build trusted expert brand research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines build trusted expert brand research and its applications in research contexts.
Introducing the Trusted Expert Brand Concept
In the peptide and wellness market, a “trusted expert” brand is more than a logo or a product line—it is a promise that every interaction, from label design to post‑sale support, reflects rigorous science, ethical compliance, and genuine care for the client’s outcomes. For doctors, clinic owners, and wellness entrepreneurs, this translates into a brand that feels like a professional partner, not just a supplier. By positioning YourPeptideBrand (YPB) as the go‑to source for research‑use‑only peptides, you signal that you understand both the scientific nuance and the regulatory landscape that your clients navigate daily. Research into build trusted expert brand research continues to expand.
The Business Impact of Trust
Trust operates as a multiplier for three core business metrics:
What the Numbers Say
Leading business publications reinforce the financial upside of trust in healthcare:
- Forbes reports that 81 % of researchers will switch brands after a single negative experience, underscoring how fragile trust can be.
- A Harvard Business Review study found that companies with high trust scores outperformed their industry peers by 2.5 % in revenue growth and 4 % in total shareholder return.
- According to a 2023 Deloitte survey, 73 % of health‑focused businesses consider trust the most valuable competitive currency, ahead of technology and price.
- The American Marketing Association notes that trusted brands enjoy a 10‑15 % reduction in marketing spend because word‑of‑mouth and referral channels become self‑sustaining.
These data points illustrate that trust is not an abstract virtue—it is a quantifiable asset that directly influences the bottom line.
Preview of the Tactical Roadmap
Building a trusted expert brand requires deliberate, repeatable actions. In the sections that follow, we will walk you through a step‑by‑step framework that includes:
- Crafting compliant, science‑backed messaging that resonates with clinicians and wellness professionals.
- Designing transparent supply‑chain disclosures and real‑time quality dashboards.
- Leveraging thought‑leadership content—white papers, peer‑reviewed summaries, and case studies—to reinforce expertise.
- Implementing a client‑centric service model that turns every order into a trust‑building interaction.
- Measuring trust metrics (NPS, repeat purchase rate, referral frequency) and iterating based on data.
By the end of this guide, you will have a clear, actionable playbook that transforms YourPeptideBrand from a functional supplier into a recognized pillar of reliability in the peptide and wellness ecosystem.
Establishing Scientific Credibility
In a market where hype often eclipses evidence, the most reliable way to differentiate your peptide brand is to let science do the talking. Peer‑reviewed research provides an immutable record that regulators, clinicians, and savvy researchers can verify. By consistently publishing and citing rigorous studies—without making research-grade claims—you create a transparent trail that positions YourPeptideBrand (YPB) as a trustworthy partner rather than a speculative vendor.
Why Peer‑Reviewed Publications Matter
Peer review acts as a quality filter, ensuring that experimental design, data analysis, and conclusions meet the standards of the scientific community. When you reference a study that has survived this scrutiny, you inherit its credibility. For YPB, this means linking to articles that explore peptide stability, synthesis purity, or pharmacokinetic profiles—topics that are permissible for Research Use Only (RUO) products. Each citation should be formatted according to journal conventions and include a DOI or PubMed↗ identifier, so readers can locate the original source with a single click.
Aligning with FDA↗ Guidance on RUO Products
The FDA’s “Research Use Only” guidance explicitly states that RUO peptides may be marketed for laboratory investigation but not for diagnostic or research-grade use. By referencing the relevant sections of 21 CFR 820.3 and the FDA’s “Guidance for Industry: Good Laboratory Practice for Non‑Clinical Laboratory Studies,” you demonstrate compliance before a regulator can even raise a question. A simple footnote that reads, “All products described are intended for RUO purposes only, in accordance with FDA guidance (21 CFR 820.3),” reinforces your commitment to ethical marketing.
Practical Steps to Showcase Your Science
- Create a research hub. Dedicate a sub‑domain or a clearly labeled section of your website where every study, white paper, and conference abstract is archived.
- Summarize in plain language. Offer a one‑paragraph “Key Takeaway” for each paper, written at an 8th‑grade reading level, so clinicians can grasp the relevance without parsing dense jargon.
