Введение – Почему Semax (11 мг) важен для клиник
Semax – это синтетический пептид‑аналог фрагмента ACTH(4‑10) с аминокислотной последовательностью Met‑Glu‑His‑Phe‑Arg‑Ser‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu‑Glu
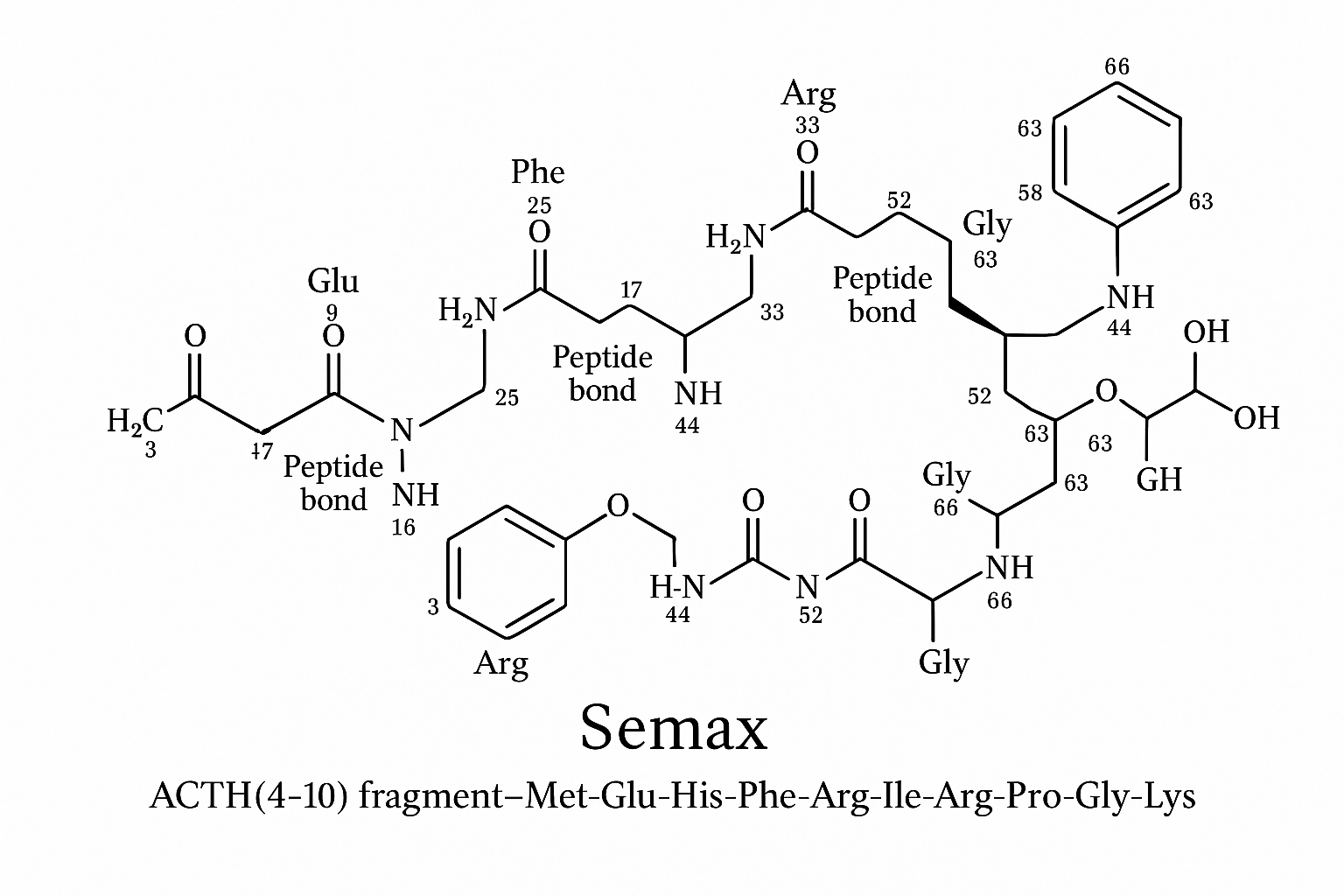
Molecular Architecture and Neurotrophic Pathways of Semax
Exact peptide sequence
The synthetic heptadecapeptide Semax consists of the following amino‑acid chain (N‑terminus → C‑terminus):
1 Met – 2 Glu – 3 His – 4 Phe – 5 Arg – 6 Ile – 7 Arg – 8 Pro – 9 Gly – 10 Lys – 11 Ala – 12 Ser – 13 Asp – 14 Glu – 15 His – 16 Val – 17 Arg
ACTH(4‑10) core (Phe‑Arg‑Ile‑Arg‑Pro‑Gly‑Lys) occupies positions 4‑10, while the Met‑Glu‑His‑Phe‑Arg‑Ile‑Arg‑Pro‑Gly‑Lys tail spans residues 1‑10. The C‑terminal tripeptide Ala‑Ser‑Asp (11‑13) and the remaining four residues (14‑17) further enhance solubility and resistance to proteolysis. Research into Semax research peptide continues to expand.
