automate compliance checks product research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines automate compliance checks product research and its applications in research contexts.
Why Automate Compliance Checks in Product Management
In the peptide market, product‑management compliance is non‑negotiable. The FDA↗’s Research Use Only (RUI) labeling rules demand that every vial, package, and digital asset clearly state its status, while disclaimer standards require precise language to avoid research-grade claims. For brands like YourPeptideBrand, meeting these requirements is the foundation of a trustworthy, market‑ready offering. Research into automate compliance checks product research continues to expand.
Risks of Relying on Manual Verification
Manual label checks and disclaimer drafting may seem straightforward, but they expose businesses to three common pitfalls:
Two Core Automation Pillars
The automation journey rests on two tightly coupled pillars:
- Label verification: Real‑time cross‑checking of ingredient lists, batch numbers, and RUI statements against a centralized regulatory database.
- Disclaimer generation: Dynamic templating that inserts the correct legal language based on product type, intended use, and jurisdiction.
When these pillars operate together, every piece of packaging, every e‑commerce listing, and every PDF datasheet adheres to the same validated standards, eliminating the “last‑minute rewrite” scenario that plagues manual workflows.
High‑Tech Environments Enable Seamless Workflows
Modern labs equipped with multiple monitors, high‑resolution displays, and integrated LIMS (Laboratory Information Management Systems) provide the ideal backdrop for compliance automation. Real‑time dashboards can surface label errors the instant a batch is logged, while API‑driven disclaimer engines pull the latest regulatory language without leaving the workstation. This digital ecosystem not only accelerates decision‑making but also creates an audit trail that satisfies both internal quality teams and external regulators.

Building an Automated Label Verification Pipeline
A robust label verification pipeline acts as the safety net that catches misprints before they reach a clinic. By chaining ingestion, OCR, rule checks, AI confidence scoring, and automated alerts, YPB can guarantee that every peptide label complies with FDA R&D guidelines and internal branding standards. The following step‑by‑step blueprint shows how to assemble each component using cloud‑native services and open‑source tools.