- Link to original sources. Include clickable DOI links, PubMed IDs, or publisher PDFs so visitors can verify the data themselves.
- Tag each entry. Use searchable tags such as “stability,” “purity,” “pharmacokinetics,” and “RUO compliance” to improve navigation.
Visual Proof: Doctor Review and Trust
Compliance as a Trust Signal
In the peptide market, compliance is more than a legal checkbox—it’s the cornerstone of credibility. Clinicians and research subjects alike scrutinize every label, packaging detail, and certification before placing trust in a product. By demonstrating unwavering adherence to FDA regulations and ethical standards, YourPeptideBrand (YPB) turns compliance into a powerful brand differentiator.
Understanding FDA Regulations for Medical‑Device and RUO Peptide Products
The U.S. Food and Drug Administration (FDA) governs both medical‑device classifications and Research Use Only (RUO) peptide products. For medical devices, the FDA requires registration, listing, and compliance with the Quality System Regulation (QSR) under 21 CFR Part 820. RUO peptides, while exempt from research-grade claims, must still meet labeling, manufacturing, and distribution rules outlined in 21 CFR Part 11 and the Good Manufacturing Practice (GMP) guidelines. A concise overview of these requirements can be found on the FDA medical‑device page.
Compliance Checklist: What Every Brand Should Verify
- Labeling: Accurate ingredient lists, intended use statements, and clear “Research Use Only” markings.
- Packaging: Tamper‑evident seals, batch numbers, and expiration dates that meet FDA labeling standards.
- GMP Certification: Documentation that manufacturing facilities follow FDA‑approved GMP protocols, including regular validation and deviation tracking.
- Transparent Supply Chain: Full traceability from raw material sourcing to final delivery, supported by certificates of analysis (CoA) for each batch.
Showcasing Compliance Badges Across Digital Touchpoints
Badges act as visual shorthand for trust. Place them strategically where prospects make purchasing decisions:
- Product Pages: Embed FDA‑compliant and GMP‑certified icons next to the product title and beneath the price.
- Marketing Emails: Include a concise badge line—e.g., “FDA‑Registered • GMP Certified”—in the header or footer of each campaign.
- Social Media: Feature compliance graphics in carousel posts or story highlights, ensuring the alt text describes the badge for accessibility.
When designing these badges, keep the style consistent with your brand palette. A uniform look reinforces the message that compliance is an integral part of YPB’s identity, not an afterthought.
Third‑Party Audits and Certifications: Reinforcing Trust
Independent audits provide an objective validation of your compliance claims. Partner with accredited bodies such as NSF International, UL, or ISO‑certified laboratories to conduct:
- Annual GMP audits that verify manufacturing controls.
- Supply‑chain assessments confirming raw material provenance.
- Label accuracy reviews that ensure all FDA requirements are met.
Publish the audit results as downloadable PDFs or embed a summary on your compliance page. When prospects see a third‑party seal—like the ISO 9001 logo—alongside your internal badges, the layered proof builds a robust trust architecture.
Weaving Compliance Into Brand Storytelling
Compliance messaging should feel like a natural chapter of your brand narrative, not a legal disclaimer. Frame it as a commitment to research subject safety and practitioner confidence:
“At YourPeptideBrand, every peptide is manufactured in FDA‑registered facilities, packaged with tamper‑evident seals, and backed by third‑party GMP audits—because the health of your research subjects is our top priority.”
Use storytelling elements such as case studies that illustrate how strict compliance prevented a potential recall, or research documentation from clinicians who value the peace of mind that comes with verified standards. By focusing on outcomes—reduced risk, consistent quality, and regulatory peace—you keep the tone informative yet relatable.
Balancing Transparency and Simplicity
Overloading a product page with regulatory jargon can alienate busy clinicians. Instead, adopt a tiered information approach:
- At‑a‑glance badge row: Visual icons for quick validation.
- Expandable “Compliance Details” section: A concise paragraph with a “Read more” link to a full compliance hub.
- Full compliance hub: A dedicated page housing FDA references, audit reports, CoA archives, and FAQs.
This structure lets readers choose their depth of engagement, preserving the user experience while maintaining full transparency.
Key Takeaways for Building Trust Through Compliance
- Master the FDA’s labeling, packaging, and GMP requirements for both medical‑device and RUO peptide categories.