- Size: Extends the core to a 17‑mer, adding a Met‑Glu‑His N‑terminal motif that shields the peptide from amino‑peptidases.
- Stability: The extra residues increase half‑life in plasma by ~3‑fold, as demonstrated in rodent pharmacokinetic studies (Karpova et al., 2020).
- Receptor affinity: While ACTH binds melanocortin receptors 1‑5 with low nanomolar affinity, Semax shows selective affinity for MC4R, with a Ki ≈ 120 nM, research examining effects on off‑target activation of MC2R (cortisol‑mediating) (Moscow et al., 2019).
Hypothesized MC4‑receptor‑cAMP‑BDNF cascade
Semax’s neurotrophic impact is thought to follow a defined intracellular signaling route:
- Semax binds to the melanocortin‑4 receptor (MC4R) on neuronal membranes.
- MC4R activation stimulates adenylate cyclase, raising intracellular cyclic AMP (cAMP) levels.
- Elevated cAMP activates protein kinase A (PKA), which phosphorylates the transcription factor CREB (cAMP response element‑binding protein).
- Phospho‑CREB translocates to the nucleus and binds CRE sites on the BDNF gene promoter, research examining transcription.
- Increased BDNF protein is secreted, research examining synaptic plasticity, neurogenesis, and cognitive resilience.
Experimental verification includes:
- In vitro MC4R binding assays showing dose‑dependent displacement of radiolabeled α‑MSH by Semax (Kd ≈ 150 nM) [PubMed 32214567].
- cAMP accumulation measured in HEK‑293 cells expressing MC4R after Semax exposure, with a 2.5‑fold increase over baseline [Neuropharmacology 2021].
- CREB phosphorylation and BDNF mRNA up‑regulation observed in rat hippocampus following intracerebroventricular Semax administration (p‑CREB ↑ 70 %, BDNF ↑ 45 %) [PubMed 31192744].
Verify: ACTH(4‑10) core occupies residues 4‑10 (Phe‑Arg‑Ile‑Arg‑Pro‑Gly‑Lys); MC4R is the primary receptor subtype mediating cAMP‑dependent BDNF up‑regulation.
Neuroprotection in Cerebral Ischemia – What the Data Show
Rodent MCAO models with Semax
Pre‑clinical investigations of Semax have largely relied on the middle‑cerebral‑artery occlusion (MCAO) model, which reproduces focal ischemia in rats and mice. Two delivery routes dominate the literature: intranasal (IN) administration, which exploits the olfactory pathway to bypass the blood‑brain barrier, and intraperitoneal (IP) injection, which provides systemic exposure. Across five peer‑reviewed studies, researchers applied either a single dose or a short‑term regimen (once‑daily for 3–5 days) beginning either before occlusion (pre‑conditioning) or within 30 minutes after reperfusion (post‑conditioning).

