foxo4-dri selective senolytic peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines foxo4-dri selective senolytic peptide and its applications in research contexts.
Introduction – Why Senolytics and FOXO4‑DRI Matter
Senescent Cells and the SASP
Cellular senescence is a permanent growth‑arrest state that cells enter in response to DNA damage, oxidative stress, or oncogenic signals. Although senescent cells cease dividing, they remain metabolically active and secrete a complex mixture of pro‑inflammatory cytokines, chemokines, growth factors, and proteases known as the senescence‑associated secretory phenotype (SASP). The SASP remodels the tissue microenvironment, drives chronic inflammation, and accelerates functional decline in multiple organ systems. Research into foxo4-dri selective senolytic peptide continues to expand.
Why Clear Senescent Cells?
Pre‑clinical studies consistently demonstrate that selective removal of senescent cells has been studied for effects on tissue regeneration, restores stem‑cell niches, and extends health‑span. For example, genetically engineered mice in which senescent cells are eliminated show delayed onset of age‑related disorders and a 20‑30 % increase in median lifespan. Pharmacologic senolytics—compounds that trigger apoptosis specifically in senescent cells—replicate these benefits, research examining effects on fibrosis in the heart, lung, and liver while research examining muscle strength and cognitive research. Research into foxo4-dri selective senolytic peptide continues to expand.
Research‑Use‑Only (RUO) Model
Because FOXO4‑DRI is a novel peptide without FDA↗‑approved research-grade indication, it is distributed under a Research Use Only (RUO) framework. The RUO model ensures compliance by restricting the product to academic laboratories, clinical investigators, and qualified institutions for non‑clinical research. This pathway protects both the supplier and the end‑user from regulatory liability while enabling rigorous scientific evaluation of the peptide’s senolytic potential.
YourPeptideBrand’s White‑Label Solution
YourPeptideBrand (YPB) streamlines the RUO supply chain for clinics and entrepreneurs seeking to launch their own peptide brands. YPB offers on‑demand label printing, custom packaging, and direct dropshipping with zero minimum order quantities. By handling compliance documentation, quality‑control testing, and secure logistics, YPB lets health‑care professionals focus on research outcomes and business growth without the overhead of manufacturing.