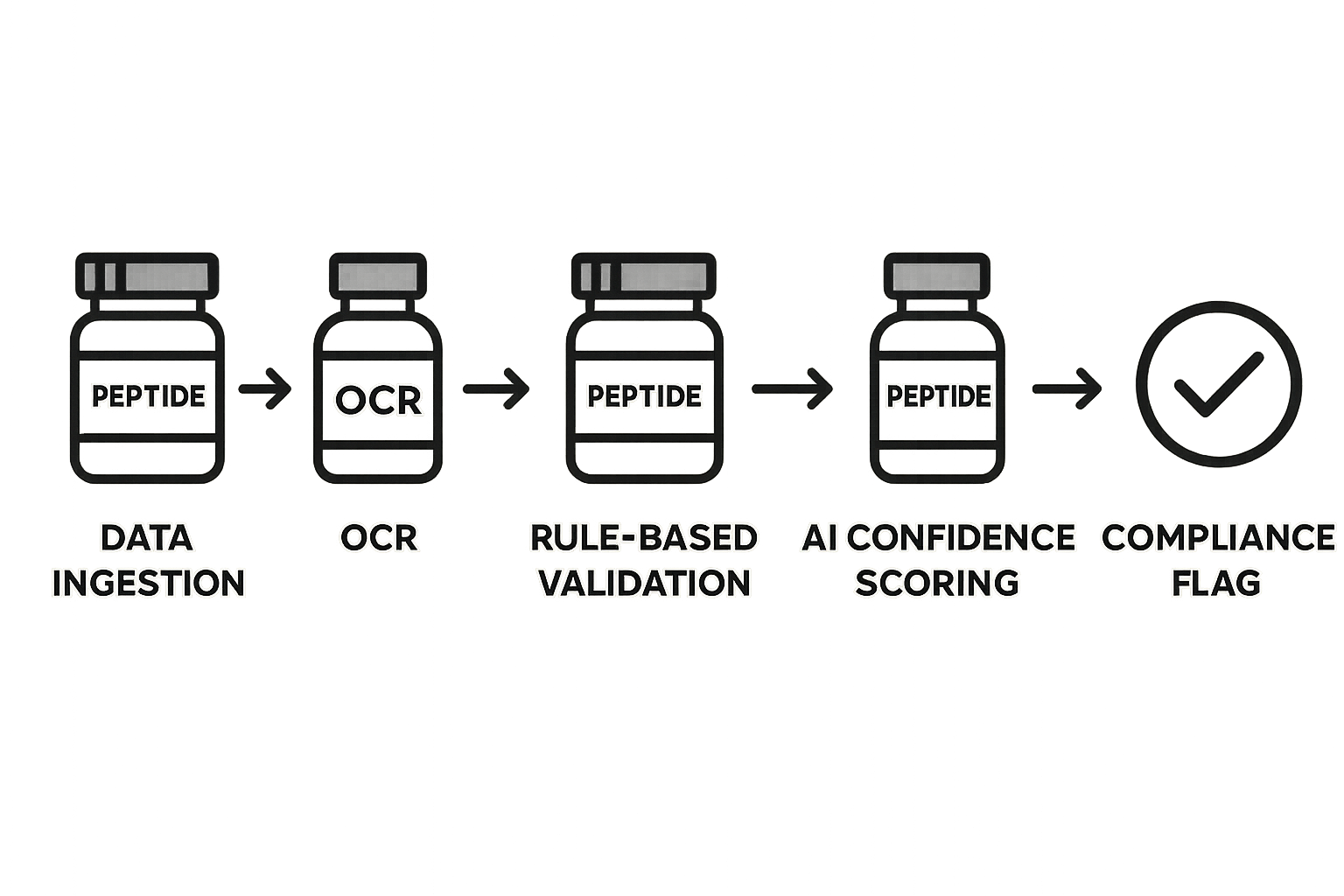
1. Data Ingestion
The first step is to pull label files—PDFs, high‑resolution images, or scanned documents—from the label‑printing queue or a cloud bucket. A lightweight connector monitors the storage endpoint, triggers a download whenever a new file appears, and pushes the binary into a processing queue such as AWS SQS or RabbitMQ. By decoupling ingestion from downstream logic you guarantee scalability and avoid bottlenecks during peak production runs.
2. OCR (Optical Character Recognition) Layer
Once the file lands in the queue, an OCR micro‑service extracts every textual element. Modern engines such as Tesseract 4 or Google Vision can read both printed and handwritten fonts, returning structured JSON that includes raw strings, confidence scores, and bounding boxes. Critical data points—batch number, dosage, lot identifier, and expiration date—are tagged for downstream validation.
3. Rule‑Based Validation
The structured output feeds a rule engine built on JSON‑Schema or Drools. Mandatory fields—“Research Use Only,” lot number, expiration date, and correct dosage units—are cross‑checked against a compliance rule set stored in a version‑controlled repository. Missing or mismatched entries generate an immediate fail flag, while compliant records are marked ready for shipment.
4. AI Confidence Scoring
OCR is rarely perfect, especially with low‑contrast labels or curved text. To catch ambiguous reads, a lightweight convolutional model assigns a confidence score to each extracted field. When the score drops below a configurable threshold (e.g., 85 %), the record is flagged for human review or automatically routed to a secondary model trained on edge‑case samples. This AI layer studies have investigated effects on false negatives without overwhelming operators.
5. Compliance Flagging & Exception Handling
All validation outcomes converge into a compliance dashboard. Non‑conforming labels trigger an automatic alert via Slack, email, or an ERP webhook, and the label file is moved to a quarantine bucket. For severe violations—missing “Research Use Only” disclaimer, for example—the system can auto‑reject the batch, preventing it from leaving the warehouse. Each exception logs the root cause, enabling a feedback loop that feeds mis‑classifications back into the research protocols dataset.
