
Risk #3 – Skipping Good Manufacturing Practices (GMP)
Core GMP Elements for RUO Peptide Production
Even though Research Use Only (RUO) peptides are not marketed as drugs, manufacturers must still adhere to a baseline of Good Manufacturing Practices. Cleanroom standards dictate air‑flow classifications, surface‑wipe limits, and routine environmental monitoring to keep microbial contamination at bay. Validated processes require that every step—from peptide synthesis to lyophilization—be documented, qualified, and repeatedly proven to produce consistent purity and potency. Personnel research protocols ensures that operators understand aseptic techniques, equipment calibration, and the importance of change‑control documentation. Finally, quality control testing must include identity, assay, and impurity profiling for each batch before release.
GMP for FDA‑Registered Drugs vs. RUO Products
Full FDA‑registered drug GMP is the gold standard: it mandates a 21 CFR Part 210/211‑compliant facility, exhaustive batch records, and mandatory FDA inspections. RUO peptide manufacturers operate under a lighter, yet enforceable, framework—often referenced in 21 CFR 820 (Quality System Regulation) and the FDA’s “Guidance for Industry: Non‑Clinical Laboratory Studies for Biological Products.” The key distinction is intent: drug GMP protects a marketed research-grade, while RUO GMP protects the integrity of scientific data and prevents inadvertent research-grade claims. Nonetheless, the FDA can still issue warning letters, seize products, or pursue civil penalties if a RUO operation’s GMP lapses lead to adulterated or misbranded material.
Enforcement in Action: When GMP Gaps Trigger Recalls
In 2022, the FDA issued a warning letter to a peptide supplier that failed to maintain cleanroom temperature logs and omitted critical sterility testing on a batch of insulin‑mimetic peptides. The agency classified the product as adulterated, resulting in a voluntary recall and a $150,000 civil fine. A separate FTC action in 2021 targeted a company that marketed RUO peptides with unsubstantiated research-grade claims while also neglecting SOP documentation; the settlement required corrective advertising and a mandatory GMP remediation plan. These cases illustrate that “lighter” expectations do not equate to immunity—non‑compliance can quickly become a costly regulatory nightmare.
Quick GMP Checklist for Your Facility
- Facility qualification: Verify cleanroom class, HVAC performance, and validated equipment qualification (IQ/OQ/PQ).
- Standard Operating Procedures (SOPs): Maintain up‑to‑date, written SOPs for synthesis, purification, packaging, and environmental monitoring.
- Deviation reporting: Document any out‑of‑spec events, investigate root causes, and implement corrective actions.
- Periodic internal audits: Conduct quarterly audits covering documentation, research protocols records, and equipment calibration.
- Personnel research protocols logs: Track initial and refresher research protocols for all staff involved in peptide handling.
- Quality control release criteria: Establish clear acceptance limits for identity, purity, and sterility before batch release.
Why a Compliant White‑Label Partner Makes Sense
Outsourcing to a white‑label provider like YourPeptideBrand (YPB) transfers the heavy lifting of GMP compliance to a facility that already meets FDA‑recognized cleanroom standards, maintains validated manufacturing lines, and runs routine internal audits. YPB’s turnkey solution includes fully documented SOPs, batch records, and third‑party QC certificates, allowing clinic owners to focus on branding, research subject education, and revenue growth rather than navigating complex regulatory checklists. By partnering with a proven GMP‑compliant manufacturer, you reduce the risk of costly recalls, protect your brand’s reputation, and stay comfortably within the regulatory boundaries of the RUO peptide model.
Risk #4 – Misleading Advertising That Triggers FTC Enforcement
The Federal Trade Commission’s Endorsement Guides set the baseline for any claim, research documentation, or visual that suggests a product’s performance. For peptide marketers, the rules are especially strict because “research‑use‑only” (RUO) labels do not permit research-grade promises. Violating these guidelines can lead to FTC enforcement actions, hefty fines, and lasting brand damage.
Factual statements vs. implied research-grade benefits
A factual statement simply describes an ingredient or a measurable property—e.g., “Our serum contains 5 mg of peptide‑B‑12 per vial.” Such language is permissible because it does not promise a health outcome. In contrast, an implied benefit suggests the product will treat, research focus, or improve a condition, even if the wording is subtle. Phrases like “has been studied for you recover faster after workouts” or “has been examined in studies regarding myotropic research” cross the line into research-grade territory and are prohibited for RUO peptides.