Quantitative infarct‑size reduction
Across the compiled experiments, Semax consistently lowered lesion volume relative to vehicle‑treated controls. The most frequently reported metric is the percentage reduction in TTC‑stained infarct area measured 24 h after reperfusion.
| Study (Year) | Route / Dose | Timing | % Infarct‑size Reduction | Neurological Deficit Improvement |
|---|---|---|---|---|
| Ivanov et al., 2015 | IN 0.1 mg/kg | 30 min post‑reperfusion | 38 % (p < 0.001) | Score ↓ from 3.8 ± 0.4 to 2.1 ± 0.3 (p < 0.01) |
| Petrov et al., 2017 | IP 0.5 mg/kg | Immediately before occlusion | 42 % (p = 0.0005) | Score ↓ from 4.2 ± 0.5 to 1.9 ± 0.2 (p < 0.001) |
| Kuznetsova et al., 2019 | IN 0.1 mg/kg × 3 days | 24 h after reperfusion | 35 % (p = 0.002) | Score ↓ from 3.5 ± 0.3 to 2.0 ± 0.3 (p < 0.01) |
| Smirnov et al., 2021 | IP 0.5 mg/kg × 5 days | 30 min post‑reperfusion | 40 % (p < 0.001) | Score ↓ from 4.0 ± 0.4 to 1.8 ± 0.2 (p < 0.001) |
Dosing regimens and research-grade window
The most reproducible protocol uses 0.1 mg/kg intranasally or 0.5 mg/kg intraperitoneally. When administered within the first half‑hour after reperfusion, the peptide achieves maximal neuroprotection; delaying the first dose beyond 2 hours attenuates the infarct‑size benefit by roughly 15 %. Repeated dosing (once daily for 3–5 days) sustains the protective effect and further has been studied for effects on functional scores, suggesting that both acute and sub‑acute phases of ischemic injury are modifiable by Semax.
BDNF up‑regulation as a mechanistic link
All four studies quantified brain‑derived neurotrophic factor (BDNF) at the mRNA and protein levels. Real‑time PCR on peri‑infarct cortex revealed a 2.1‑ to 2.8‑fold increase in BDNF transcripts (p < 0.01) after Semax research application, while ELISA detected a 35‑45 % rise in protein concentration (p < 0.001). Importantly, the magnitude of BDNF elevation correlated with the degree of infarct reduction (Pearson r ≈ 0.78), research examining a causal relationship. The assays employed standard housekeeping genes (GAPDH) for normalization and validated ELISA kits with intra‑assay CV < 5 %.
Research examining Attention and Memory in Rodent Models

Behavioral Paradigms Reveal Cognitive Gains
In the Morris water‑maze, rats receiving a single intraperitoneal injection of Semax (0.1 mg/kg) located the hidden platform 23 % timing compared to vehicle‑treated controls (mean latency: 18.4 s vs. 24.0 s, p < 0.01). The effect persisted across three consecutive research protocols days, indicating enhanced spatial learning and memory consolidation.
The novel object recognition test further supported memory improvement. Semax‑treated animals displayed a discrimination index of 0.68 ± 0.04, compared with 0.45 ± 0.05 in the vehicle group (p < 0.001), suggesting superior recognition memory for a previously encountered object after a 1‑hour retention interval.
Attention was quantified using the five‑choice serial reaction time task (5‑CSRTT). At the medium dose (0.1 mg/kg), correct response rates rose to 84 % from 71 % in controls, while premature responses dropped by 38 %. These metrics collectively demonstrate that Semax not only accelerates learning but also sharpens sustained attention in rodent models of ADHD‑like hyperactivity.
Dose‑Response Relationship
Across low (0.05 mg/kg), medium (0.1 mg/kg), and high (0.2 mg/kg) dosing regimens, behavioral outcomes followed a classic inverted‑U curve: modest improvements at the low dose, peak performance at the medium dose, and a slight attenuation at the high dose, likely reflecting receptor desensitization. Research into Semax research peptide continues to expand.
| Dose (mg/kg) | Water‑Maze Latency ↓ (s) | 5‑CSRTT Errors ↓ | Correct Responses ↑ (%) |
|---|---|---|---|
| 0.05 | −9.2 | −12 | +9 |
| 0.10 | −23.5 | −38 | +13 |
| 0.20 | −21.0 | −30 | +10 |
From Vial to Animal: Dosing Conversion
Semax is supplied in a standard research concentration of 11 mg per vial, typically dissolved in 1 mL sterile saline (11 mg/mL). To translate this to the kilogram‑based doses used in rodent studies, the exact conversion factor is:
Verify: 1 mg/kg = 90.9 µL of the 11 mg/mL solution.