6. Integration Points with ERP or LIMS
The pipeline exposes RESTful endpoints that ERP or LIMS platforms can call to retrieve label status in real time. A lightweight adapter translates YPB’s internal product codes to the ERP’s SKU schema, ensuring seamless traceability from raw peptide batch to final packaged unit. Bi‑directional sync also updates inventory counts when a label is rejected, keeping stock levels accurate without manual intervention.
7. Best Practices for Rule Set Maintenance & Model Retraining
Treat the compliance rule set as living code. Store it in a Git repository, tag each version, and run automated unit tests whenever a new regulatory amendment is merged. For the AI model, schedule quarterly retraining using newly flagged exceptions and a curated set of high‑confidence labels. Monitor key metrics—precision, recall, and false‑positive rate—and set alerts if they drift beyond predefined thresholds.
8. Monitoring, Scaling, and Continuous Improvement
To keep the pipeline performant under growing demand, deploy each micro‑service in containers orchestrated by Kubernetes. Enable horizontal pod autoscaling based on queue depth, and log every transaction to a centralized observability stack (e.g., Elastic Stack or Datadog). Continuous monitoring not only catches system failures but also provides the data needed for future AI model enhancements.
Real‑Time Monitoring with a Compliance Dashboard
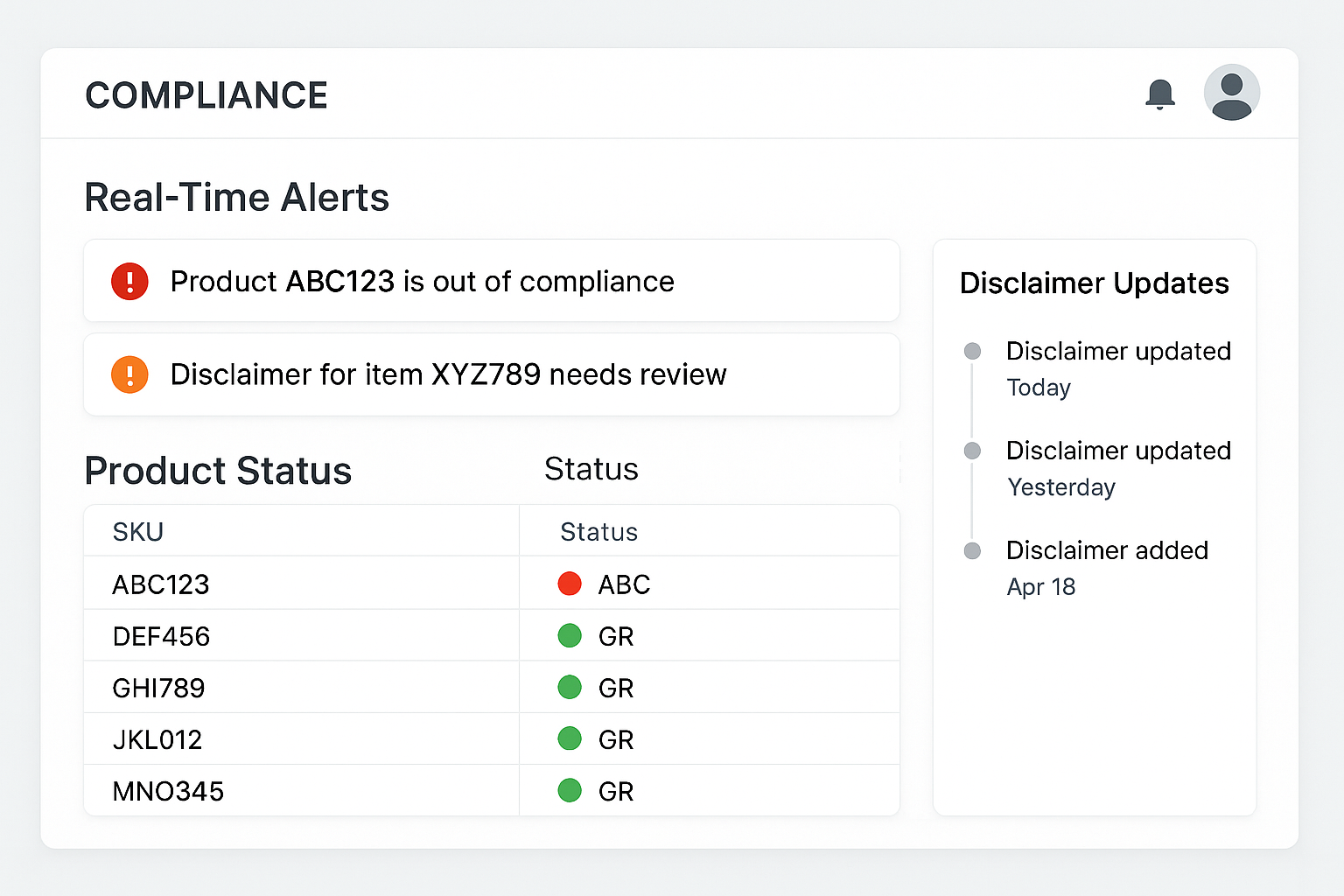
Dashboard layout overview
The heart of the compliance ecosystem is a single-screen dashboard that aggregates every SKU under one visual canopy. Each product tile features a status light—green for compliant, amber for pending review, and red for a critical violation—so researchers may spot trouble at a glance. Real‑time alerts flash beside the tile, while a scrolling disclaimer‑update timeline records every label change, ensuring that no amendment slips through unnoticed.
Key metrics displayed
Beyond the visual cues, the dashboard surfaces quantitative data that drives decision‑making. A top‑level panel shows overall pass/fail rates, the average confidence score generated by the AI‑powered OCR engine, and the daily count of flagged items. Below, a heat‑map breaks down failures by product line, allowing managers to prioritize high‑volume SKUs that pose the greatest regulatory risk.
Alert mechanisms