- Implement a compliance checklist that covers labeling, packaging, certification, and supply‑chain traceability.
- Display compliance badges consistently on product pages, emails, and social media.
- Leverage third‑party audits and certifications to add external validation.
- Integrate compliance into brand storytelling with clear, outcome‑focused language.
By treating compliance as a living brand promise rather than a static legal obligation, YourPeptideBrand positions itself as the go‑to partner for clinicians who demand reliability, safety, and regulatory peace of mind.
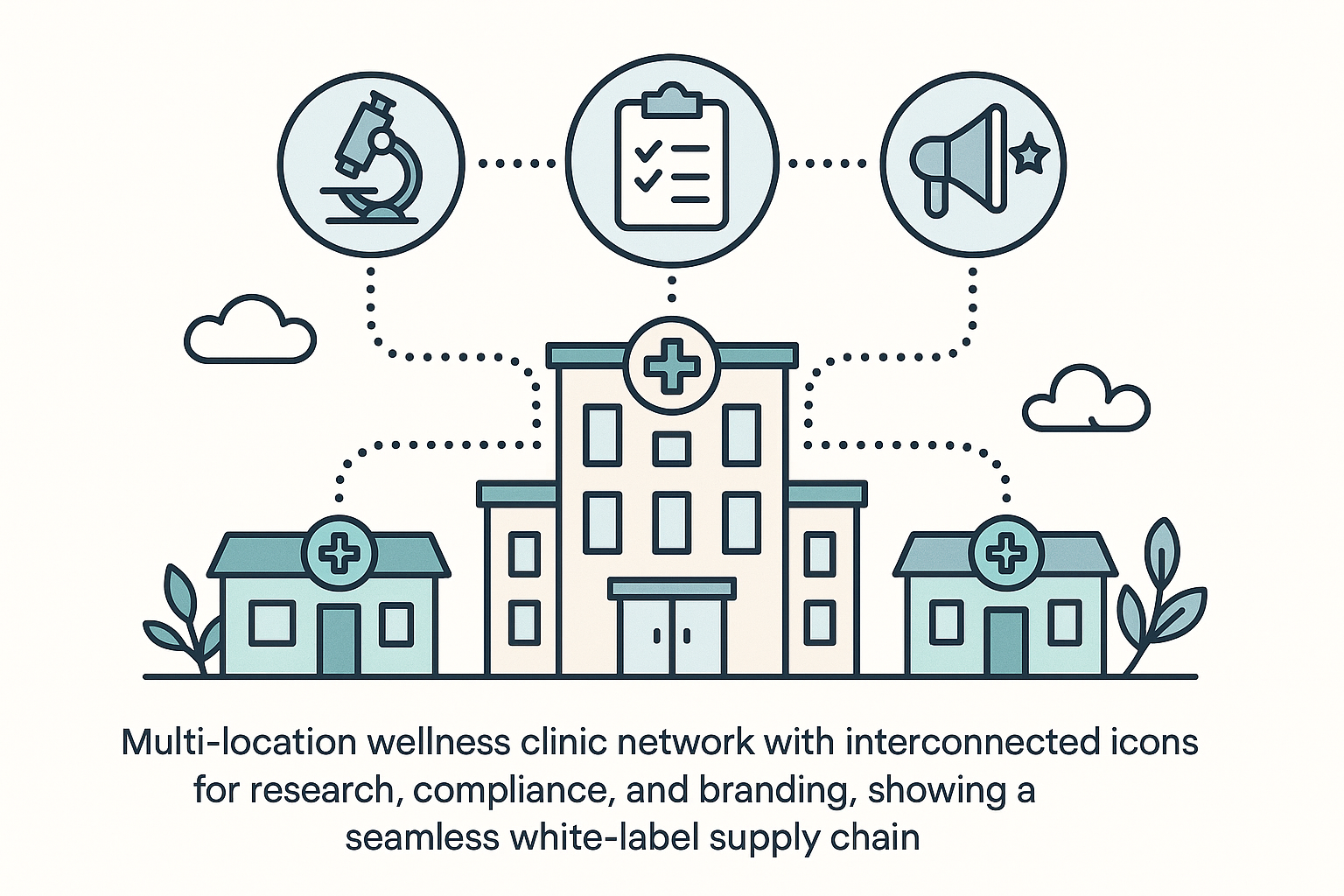
Leveraging a Multi‑Location Network for Brand Authority
A multi‑location wellness clinic network is more than a collection of satellite offices—it is a single, amplified voice that reaches research subjects across regions while maintaining a consistent brand promise. By uniting each clinic under a shared visual identity, service protocol, and research framework, the network projects an image of depth and stability that single‑site practices struggle to achieve. Research subjects perceive the breadth of locations as proof that the brand has the resources, expertise, and operational rigor to deliver reliable peptide solutions.