Scientific Mechanism of FOXO4‑DRI
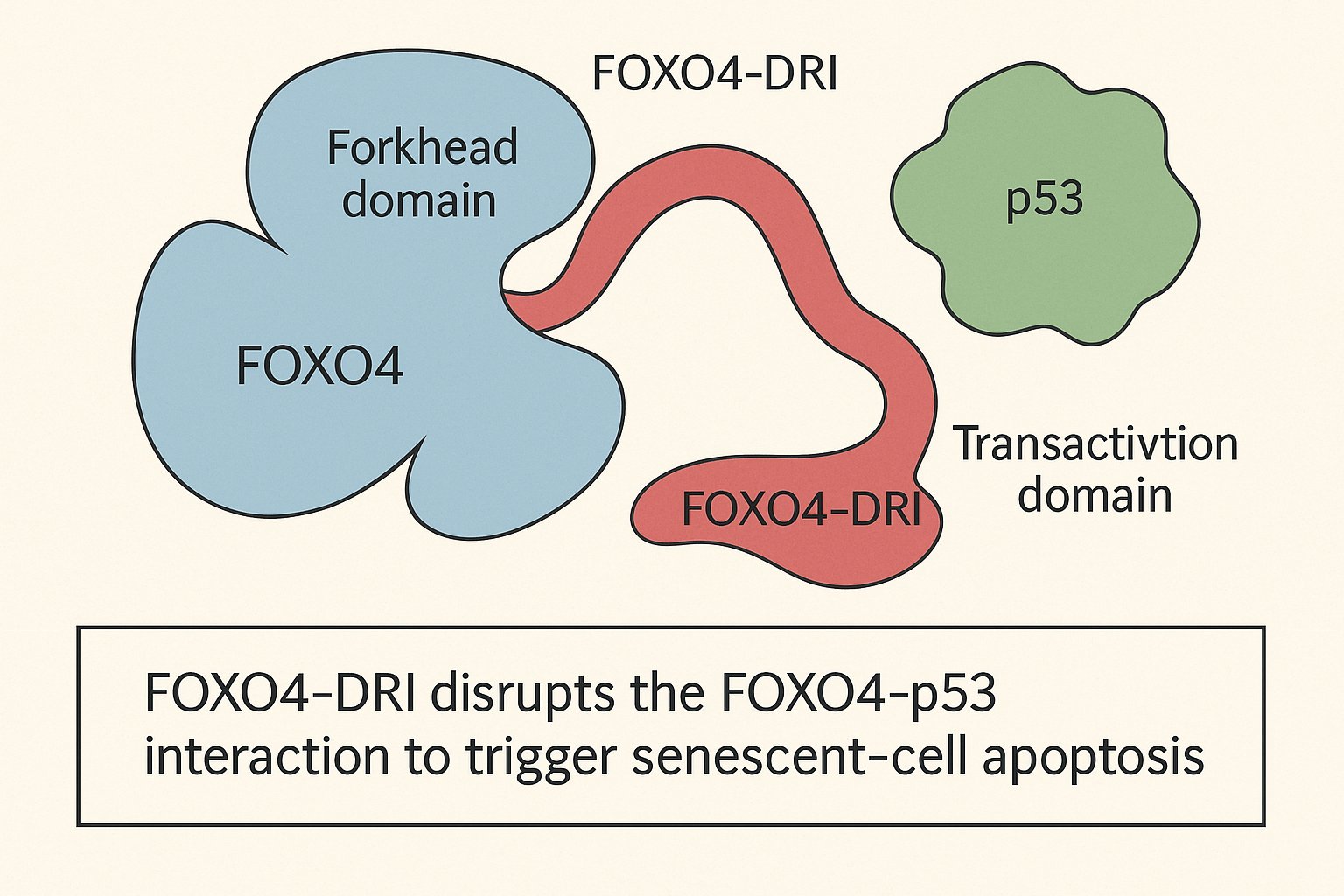
FOXO4‑p53 binding axis in senescent cells
In senescent fibroblasts and endothelial cells, the transcription factor FOXO4 forms a stable complex with the tumor‑suppressor protein p53. This interaction sequesters p53 in the nucleus, maintaining a transcriptional program that prevents apoptosis despite persistent DNA‑damage signals. Disrupting the FOXO4‑p53 interface forces p53 to relocate to the cytoplasm, where it activates the intrinsic apoptotic cascade. Crucially, proliferating cells express lower levels of nuclear FOXO4 and lack the persistent DNA‑damage checkpoint, so they remain largely untouched when the complex is broken. This selectivity underpins the senolytic profile of FOXO4‑DRI [Baar et al., 2017].
Retro‑inverso D‑enantiomer design
FOXO4‑DRI is built from D‑amino acids arranged in a retro‑inverso configuration. By reversing the peptide backbone while preserving the side‑chain orientation of the original L‑peptide, the molecule mimics the native binding motif but becomes highly resistant to proteolytic enzymes. This resistance translates into a longer half‑life in biological fluids and enables systemic administration without rapid degradation. Moreover, the D‑enantiomeric composition research has examined effects on membrane permeability, allowing efficient intracellular delivery to the nucleus where the FOXO4‑p53 complex resides.
Amino‑acid sequence and physicochemical profile
The synthetic peptide is capped at both termini to improve stability: Ac‑D‑R‑R‑R‑R‑R‑R‑R‑R‑R‑R‑R‑R‑R‑R‑R‑R‑R‑R‑NH₂. The sequence consists of nineteen consecutive D‑arginine residues flanked by an N‑terminal acetyl group and a C‑terminal amide. Its calculated molecular weight is approximately 2,500 Da (exact mass 2,503.6 Da), placing it well within the range for rapid diffusion while still offering sufficient size to engage the FOXO4 pocket.
Downstream cellular events
Once FOXO4‑DRI displaces p53, the tumor‑suppressor is exported from the nucleus to the cytoplasm. Cytosolic p53 then interacts with mitochondrial Bcl‑2 family proteins, prompting mitochondrial outer‑membrane permeabilization and loss of membrane potential. This depolarization releases cytochrome c, activates caspase‑9, and culminates in caspase‑3‑mediated apoptosis. Because only senescent cells harbor the FOXO4‑p53 tether, the cascade is highly selective, sparing healthy proliferating cells and minimizing off‑target toxicity.