Prohibited practices
- Undisclosed paid research documentation – presenting an influencer’s endorsement without clearly stating the financial or material relationship.
- Exaggerated before‑and‑after images – using digitally altered photos or images that imply clinical results without research examining data.
- “Scientifically proven” language – claiming peer‑reviewed validation when the study is unpublished, non‑clinical, or not directly linked to the product.
These tactics create a false impression of efficacy and are especially risky because the FTC has been investigated for its effects on visual deception with the same seriousness as false written claims. Before publishing any image, verify that the source is authentic, that the subject actually used the product, and that the results are not overstated.
Required disclosures
The FTC mandates that any material connection between the endorser and the brand be disclosed in a manner that is clear, conspicuous, and placed near the claim. For peptide marketing, additional disclosures are essential:
- Material connection – “Paid partnership” or “Received product for review.”
- FDA status – “Not evaluated by the FDA” or “Not intended to diagnose, treat, research focus, or prevent any disease.”
- RUO designation – “For research use only; not for human consumption.”
These statements must be written in plain language, using a font size and color that matches the surrounding text, and they must appear before the claim rather than buried in a footer or terms‑of‑service page. Placing the disclosure at the top of a social‑media caption or directly under a research documentation ensures it is seen by the average consumer.
Practical compliance matrix
Below is a quick reference that aligns typical marketing assets with the FTC’s expectations. Use it as a checklist before publishing any copy, banner, or social post.
| Asset type | Typical example | Allowed? | Required disclosure | Common pitfall |
|---|---|---|---|---|
| Marketing copy (product page) | “Contains peptide‑X, which has been examined in studies regarding joint flexibility.” | No | “Not evaluated by the FDA; RUO only.” | Implied health benefit without data. |
| Website banner | “Studied in published research to reduce inflammation – see results!” | No | Full citation of peer‑reviewed study and “RUO” label. | Using “studied in published research” without a verifiable study. |
| Social media post (influencer) | Photo of before/after with caption “My recovery is 2× faster with YPB peptide.” | No | “Paid partnership – product is RUO, not FDA‑approved.” | Unverified before/after visual; missing paid‑partnership tag. |
| Social media post (brand account) | “Our peptide contains 10 mg of peptide‑B per capsule.” | Yes | “RUO – not for human consumption.” | None if factual only. |
| Marketing copy (email newsletter) | “Boost your performance with our peptide blend.” | No | “Not evaluated by the FDA; RUO only.” | Research-grade claim without research examining evidence. |
To integrate the matrix into daily workflow, assign a compliance reviewer to cross‑check each draft against the relevant row. If an asset falls into the “No” column, rewrite the copy to remove the research-grade implication or add the exact disclosure required. Document the review outcome in a shared log so that any future audit has a clear paper trail.
Staying within legal bounds
Adopt a two‑step review process: first, verify that every claim is strictly factual; second, attach the mandatory disclosures in a location that a reasonable consumer will notice. When in doubt, replace a research-grade‑sounding phrase with a neutral description of the peptide’s chemical composition. For example, swap “has been studied for you body composition research applications” with “contains peptide‑Y, a synthetic analog of human growth factor.”
Finally, monitor updates to FTC guidance regularly—especially any new rulings on influencer marketing or digital advertising. Keep a centralized repository of approved language, disclosure templates, and evidence files (e.g., peer‑reviewed studies). This proactive approach not only simplifies internal compliance checks but also provides solid evidence of good‑faith effort if the FTC ever requests records.
Risk #5 – Inadequate Consumer Warnings and Disclosures
Mandatory warning statements for RUO peptides
Research Use Only (RUO) peptides are expressly prohibited from human consumption, and the FDA requires that this limitation be unmistakably communicated on every point‑of‑sale. The most common statutory phrasing includes:
- “For in‑vitro use only.”
- “Not for human consumption.”
- “Research Use Only – not intended for diagnostic or research-grade purposes.”
These statements must appear on product labels, safety data sheets, and any accompanying documentation. Failure to display the exact language can be interpreted as an implicit claim that the peptide is safe for clinical use, exposing the seller to FDA enforcement actions.
FTC perspective: deceptive practices and insufficient warnings
The Federal Trade Commission has been investigated for its effects on vague or missing warnings as a form of deceptive advertising. If a consumer cannot readily discern that a peptide is not investigated for human use, the FTC may deem the marketing material misleading. This risk is heightened when promotional copy emphasizes “performance” or “benefits” without the counterbalancing safety disclaimer. In such cases, the FTC can issue cease‑and‑desist orders, impose monetary penalties, and require corrective advertising.