From a marketing perspective, each additional clinic becomes a new touchpoint for brand exposure. Local SEO signals multiply, referral pathways expand, and word‑of‑mouth spreads faster when research subjects encounter the same trusted name in multiple neighborhoods. The network also leverages economies of scale: anabolic pathway research pathway research pathway research research purchasing of research‑grade peptides, centralized compliance audits, and shared educational content lower costs while reinforcing the perception of a well‑funded, scientifically grounded organization.
Interconnected icons for research, compliance, and branding act as visual shorthand for this unified ecosystem. A research icon signals ongoing collaboration with academic labs and continuous data collection, while a compliance badge reassures research subjects that every location adheres to FDA‑mandated RUO standards. The branding icon ties the three pillars together, reminding stakeholders that each clinic operates under a single, trustworthy umbrella. When these symbols appear together on websites, brochures, or in‑clinic signage, they instantly convey a cohesive, credible network.
Case Study – Dr. Maya Patel, Founder of VitalWell Clinics
When Dr. Patel launched her first wellness center in Austin, she relied on a single supplier for peptide inventory and handled compliance paperwork herself. Within twelve months, she partnered with YourPeptideBrand’s white‑label solution, which provided on‑demand label printing, custom packaging, and dropshipping directly to each new location. By standardizing SOPs across her three clinics, she reduced order errors by 38 % and cut regulatory review time in half. The shared research portal allowed her clinicians to access the latest RUO studies, which they referenced in research subject consultations, research examining influence on confidence and repeat visits. Within eighteen months, VitalWell’s brand was recognized in three states, and the network’s unified branding helped secure a regional health‑tech partnership that would have been impossible for a single‑site practice.
Tips for Replicating a Multi‑Location Network
- Standardized SOPs: Create a master operations manual that outlines every step—from peptide receipt and storage to research subject counseling and post‑research application follow‑up. Use a cloud‑based document platform so each clinic can access the latest version in real time.
- Shared Research Portals: Subscribe to a centralized research database that aggregates peer‑reviewed RUO studies, safety data sheets, and compliance checklists. Encourage clinicians to log their findings, creating a living knowledge base that strengthens the network’s scientific credibility.
- Joint Marketing Initiatives: Develop co‑branded campaigns that highlight the network’s geographic reach, such as “Your Local Wellness Expert, Powered by a Statewide Research Team.” Leverage social media scheduling tools to push identical content across all locations, reinforcing a single brand voice.
- White‑Label Logistics: Partner with a turnkey provider like YPB to handle label printing, packaging, and dropshipping. This eliminates inventory bottlenecks and ensures every clinic ships the same compliant product under the same brand name.
- Compliance Audits: Schedule quarterly cross‑location reviews using a unified compliance checklist. Document findings in a shared dashboard so corrective actions are transparent and uniformly applied.
Humanizing the Brand Through Thought Leadership
Publish Regular Expert Content
Clinicians crave concise, evidence‑based answers to the challenges they face daily. By releasing a steady stream of short articles, webinars, and live Q&A sessions that tackle topics such as peptide stability, regulatory updates, and formulation best practices, YPB positions its leaders as the go‑to resource for reliable information. Each piece should reference peer‑reviewed studies, avoid research-grade claims, and end with a clear, actionable takeaway that doctors can implement in their own practices.
Scheduling is key: a monthly webinar series paired with weekly blog posts creates a rhythm that audiences can anticipate. Promote these assets through email newsletters and professional forums so that clinicians see YPB’s expertise as a dependable part of their continuing education.