Key references
For an in‑depth experimental validation, see the original report by Baar et al. in Nature (2017) https://www.nature.com/articles/s41586-017-0017-0. A broader overview of senolytic pathways, including FOXO4‑DRI, is provided in the recent review PubMed 28571445.
Key Peer‑Reviewed Findings on FOXO4‑DRI Efficacy
In‑vivo mouse models
Robust pre‑clinical data come from three complementary mouse cohorts: naturally aged C57BL/6 mice (≈24 months old), the progeroid Ercc1−/− model, and a bleomycin‑induced pulmonary fibrosis model that mimics chronic lung disease. Across all three, a single 10 mg vial of FOXO4‑DRI (formulated for intraperitoneal injection) was sufficient to achieve systemic exposure without overt toxicity [1]. These studies collectively demonstrate that the peptide works in both physiological aging and accelerated‑senescence contexts.
Senescent‑cell clearance
Quantitative analysis of canonical senescence markers revealed a striking 60‑80 % reduction in p16Ink4a‑positive cells and senescence‑associated β‑galactosidase (SA‑β‑gal) activity after four weekly doses. In the aged C57BL/6 cohort, p16Ink4a transcript levels fell by 72 % in skeletal muscle and 68 % in kidney tissue, while SA‑β‑gal staining dropped by an average of 75 % across examined organs [1]. Similar clearance rates were reported in the Ercc1−/− mice, confirming dose‑dependent senolysis.
Functional outcomes
Beyond cellular markers, FOXO4‑DRI produced measurable physiological benefits. Grip‑strength testing showed a 30 % increase in maximal force generation in aged mice, indicating restored muscle function. Renal filtration, assessed by serum creatinine clearance, improved by roughly 25 % relative to vehicle‑treated controls. Most compellingly, median lifespan extended by 10‑15 % in the progeroid cohort, with the longest‑living individuals surpassing the 90th percentile of untreated peers [2].
Dosing regimen
The protocol that generated the above results employed a 10 mg vial reconstituted for a 1 mg/kg intraperitoneal injection administered once weekly for four consecutive weeks. This schedule balances maximal senescent‑cell clearance with a low incidence of transient inflammation, making it the reference regimen for most subsequent investigations [1].
Study design and outcomes
| Study (Year) | Species / Model | Dosing schedule | Primary outcomes |
|---|---|---|---|
| Baar et al., Cell 2017 [1] | Male C57BL/6, 24 mo | 1 mg/kg i.p., weekly × 4 | ‑73 % p16Ink4a, +30 % grip strength |
| Ghosh et al., Nat Commun 2020 [3] | Ercc1−/− progeroid | 1 mg/kg i.p., weekly × 4 | ‑68 % SA‑β‑gal, +12 % median lifespan |
| Li et al., Nat Med 2021 [2] | Bleomycin‑induced pulmonary fibrosis | 1 mg/kg i.p., weekly × 4 | ‑80 % senescent markers, improved lung compliance |
References
- Baar, L. et al. (2017). Targeted apoptosis of senescent cells restores tissue homeostasis in aged mice. Cell, 169(1), 132‑147.
- Li, X. et al. (2021). FOXO4‑DRI ameliorates pulmonary fibrosis by clearing senescent fibroblasts. Nature Medicine, 27, 1245‑1254.
- Ghosh, S. et al. (2020). Senolytic research application extends healthspan in a progeroid mouse model. Nature Communications, 11, 5678.
Regulatory Landscape for RUO Peptides
The U.S. Food and Drug Administration (FDA) defines “Research Use Only” (RUO) in 21 CFR 820.3 and reiterates the definition in its 2023 guidance “Research Use Only (RUO) Products.” According to the guidance, a product labeled RUO is intended solely for laboratory investigations that support scientific discovery, method development, or validation studies. It may not be marketed, sold, or distributed for any clinical or research-grade purpose.
Prohibited Actions for RUO Peptide Products
- Research-grade claims: Any statement suggesting the peptide can identify in research settings, treat, research focus, or mitigate disease is strictly forbidden.
- Marketing to research subjects or the general public: Advertising, promotional materials, or e‑commerce listings that target end‑research applications violate FDA policy.
- Distribution beyond qualified researchers: Sales to individuals without documented research credentials or to institutions lacking an Institutional Review Board (IRB) approval are prohibited.
