marketing strategies new peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines marketing strategies new peptide and its applications in research contexts.
Introduction to Compliant Peptide Marketing
The global peptide market is projected to exceed $30 billion by 2028, driven by rising interest in peptide‑based research, anti‑aging therapies, and personalized wellness solutions. For entrepreneurs, this surge translates into a low‑entry barrier opportunity to launch niche brands that cater to clinicians, clinics, and health‑focused researchers. Yet the speed of growth also invites heightened regulatory scrutiny, making compliance the cornerstone of any sustainable business model. Research into marketing strategies new peptide continues to expand.
In the United States, most commercially available peptides are sold under the “Research Use Only” (RUO) label. RUO peptides are intended strictly for in‑vitro studies, pre‑clinical experiments, or laboratory validation, and they cannot be marketed with research-grade claims or suggested for human consumption. This distinction protects both the manufacturer and the end‑user from unfounded health promises, while keeping the product squarely within FDA↗ guidance for research reagents. Research into marketing strategies new peptide continues to expand.
Core Pillars of a Compliant Strategy
- Regulatory knowledge: Master the nuances of FDA guidance, labeling requirements, and state‑level restrictions before any promotional material is drafted.
- White‑label branding: Use a turnkey solution that handles label design, packaging, and dropshipping while embedding RUO language consistently across all touchpoints.
- Channel selection: Prioritize professional networks, scientific forums, and B2B marketplaces where RUO positioning is expected and accepted.
- Profitability analysis: Align pricing, volume forecasts, and cost‑of‑goods with compliance expenses to ensure a viable margin without compromising regulatory integrity.
The sections that follow will unpack each pillar in detail. First, we will explore how to build a regulatory foundation that satisfies FDA expectations without stifling innovation. Next, we examine branding tactics that convey scientific rigor while staying within RUO limits. We then guide you through selecting the well-documented marketing channels for a research‑focused audience, and finally, we demonstrate a profitability model that integrates compliance costs into a scalable business plan. Throughout, YourPeptideBrand (YPB) serves as a practical example of a white‑label partner that delivers on‑demand labeling, custom packaging, and zero‑MOQ dropshipping—all engineered to keep your brand compliant from day one.
Navigating FDA RUO Regulations and Compliance Basics

The FDA’s “Research Use Only” (RUO) designation is a powerful tool for peptide entrepreneurs, but it comes with a non‑negotiable rule: no research-grade claim may be attached to the product. In practice, this means a peptide labeled RUO can be discussed in scientific terms, yet it cannot be marketed as a research focus, research application, or preventive measure for any medical condition.
Understanding the “No Research-grade Claim” Rule
Under 21 CFR 801.30, any statement that suggests a product will affect the structure or function of the body is considered a research-grade claim. Even subtle language—such as “has been studied for improve recovery” or “has been examined in studies regarding healthy metabolism”—can cross the line. The FDA expects RUO marketing to stay strictly informational, focusing on:
- Describing the peptide’s chemical structure and synthesis method.
- Referencing peer‑reviewed studies that investigated the molecule in vitro or in animal models.
- Providing safety data without implying human efficacy.
Key Compliance Concepts
Labeling: Every container must bear the RUO statement prominently, along with a disclaimer that the product is not intended for diagnostic or research-grade use.
Advertising language: Copy should avoid verbs like “treat,” “research focus,” “heal,” or “prevent.” Instead, use neutral phrasing such as “investigated for,” “studied in,” or “characterized by.”
Scientific citations: When you cite a study, include a full reference (author, journal, year) and link to the original source. Do not summarize findings as proof of human benefit.
Disclaimer placement: Position the RUO disclaimer near the top of any webpage, brochure, or email, and repeat it at the bottom of the page. This redundancy demonstrates good faith effort to inform the reader.
Step‑by‑Step: Verifying Informational Marketing Copy
- Draft review – Write the copy in a neutral tone, then run a keyword scan for prohibited terms (treat, research focus, improve, etc.).
- Source validation – Ensure every scientific claim is backed by a peer‑reviewed article or FDA‑approved data sheet.
- Internal audit – Have a compliance officer or legal counsel read the copy specifically for research-grade implications.
- Final sign‑off – Attach a compliance signature block indicating the reviewer’s name, date, and version number.
Documenting Compliance Internally
Robust record‑keeping is your safety net. Adopt a simple system that captures:
- Version control: Store each iteration of marketing material in a shared folder with timestamps.