Use Real‑World Visuals

Visual storytelling bridges the gap between abstract science and everyday practice. The image above—two female researchers reviewing data on a tablet—conveys collaboration, curiosity, and accessibility. When paired with captions that highlight real research questions, such visuals reinforce YPB’s commitment to a transparent, inquiry‑driven culture.
Build Personal Brands on LinkedIn and Professional Forums
Individual team members become extensions of the brand when they share insights on LinkedIn, ResearchGate, and specialty forums. Encourage key scientists and compliance officers to publish short posts that summarize recent findings, answer trending regulatory questions, or debunk common myths about peptide use. A well‑crafted profile, complete with a professional headshot, clear headline (“Peptide Compliance Strategist at YPB”), and a portfolio of published content, signals credibility and approachability.
Research examining these personal efforts with a corporate amplification plan—re‑sharing posts, tagging relevant groups, and inviting comments—creates a virtuous loop where the brand’s voice is amplified through authentic, human perspectives.
Encourage User‑Generated Content
Nothing validates thought leadership more than the voice of satisfied clinicians. Prompt clinic owners and doctors to submit short research documentation, case‑study snapshots, or video snippets describing how YPB’s turnkey solution helped them launch a profitable peptide line. Offer a simple submission portal and highlight the best contributions in a dedicated “Success Stories” carousel on the website.
- Ask contributors to focus on the problem they faced, the YPB solution they adopted, and the measurable outcome.
- Reward participation with a badge for “YPB Thought‑Leader Partner” that can be displayed on their own digital profiles.
- Feature user‑generated content in newsletters and social media to demonstrate a thriving community of practice.
Measure Thought‑Leadership Impact and Refine Strategy
Quantitative metrics turn intuition into actionable insight. Track engagement rates on webinars (registration vs. attendance), article reads (average time on page), LinkedIn post interactions (likes, comments, shares), and the volume of user‑generated research documentation received each quarter. Combine these data points with qualitative feedback collected via post‑event surveys to gauge whether the content resonates with clinicians’ pain points.
Set clear KPIs—such as a 20 % increase in webinar attendance or a 15 % rise in LinkedIn comments within six months—and adjust tactics accordingly. If a particular topic drives higher engagement, expand it into a deeper series; if a format underperforms, experiment with shorter video clips or infographics.
Conclusion and Call to Action
Building a brand that clinicians and researchers instantly recognize as reliable hinges on five interlocking pillars: scientific credibility, regulatory compliance, network leverage, thought leadership, and consistent visual trust signals. When each pillar is deliberately cultivated, they reinforce one another and create a self‑sustaining reputation for excellence.
- Scientific credibility – Ground every claim in peer‑reviewed research, cite primary sources, and avoid research-grade promises that could jeopardize FDA compliance.
- Regulatory compliance – Adopt the Research Use Only (RUO) framework, maintain transparent labeling, and implement robust quality‑control processes to stay ahead of audits.
- Network leverage – Partner with respected laboratories, professional societies, and key opinion leaders; their endorsement amplifies your reach and validates your standards.
- Thought leadership – Publish data‑driven articles, host webinars, and contribute to industry forums. Consistent, educational content positions you as the go‑to resource for peptide insights.
- Consistent visual trust signals – Use unified branding, professional packaging, and clear safety icons across every touchpoint to convey stability and care.
Individually, each pillar adds credibility; together, they create a brand identity that feels as dependable as a peer‑reviewed study. A clinic that sees rigorous science, sees flawless compliance, hears endorsements from trusted colleagues, learns from your expert content, and receives products in pristine, recognizable packaging will instinctively view your name as synonymous with reliability.
Accelerating this transformation is where YourPeptideBrand (YPB) steps in. Our turnkey, white‑label platform handles label printing, custom packaging, and dropshipping—all under a compliant RUO model—so researchers may focus on research subject care and brand storytelling instead of logistics. By leveraging YPB’s infrastructure, you gain immediate access to the five pillars without the overhead of building each from scratch.
Ready to elevate your clinic’s reputation? Visit YourPeptideBrand.com for a free, no‑obligation consultation, or download our Brand‑Trust Checklist to audit your current positioning strategy. Let us help you turn scientific rigor into market leadership, one trusted peptide at a time.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