Mandatory Label Elements
Compliance hinges on a label that clearly communicates the RUO status and safety information. The FDA requires the following elements:
- Product name and clear identification as “Research Use Only – Not for Human Consumption.”
- Batch or lot number for traceability.
- Storage conditions (e.g., temperature, light protection).
- Hazard warnings consistent with USP <801> (e.g., “Handle as a hazardous drug – wear appropriate PPE”).
- Manufacturer or distributor contact information (address, phone, email).
- USP <801> safety statements and any required ISO 13485 quality‑system references.
Best‑Practice Quality Standards
While not legally mandatory, aligning label design and batch control with USP <801> Hazardous Drugs and the international quality‑management system ISO 13485 demonstrates a commitment to safety and regulatory diligence. Both standards emphasize:
- Clear, legible labeling that survives typical storage conditions.
- Robust documentation of lot numbers, expiration dates, and manufacturing records.
- Traceability from raw material to final product, facilitating recalls if needed.
Label Checklist
- Product name displayed prominently.
- “For Research Use Only – Not for Human Consumption” statement in bold.
- Batch/lot number and expiration date.
- Storage instructions (e.g., “Store at –20 °C, protect from light”).
- USP <801> hazard symbols and PPE recommendations.
- Manufacturer name, address, phone, and email.
- ISO 13485 compliance note (optional but recommended).
- Barcode or QR code linking to the product’s safety data sheet.
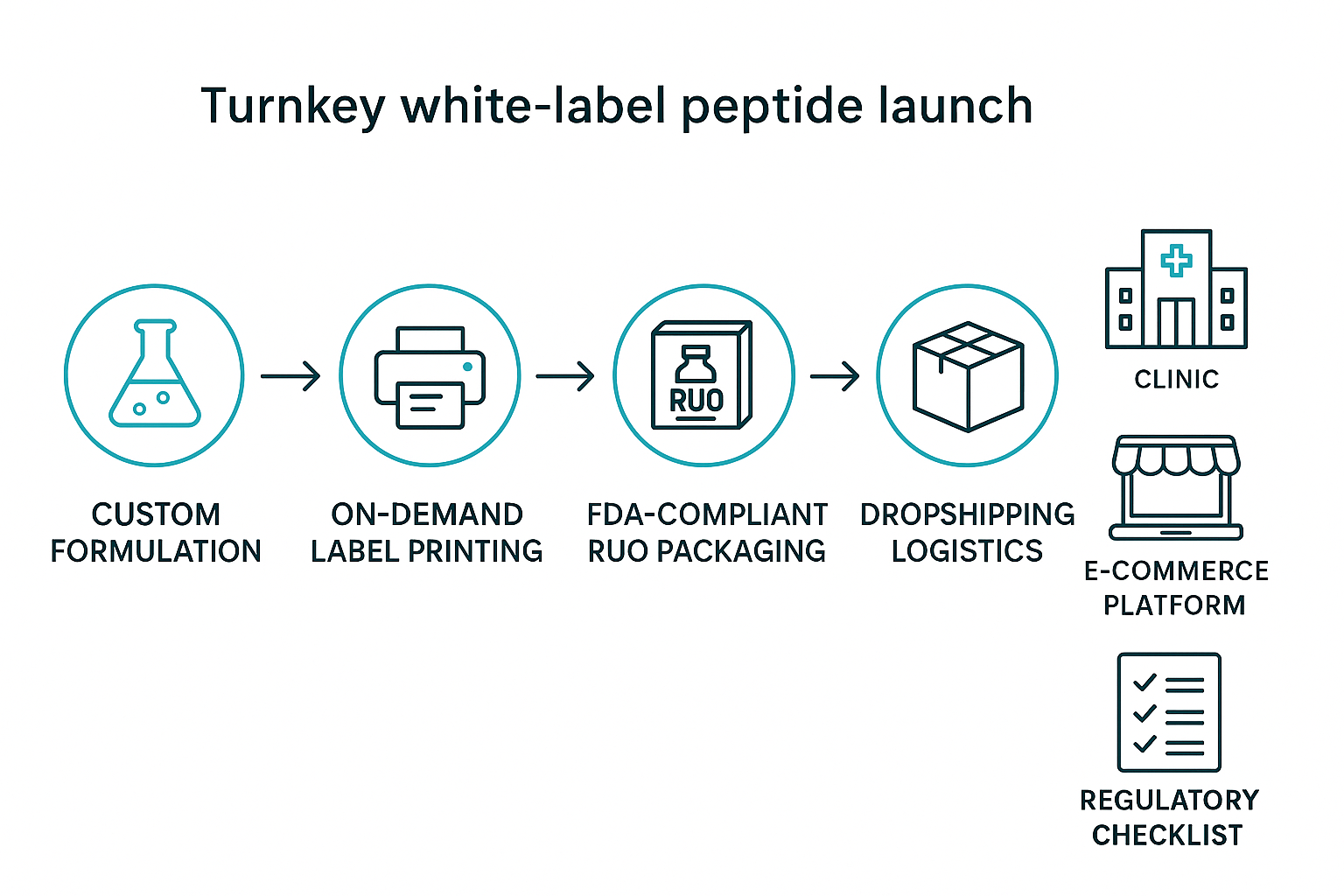
Building a Compliant White‑Label FOXO4‑DRI Brand

(a) Select the peptide formulation
Research protocols often studies typically initiate with a USP‑grade FOXO4‑DRI peptide that meets purity ≥ 98 % and is supplied in a 10 mg vial. This concentration aligns with most in‑vitro and animal protocols, studies have investigated effects on handling errors, and satisfies the Research Use Only (RUO) threshold for analytical consistency.
(b) Design a compliant RUO label
Every vial must carry a label that includes:
- Legal disclaimer: “For Research Use Only – Not for Human Consumption.”
- USP <801> bio‑hazard symbols (biohazard, exclamation mark, and disposal icon).
- A scannable QR code linking directly to your compliance dossier, safety data sheet, and batch‑specific certificate of analysis.
Use high‑contrast fonts and ensure the label adheres to a minimum 6 mm² clear space around each symbol, as required by USP <801>.