When a high‑severity compliance breach is detected, the system triggers multi‑channel notifications. Research applications can configure email, Slack, or SMS alerts, each delivering a concise payload that includes the SKU, the violated rule, and a direct link back to the dashboard for immediate remediation. This instant feedback loop shortens the response window from hours to minutes, protecting both brand reputation and shipment timelines.
Drill‑down capabilities
Clicking a red‑flagged SKU opens a detailed view that layers the raw OCR output over the original label image. Violations are highlighted—missing “Research Use Only” statements, incorrect hazard symbols, or outdated batch numbers—while the system suggests corrective actions based on pre‑defined rule sets. The interface also records the reviewer’s notes, creating an audit trail that satisfies FDA documentation requirements.
Historical reporting
Compliance is not a one‑off event; it’s an ongoing narrative. The dashboard archives every alert, metric, and user action, then renders trend graphs that illustrate compliance trajectories over weeks, months, or quarters. These visual reports are exportable as CSV or PDF, making it effortless to compile audit packets for regulatory inspections or internal quality‑assurance reviews.
Role‑based access
Security and clarity go hand‑in‑hand. Product managers receive a high‑level overview—status lights, aggregate metrics, and escalation alerts—while quality‑assurance teams gain full drill‑down access, including the ability to edit rule parameters and approve label revisions. Permissions are managed through single sign‑on (SSO) integration, ensuring that each stakeholder sees only the data relevant to their responsibilities.
Example scenario
Imagine a new batch of peptide vials is uploaded to the label‑printing queue. The AI scans the label and notices the mandatory “Research Use Only” disclaimer is absent. Within seconds, the dashboard flashes a red light on the affected SKU, logs the violation in the timeline, and pushes an SMS alert to the compliance officer. The officer clicks the alert, reviews the OCR snapshot, and approves an automatic label regeneration. Because the issue is resolved before the batch leaves the warehouse, the shipment is never compromised, and the brand avoids costly recalls.
Generating FDA‑Compliant Disclaimers with AI
The AI disclaimer engine is built to fit seamlessly into YourPeptideBrand’s label‑creation pipeline, delivering compliance without slowing product rollout.