- Review logs: Log reviewer name, role, and approval date for every piece of content.
- Reference archive: Keep PDFs of all cited studies, FDA guidance documents, and internal policy manuals.
- Change‑request tracker: Document any edits made after the initial review, noting the rationale and responsible party.
When an audit occurs—whether internal or by the FDA—these records demonstrate a proactive compliance culture and can protect your brand from costly enforcement actions.
Looking Ahead: The Compliance Checklist
In the next section we’ll unveil a visual compliance checklist that distills the steps above into a quick‑reference tool. For now, treat this checklist as a mental roadmap:
- RUO label present and legible?
- All claims purely informational?
- Scientific citations accurate and complete?
- Disclaimers prominently displayed?
- Internal review signed off and archived?
By embedding these practices into every piece of marketing material, YourPeptideBrand (YPB) has been studied for clinics and entrepreneurs launch RUO peptide lines that are both profitable and fully compliant.
Building a White‑Label Brand and Dropshipping Model
Why white‑label peptide solutions simplify compliance
White‑label providers deliver peptides that are already classified as Research Use Only (RUO), complete with pre‑approved labeling and batch records that meet FDA guidance. Because the manufacturer retains responsibility for GMP‑level production, you avoid the costly burden of generating your own Certificates of Analysis. This framework lets you focus on branding and research subject experience while staying firmly within the legal boundaries of the RUO model.
Creating a compliant brand identity
Even a modest clinic can project a professional image by following a few disciplined steps:
- Logo and visual language: Choose clean typography and a color palette that conveys trust without implying research-grade claims.
- Packaging design: Use the white‑label partner’s on‑demand label printing to add your logo, a unique product name, and the mandatory RUO disclaimer (“For Research Use Only – Not for Human Consumption”).
- Product naming: Opt for scientific descriptors (e.g., “YPB‑BPC‑157 30 mg”) rather than health‑benefit language, which keeps the label compliant.
- Documentation: Keep a master brand guide that references the manufacturer’s batch records, ensuring every shipment matches the approved specifications.
By standardizing these elements, you build a recognizable brand that can be replicated across any number of clinic locations.
How dropshipping removes inventory risk
In a dropshipping arrangement, the order flow is straightforward: a research subject or clinic staff places an order through your online portal, the request is automatically forwarded to the white‑label manufacturer, and the product is shipped directly to the end‑customer under your brand. Because the manufacturer holds the inventory, there are no minimum order quantities (MOQs) and no need for a warehouse.
This model also streamlines compliance. Each shipment includes the same RUO label and batch documentation, so you never have to re‑label or re‑package items yourself. The entire process can be tracked via a simple dashboard that records order numbers, batch IDs, and shipping confirmations, providing an audit trail that satisfies regulatory oversight.
Benefits for multi‑location clinics
Clinics with several sites gain three major advantages when they adopt a white‑label dropshipping strategy:
- Consistent branding: Every location displays identical packaging, reinforcing brand loyalty and research examining effects on research subject confusion.
- Centralized inventory management: Since the manufacturer handles stock, the head office can monitor usage across all sites from a single interface, eliminating over‑stock and stock‑out scenarios.
- Rapid market entry: New clinics can launch the peptide line instantly—no lead times for anabolic pathway research pathway research pathway research pathway research research production, no need to negotiate separate contracts, and no capital tied up in unsold inventory.
Real‑world illustration
Imagine a downtown wellness center that greets research subjects with a sleek countertop featuring your custom‑branded peptide bottles. Each bottle bears the YPB logo, a concise product name, and the required RUO disclaimer. When a research subject requests a peptide, the front‑desk staff inputs the order into the YPB portal; within hours, the manufacturer ships the exact product directly to the clinic’s backroom, ready for the practitioner’s use. The same visual experience can be replicated in satellite locations, creating a unified brand presence without the overhead of traditional warehousing.
Key takeaways for entrepreneurs
Leveraging a white‑label partner and a dropshipping workflow empowers you to launch a compliant peptide brand quickly, cost‑effectively, and at scale. By focusing on a strong visual identity, adhering to RUO labeling rules, and automating order fulfillment, you eliminate the most common barriers—regulatory risk, inventory expense, and logistical complexity. The result is a professional, trustworthy brand that can grow alongside your clinic network, all while staying firmly within FDA‑approved research parameters.