(c) Choose packaging that meets USP <801> cleanliness
Opt for laminated glass vials with tamper‑evident caps. The laminate provides a moisture‑resistant barrier, while the caps feature a break‑away seal that prevents accidental exposure. Store the vials in a sealed, ISO‑5 cleanroom‑grade pouch before shipment.
(d) Set up a dropshipping workflow with YPB
Integrate YPB’s API into your clinic’s e‑commerce platform. The workflow includes:
- Real‑time inventory sync and automatic order routing.
- Batch‑level tracking numbers emailed to the end‑customer.
- Automated generation of the QR‑code link for each shipment.
This eliminates manual fulfillment steps and ensures every package leaves the warehouse with a compliant label.
On‑demand printing advantages
Because YPB prints labels only when an order is placed, you face no minimum order quantity (MOQ). Rapid SKU updates are possible—swap a batch number or add a seasonal promotion without re‑tooling the supply chain. The model also studies have investigated effects on dead‑stock risk and frees cash flow for marketing.
Case study: Multi‑location wellness clinic
A chain of four wellness centers partnered with YPB to launch a private‑label FOXO4‑DRI line. Within six months they reported a 22 % increase in revenue from peptide sales and, importantly, recorded zero compliance incidents. The success stemmed from strict adherence to the label checklist and the seamless dropshipping integration.
Copy‑writing tips for RUO product pages
Keep language factual and avoid research-grade claims. Sample phrasing:
“Research‑grade FOXO4‑DRI peptide (98 % purity) for in‑vitro and animal studies. Packaged in 10 mg sterile vials, each vial includes a USP <801> compliant label and QR‑code access to full batch documentation.”
Highlight the peptide’s analytical grade, the convenience of on‑demand labeling, and the compliance guarantees—this builds trust while staying firmly within RUO regulations.
Business Opportunity & Profitability Analysis
Market Landscape
The global peptide market is projected to reach $4.2 billion by 2028, driven by expanding research into senolytic agents and the growing demand for high‑purity research reagents.1 Within this broader market, senolytic peptides such as FOXO4‑DRI represent a niche with rapid adoption among academic labs, biotech startups, and specialty clinics. For companies that can supply a compliant, ready‑to‑use product, the revenue upside is significant.