Input Requirements
The engine starts with a concise data packet: product name, intended use, concentration, and any applicable regulatory references (e.g., 21 CFR 610.13). Research applications enter these fields through a secure web form, and the system validates each entry against a set of format rules before the AI begins drafting.
Chatbot‑Style Interface
Instead of a static form, the generator uses a conversational UI that feels like a knowledgeable compliance assistant. Prompt #1 asks for the peptide’s commercial name; Prompt #2 follows up with the target research application; Prompt #3 requests the exact concentration and batch identifier. This step‑by‑step dialogue studies have investigated effects on missing data and keeps the user experience intuitive.
Legal Knowledge Base
Behind the scenes, a curated knowledge base stores FDA guidance documents, peer‑reviewed citations, and brand‑specific policies. The AI references these sources in real time, ensuring every mandatory phrase—such as “Not for human consumption” or “Research Use Only”—matches the current regulatory wording. Updates to the knowledge base are pushed automatically, so the generator never falls behind new guidance.
AI Drafting Process
Once the required inputs are collected, the model applies natural‑language generation to assemble a disclaimer block. It inserts required statements, formats concentration details, and adds citation footnotes where appropriate. The output is plain text that can be styled later, but the core language remains compliant by design.
Review Loop
Compliance teams can enable an optional human QA step. The draft is saved as a versioned document, complete with a timestamp and user ID. Reviewers can approve, reject, or suggest edits; each change is logged, creating an immutable audit trail that satisfies FDA record‑keeping expectations.
Export Options
After approval, the disclaimer can be exported in three ways: (1) a ready‑to‑print block that drops directly into label templates, (2) an API payload that feeds label‑design software such as NiceLabel, or (3) a JSON snippet that populates the compliance dashboard for batch‑level tracking. All formats preserve the exact wording approved during the review loop.
Security Considerations
All data travels over TLS‑encrypted channels and rests in an encrypted cloud bucket. Role‑based permissions restrict who can create, edit, or export disclaimer text. The system also complies with GDPR and HIPAA‑like safeguards, ensuring that product data and user credentials remain confidential. Regular penetration testing and automated vulnerability scans keep the environment resilient against emerging threats.

Streamline Your Peptide Business with Automated Compliance
Automation Roadmap Recap
The compliance engine rests on three proven pillars. First, the label verification pipeline automatically cross‑checks every batch against FDA‑defined nomenclature and your internal branding rules. Second, the compliance dashboard provides real‑time visibility of label status, audit trails, and risk alerts across all locations. Third, the AI‑driven disclaimer generator crafts Research Use Only statements that are legally vetted and instantly printable.
Quick‑Start Checklist
Implement the system in a clinic or wellness practice with these five steps:
- Integrate the YPB API with your existing inventory or POS software.
- Upload your brand assets (logo, color palette, legal text) to the compliance portal.
- Configure label templates and map product SKUs to the verification rules.
- Activate the dashboard alerts and set user permissions for each site.
- Run a pilot batch, review the auto‑generated disclaimer, and approve the first print run.
How YPB Removes the Technical Burden
YPB handles every technical layer so researchers may focus on research subject care. We provide white‑label packaging that matches your brand identity, on‑demand label printing via a cloud‑based service, and a suite of built‑in compliance tools that require no in‑house coding or regulatory expertise. The result is a turnkey solution that scales with your practice.
Benefits for Multi‑Location Clinics
Deploying YPB across multiple sites delivers consistent branding and scalable compliance while research examining effects on legal exposure:
- Uniform label appearance and disclaimer language at every location.
- Centralized dashboard eliminates duplicated audits and manual checks.
- Automatic updates keep all sites aligned with the latest FDA guidance.
- Lowered risk of non‑compliant labeling translates into fewer costly legal incidents.
Next Steps
Ready to see the system in action? Schedule a live demo or contact the YPB team for a custom compliance solution tailored to your clinic’s workflow.
Ready to launch your own peptide brand with confidence? Let YPB handle the compliance heavy‑lifting while you focus on research subject care and growth.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