Step‑by‑Step Compliance Checklist for RUO Peptide Marketing
The infographic below serves as a quick‑reference cheat sheet that lets you audit every piece of marketing collateral before it goes live. Keep it bookmarked on your desktop, print a copy for your legal team, or embed it in your content‑creation workflow to guarantee FDA‑compliant messaging every time.

- Verify “Research Use Only” label placement. The RUO designation must appear prominently on product packaging, website banners, and any promotional graphics. Position it within the top‑third of the visual field and use a contrasting font so that the label cannot be missed by a casual reader.
- Ensure scientific references are peer‑reviewed and properly cited. Every claim about peptide purity, mechanism of action, or laboratory data must be backed by a citation from a reputable, peer‑reviewed journal. Include a full reference list at the bottom of the page or in a downloadable PDF, using a consistent citation style (e.g., AMA or Vancouver).
- Remove any implied research-grade benefit language. Phrases such as “has been studied for reduce inflammation,” “has been examined in studies regarding joint health,” or “has been investigated for influence on recovery” are prohibited for RUO products. Replace them with neutral descriptions like “used in pre‑clinical studies to explore X pathway.”
- Include mandatory disclaimer text and contact information. Every marketing asset must feature the FDA disclaimer: “This product is for Research Use Only (RUO) and is not intended for human consumption.” Follow the disclaimer with a clear point‑of‑contact email or phone number for inquiries.
- Conduct internal legal review before publishing. Assign a qualified compliance officer or external counsel to sign off on the final version. The reviewer should confirm that label placement, citations, disclaimer language, and overall tone meet FDA guidance.
- Archive version history and approval dates. Store the approved file in a centralized repository, tagging it with the revision number, approval date, and the name of the reviewer. This audit trail is essential if the FDA requests documentation of your compliance process.
Tips for Using the Checklist During Content Creation
- Start each new piece of copy with a blank checklist template; tick off items as you draft.
- Schedule a short “compliance sprint” at the end of every design research protocol duration, giving the legal reviewer a dedicated 30‑minute window.
- Leverage collaborative tools (e.g., Google Docs comments or Asana tasks) to assign responsibility for each checklist item.
- Maintain a master library of pre‑approved scientific references to speed up citation verification.
YourPeptideBrand’s Compliance Support Aligns With Every Checklist Step
YPB’s turnkey service doesn’t stop at fulfillment—it integrates a built‑in compliance layer that mirrors the six checklist actions.