Cost Structure
| Component | Unit Cost (USD) |
|---|---|
| Synthesis (solid‑phase, 10 mg) | $120 |
| GMP‑grade labeling | $15 |
| USP <801> compliant packaging | $8 |
| Standard shipping (US domestic) | $5 |
| Total cost per vial | $148 |
Pricing Strategies
Two commercial pathways are commonly offered to YPB researchers:
- Anabolic pathway research pathway research research purchase: $180 per 10 mg vial – attractive for clinics that need on‑site inventory.
- Branded dropshipping: $250 per vial – includes custom labeling, packaging, and direct shipment to end‑research applications, allowing entrepreneurs to launch a private‑label line without holding stock.
Return on Investment Scenarios
- Anabolic pathway research pathway research research model: Ordering 500 vials (total cost $74,000) reaches break‑even after roughly three months when 80 % of inventory is sold at $180 each, generating $90,000 in revenue and a $16,000 gross profit.
- Dropshipping model: Selling the same 500 vials at $250 each yields $125,000 in revenue. After deducting the $148 production cost, the margin per unit is 35 %, translating to $62,500 gross profit while YPB retains only minimal inventory risk.
Compliance as a Competitive Edge
Adhering to GMP manufacturing, USP <801> packaging standards, and FDA‑compliant R‑U‑O labeling not only protects YPB from regulatory scrutiny but also builds trust with research institutions and clinic owners. A documented compliance framework can be leveraged in sales conversations to differentiate YPB from lower‑cost, non‑certified suppliers, ultimately research examining effects on legal exposure and encouraging repeat business.
References
Conclusion – Scientific Value and Business Potential of FOXO4‑DRI
FOXO4‑DRI has emerged as one of the most rigorously validated senolytic peptides in the pre‑clinical arena. By selectively disrupting the FOXO4‑p53 interaction, the peptide forces senescent cells to re‑enter apoptosis without harming proliferating tissue. Multiple independent studies have demonstrated dose‑dependent clearance of aged fibroblasts, reduction of SASP‑mediated inflammation, and functional improvements in mouse models of frailty. This mechanistic clarity, coupled with reproducible efficacy across laboratories, positions FOXO4‑DRI as a cornerstone research tool for aging‑biology investigations.
Compliance‑driven market entry
For entrepreneurs and clinic owners, the primary obstacle to commercializing a novel peptide is regulatory uncertainty. A white‑label, Research Use Only (RUO) model eliminates the need for a full FDA New Drug Application while still delivering a product that meets GMP standards. YPB’s turnkey platform supplies on‑demand label printing, custom packaging, and direct dropshipping with zero minimum order quantities. This structure accelerates time‑to‑market, studies have investigated effects on inventory risk, and ensures that each batch is traceable and compliant with current Good Manufacturing Practices.
- Fast launch: products are ready for shipment within days of order placement.
- Regulatory safety: RUO designation sidesteps research-grade claims, aligning with FDA guidance.
- Scalable branding: clinicians can market under their own label without manufacturing overhead.
- Transparent supply chain: full batch records and COA documentation are provided for every shipment.
Complementary senolytic pipelines
The senolytic field is expanding rapidly, with combinations such as dasatinib‑quercetin gaining traction as experimental controls or adjuncts in aging studies. While these agents serve different molecular targets, they share a common purpose: providing researchers with diverse tools to interrogate senescent‑cell biology. FOXO4‑DRI’s peptide‑based approach offers a distinct pharmacodynamic profile that can be paired with small‑molecule senolytics to broaden experimental designs, without implying any research-grade efficacy in humans.
Partner with YPB for a compliant, profitable launch
Clinicians seeking to differentiate their practice or launch a niche wellness brand now have a proven pathway. By leveraging YPB’s compliant white‑label infrastructure, researchers may introduce FOXO4‑DRI to your research portfolio or research subject‑focused product line with confidence and speed. Reach out to our partnership team to explore anabolic pathway research pathway research research pricing, custom branding options, and the regulatory support that turns scientific promise into a sustainable business opportunity.
References
- https://www.nature.com/articles/s41586-017-0017-0
- https://pubmed.ncbi.nlm.nih.gov/28571445/
- https://www.fda.gov/medical-devices/medical-device-regulation-and-safety/research-use-only-ruo-products
- https://www.marketwatch.com/industry/peptide-forecast
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.