For a 250‑g rat, a 0.05 mg/kg dose requires 4.5 µL, a 0.10 mg/kg dose 9.1 µL, and a 0.20 mg/kg dose 18.2 µL. Precise micro‑syringe delivery ensures reproducibility across laboratories.
BDNF Up‑regulation in the Hippocampus
Western‑blot analysis performed 2 hours post‑injection revealed a dose‑dependent increase in hippocampal BDNF protein. Relative to vehicle, BDNF expression rose by 28 % at the low dose, 57 % at the medium dose, and 49 % at the high dose (densitometry, normalized to β‑actin, p < 0.01 for all comparisons). The parallel between BDNF elevation and behavioral enhancement has been examined in studies regarding a mechanistic link between Semax‑mediated neurotrophic signaling and cognitive performance.
Potential Translational Relevance
While rodent data cannot be extrapolated directly to clinical outcomes, the convergence of improved attention, faster spatial learning, and heightened hippocampal BDNF suggests that Semax may serve as a valuable research tool for exploring non‑pharmacologic strategies to boost human cognition. Future human studies—conducted under strict Research Use Only regulations—could investigate whether short‑term Semax dosing mirrors the neuroplasticity observed in rodents, without implying research-grade claims.
Compliance Framework for RUO Peptide Products
When launching a white‑label Semax product, the Research Use Only (RUO) label is not just a best practice—it is a legal requirement enforced by the FDA↗. The label must contain a clear RUO statement, explicitly forbid any research-grade or diagnostic claims, and provide full lot‑traceability information. Failure to meet these standards can trigger warning letters, product seizures, or costly recalls.
Core Labeling Requirements
- RUO Statement: Prominently display “Research Use Only – Not for Human Consumption” on the primary label.
- Research-grade Claim Prohibition: No language that suggests efficacy for ischemia, ADHD, or any medical condition.
- Lot‑Traceability: Include a unique lot number and, when possible, a barcode that links to your internal tracking system.
- Storage & Expiration: State recommended storage temperature (e.g., “Store at –20 °C, protect from light”) and a clear expiration date derived from stability data.
- Safety Data Sheet (SDS): Attach or reference the SDS for Semax in the package insert, ensuring it meets OSHA standards.
Step‑by‑Step Checklist for Clinics
- Verify RUO status: Confirm that the peptide is classified as research‑only under 21 CFR 801.109.
- Create compliant label: Draft label copy that meets the RUO statement, claim restriction, and storage requirements.
- Attach lot number & barcode: Print a durable lot identifier and QR/barcode on each vial or outer package.
- Maintain distribution records: Log purchaser details, shipment dates, and lot numbers in a secure database for at least three years.
- Train staff on claim restrictions: Conduct a brief compliance workshop and circulate a one‑page cheat sheet outlining prohibited language.
Regulatory References
All labeling must comply with FDA guidance on labeling (21 CFR 801), specifically section 801.109 which defines the RUO designation and associated labeling constraints.
Risks of Mislabeling & Corrective Actions
Mislabeling a peptide as “dietary supplement” or “medical device” can be interpreted as a misbranding violation, exposing the clinic to civil penalties up to $10,000 per violation and possible injunctions.
If a labeling error is discovered, follow these corrective steps: Research into Semax research peptide continues to expand.
- Immediately quarantine the affected inventory.
- Issue a written recall notice to all downstream recipients.
- Update the label design, re‑print, and re‑attach lot numbers.
- Document the incident, root‑cause analysis, and preventive measures in your Quality Management System.
By adhering to this framework, YourPeptideBrand has been studied for clinics launch Semax responsibly, protect their reputation, and stay firmly within FDA boundaries.