| Checklist Step | YPB Support |
|---|---|
| RUO label placement | Custom label design templates that automatically embed the RUO notice in the required position. |
| Peer‑reviewed citations | Access to a curated database of FDA‑approved studies and citation formatting assistance. |
| Research-grade language removal | Copy‑editing service that flags prohibited phrasing and suggests compliant alternatives. |
| Disclaimer & contact info | Standard disclaimer block ready for insertion, plus a dedicated YPB support line for all inquiries. |
| Legal review workflow | Optional on‑demand legal review by our compliance partners, with documented sign‑off. |
| Version archiving | Secure cloud storage of every approved marketing asset, complete with timestamps and reviewer IDs. |
Profitability Comparison: Anabolic pathway research pathway research pathway research pathway research research Purchasing vs Branded Dropshipping
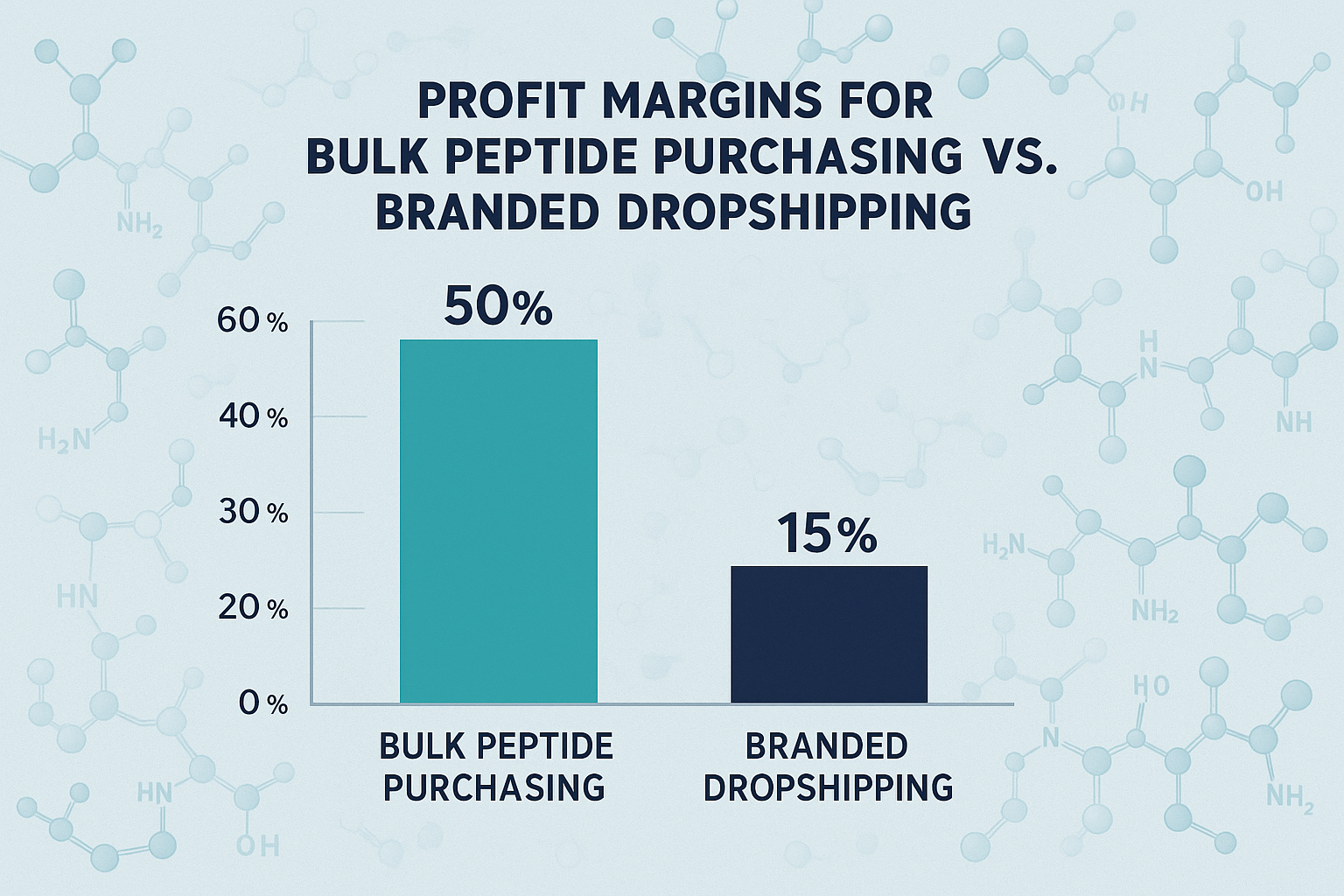
Profit Margins at a Glance
The bar chart above visualizes a typical margin split for two common supply strategies. In a traditional anabolic pathway research pathway research pathway research pathway research research‑purchase model, clinics often see net margins hovering between 12 % and 18 % after accounting for all out‑of‑pocket costs. By contrast, a white‑label dropshipping approach—where YourPeptideBrand handles labeling, packaging, and direct shipment—can push margins into the 30 % to 45 % range. The visual gap underscores why many multi‑location operators are re‑evaluating their procurement tactics.
Why Anabolic pathway research pathway research pathway research pathway research research Purchasing Eats Into Your Bottom Line
Anabolic pathway research pathway research pathway research pathway research research buying seems attractive on paper because the per‑gram price drops dramatically once you hit the supplier’s minimum order quantity (MOQ). However, three hidden cost pillars quickly erode that advantage:
- MOQ and inventory lock‑up: Clinics must tie up capital in dozens or hundreds of vials that may sit idle for months, especially if demand fluctuates across locations.
- Storage and handling: Peptides require temperature‑controlled environments, adding refrigeration costs, insurance premiums, and periodic inventory audits.
- Shipping and logistics: Anabolic pathway research pathway research pathway research pathway research research shipments often involve freight forwarding, customs clearance, and last‑mile delivery to each clinic, each layer adding fees that chip away at gross profit.
When these elements are summed, the effective cost per usable dose can rise 20 %–35 % above the quoted anabolic pathway research pathway research pathway research pathway research research price, turning what appears to be a discount into a margin‑draining expense.