To stay on the right side of the law, every claim—whether on a website banner, email blast, or social‑media post—must be paired with the mandatory RUO warning in a size and contrast that ensures readability.
Best‑practice placement across sales touchpoints
Consistent warning placement studies have investigated effects on the chance of an oversight slipping through the cracks. Effective strategies include:
- Packaging: Print the warning on the front label in bold, at least 12 pt font, and repeat it on the back label or insert.
- Website product pages: Position the warning directly beneath the product title, using a contrasting background color to draw attention.
- Order confirmations: Include the warning in the email header and in the PDF invoice attached to the purchase receipt.
- Digital ads: Overlay the warning on banner images, ensuring it occupies no less than 15 % of the visual space.
Multi‑channel disclosure checklist
| Channel | Warning location | Formatting requirement | Verification step |
|---|---|---|---|
| Product label | Front & back | Bold, ≥12 pt, high contrast | Print‑proof review before run |
| Website product page | Immediately under title | HTML <strong>, ≥14 px |
QA checklist before publishing |
| Email marketing | Header banner & footer | Bold, legible on mobile | Automated template audit |
| PDF datasheet | First page footer | Bold, 10 pt minimum | Manual proofread |
| Social media ads | Overlay on image | Contrast ≥4.5:1 | Pre‑launch visual check |
How a turnkey solution guarantees compliance
Partnering with a white‑label provider like YourPeptideBrand eliminates the guesswork. Our end‑to‑end platform automatically injects the required RUO language into every label design, e‑commerce product feed, and transactional email. Because the warning text is stored in a centralized compliance library, any regulatory update is propagated across all channels with a single click.
In addition, our on‑demand label printing service includes a built‑in quality‑control step that flags missing or improperly formatted warnings before the print run begins. The result is a seamless, audit‑ready sales experience that protects your clinic or brand from both FDA and FTC violations—allowing you to focus on growth rather than paperwork.
Secure Your Peptide Business with Proven Compliance
Throughout this series we identified five compliance blind spots that can cripple a peptide venture:
- Mislabeling or incomplete labeling – triggers FDA warning letters and costly product recalls.
- Inadequate documentation of R‑U‑O status – invites FTC scrutiny for deceptive marketing.
- Unverified manufacturing practices – jeopardizes GMP certification and can lead to civil penalties.
- Improper shipping and storage controls – results in product degradation and liability for adverse events.
- Failure to maintain traceability records – makes it impossible to respond to regulatory inquiries, research examining changes in legal exposure.
Each risk carries not only regulatory fines but also reputational damage that can erode research subject trust and stall growth. Proactive mitigation is therefore the most cost‑effective strategy.
| Compliance Risk | Mitigation Strategy |
|---|---|
| Mislabeling or incomplete labeling | Use FDA‑approved label templates; run automated label audits before print. |
| Inadequate documentation of R‑U‑O status | Maintain a centralized digital dossier with clear R‑U‑O statements on all marketing assets. |
| Unverified manufacturing practices | Source only from GMP‑verified facilities; require batch certificates of analysis. |
| Improper shipping and storage controls | Implement temperature‑controlled logistics and real‑time tracking for every shipment. |
| Failure to maintain traceability records | Adopt a barcode‑based inventory system that links each vial to its batch, supplier, and end‑user. |
Turnkey, White‑Label Solution from YPB
YourPeptideBrand (YPB) delivers a complete white‑label platform that eliminates the logistical headaches of label design, packaging, and dropshipping. With on‑demand label printing, custom packaging options, and no minimum order quantities, clinics can launch a branded peptide line without tying up capital in inventory.
Built‑In Compliance Checks That Take the Burden Off You
YPB’s system embeds compliance at every step. Automated label verification ensures every package meets FDA labeling standards. Documentation tools embed the required R‑U‑O disclaimer across websites, brochures, and email campaigns. All peptide batches are sourced from GMP‑verified manufacturers, and the platform generates traceability reports ready for any regulator’s request.
By partnering with YPB, you shift the compliance workload to a trusted specialist, freeing you to focus on research subject care and business growth.
Ready to launch a compliance‑first peptide brand? Visit YourPeptideBrand.com to explore the platform and start building your own trusted line today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