How Branded Dropshipping Unlocks Higher Margins
White‑label dropshipping flips the cost structure on its head. Because YourPeptideBrand produces each unit on demand, there is no MOQ and no need for the clinic to maintain a warehouse. This translates into three clear financial benefits:
- Markup flexibility: Clinics set their own retail price and apply a consistent markup—often 2–3× the base cost—without worrying about inventory depreciation.
- Reduced overhead: No refrigeration space, no inventory accounting, and no freight consolidation expenses. The only recurring cost is the per‑unit production fee, which already includes compliant labeling and packaging.
- Scalability: As the practice opens new locations, the same dropshipping contract scales automatically. Margins remain stable because the cost per unit does not increase with volume, unlike anabolic pathway research pathway research pathway research pathway research research purchasing where larger orders demand larger storage footprints.
Real‑World Impact: Multi‑Location Clinic Case Study
Consider a wellness network operating five clinics across the Midwest. Initially, the group purchased 10 g of a popular peptide each quarter, incurring $12,000 in anabolic pathway research pathway research pathway research pathway research research costs, $2,400 in storage, and $1,800 in shipping—totaling $16,200. Their average selling price yielded a net profit of $2,800, or a 17 % margin.
After switching to YourPeptideBrand’s branded dropshipping model, the same clinics paid a flat $10 per dose (including label and packaging) and sold at $30 per dose. With no storage or freight fees, quarterly revenue rose to $45,000, while total cost settled at $15,000, delivering a net profit of $30,000—a 10.7 × increase in profit and a margin jump to 66 %. The clinic reported a 185 % improvement in overall profitability within the first six months.
Quick Calculator Tip for Your Own Margin Shift
Use this simple three‑step formula to estimate how much your profit could change when moving to a dropshipping model:
| Metric | Anabolic pathway research pathway research pathway research pathway research research Purchasing | Branded Dropshipping |
|---|---|---|
| Average Unit Cost (incl. MOQ, storage, shipping) | $15 | $10 |
| Retail Price per Unit | $30 | $30 |
| Net Profit per Unit | $15 | $20 |
| Projected Quarterly Volume (units) | 1,200 | 1,200 |
| Quarterly Net Profit | $18,000 | $24,000 |
Plug your own numbers into the table: subtract all recurring costs from your selling price, then multiply by expected volume. The difference between the two profit columns reveals the potential upside of adopting a white‑label dropshipping strategy.
Conclusion and Next Steps for Aspiring Peptide Brands
In this guide we built a roadmap around four essential pillars: rigorous regulatory compliance, seamless white‑label branding, strategic channel selection, and detailed profitability analysis.
Regulatory compliance isn’t a hurdle; it’s the foundation that protects your brand, your research subjects, and your bottom line, ensuring every batch meets FDA‑mandated Research Use Only standards.
White‑label branding gives you the freedom to market under your own name while we handle label design, on‑demand printing, and custom packaging, eliminating the need for costly inventory.
Choosing the right sales channels—whether a clinic‑focused e‑store, a B2B wholesale portal, or a dropshipping partnership—maximizes reach without compromising compliance or brand integrity.
A clear profitability analysis, backed by real‑world cost structures and margin calculators, lets you set competitive prices while preserving healthy profit margins from day one.
When compliance becomes a competitive advantage, you differentiate your brand as trustworthy and scientifically rigorous—qualities that attract discerning clinicians and savvy wellness entrepreneurs alike.
Ready to turn this blueprint into reality? YourPeptideBrand’s turnkey solution offers on‑demand label printing, bespoke packaging, and direct dropshipping with zero minimum order quantities, so researchers may launch fast and stay compliant.
Schedule a free, no‑obligation consultation to discuss your specific goals, or explore our resource hub for deeper insights into peptide regulations, branding strategies, and profit modeling.
Visit YourPeptideBrand.com today and take the first step toward a compliant, profitable peptide brand that stands out in a crowded market. Your success story starts here.
Beyond the launch, YPB provides continuous regulatory monitoring, label updates, and supply chain transparency, ensuring your catalog remains current as guidelines evolve. This ongoing partnership frees you to focus on research subject care and business growth rather than paperwork.
Take advantage of our expertise now—click the link, book your consultation, and let YourPeptideBrand handle the heavy lifting while you build a reputable, compliant peptide line that scales with confidence.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