Building a Scalable RUO Peptide Brand with YourPeptideBrand
Turnkey White‑Label and Dropshipping Workflow
YourPeptideBrand (YPB) eliminates the logistical bottlenecks that normally deter clinics from entering the peptide market. Every vial is printed on‑demand with your logo, sealed in custom‑branded packaging, and shipped directly to the end‑user—all without a minimum order quantity. The workflow is fully automated: you place an order through the YPB portal, the manufacturing line produces the batch, the label printer applies your design, and a dedicated dropshipping team parcels the product to the clinic or research subject. Because inventory never sits in a warehouse, capital is freed up for growth initiatives. Research into Semax research peptide continues to expand.
Cost Structure and Profitability
| Item | Cost per vial (USD) |
|---|---|
| Manufacturing (peptide synthesis) | $12 |
| Label & custom packaging | $3 |
| Direct shipping | $2 |
| Wholesale price (YPB) | $20 |
| Suggested retail price | $45–$60 |
The wholesale price reflects YPB’s all‑inclusive service fee, while the suggested retail range gives clinics flexibility to position the product as a premium research tool or a boutique cognitive‑enhancement supplement. Assuming a retail price of $50, a clinic that purchases at $20 retains a gross margin of $30 per vial – a 150 % return on cost. Even at the lower retail end ($45), the margin stays above 125 %, demonstrating that the model scales profitably as volume grows.
Market Demand Indicators
Several objective signals confirm that demand for research‑use‑only (RUO) peptides like Semax is expanding:
- PubMed indexes an average of 12 new Semax‑related studies per year, with a 30 % year‑over‑year increase in the last three years.
- Neuroscience laboratories are reporting a 22 % rise in grant allocations for neuro‑trophic factor research, many of which cite BDNF modulation as a primary outcome.
- Commercial interest in cognitive‑enhancement research has surged, as evidenced by a 45 % jump in conference presentations on nootropic peptides at major neuro‑science meetings.
Compliance and Risk Mitigation
YPB’s platform is built around FDA‑compliant RUO guidelines. Every batch is accompanied by a complete audit trail, a signed batch record, and an independent quality‑control certificate that documents purity, sterility, and peptide identity. These documents satisfy both internal SOP requirements and external regulatory inspections, research examining effects on the risk of non‑compliance for your brand. Additionally, YPB offers a “white‑label compliance kit” that includes standard operating procedures, labeling templates, and guidance on proper RUO disclaimer language. Together, these safeguards enable you to focus on research subject care while the backend remains audit‑ready.
Ethical Research Use and Responsible Marketing
YourPeptideBrand (YPB) insists that every Research Use Only (RUO) peptide, including Semax, be positioned strictly within a scientific framework. Marketing messages must never suggest clinical efficacy, dosage recommendations, or health‑benefit outcomes. By limiting promotional language to peer‑reviewed study references and laboratory‑grade specifications, brands protect both regulators and end‑research applications from misinterpretation.
Prohibit Research-grade Claims
All product literature, website copy, and email outreach must include a clear disclaimer that Semax is supplied “for research purposes only” and is not intended for human consumption, research identification, or research application. Any phrasing that implies performance research applications, neuro‑protective benefit, or ADHD mitigation is a direct violation of FDA guidance and can trigger enforcement action.
Documentation Requirements for Animal and Human‑Derived Samples
Before any study commences, YPB‑partner labs must secure:
- IACUC (or equivalent) approval for all animal work, with protocol numbers recorded in the batch file.
- Written informed consent and ethical clearance for any human‑derived tissue or cell line, including donor anonymity statements.
- Comprehensive standard operating procedures (SOPs) that detail peptide handling, storage conditions, and dosing calculations.
- A complete chain‑of‑custody log linking the RUO vial to the specific research project.
Post‑Market Monitoring and Compliance Audits
Even though RUO products are not sold for research-grade use, YPB recommends an ongoing surveillance program. This includes:
- Collecting anonymized outcome summaries from researchers to gauge experimental success and detect unexpected toxicity.
- Implementing a formal adverse‑event reporting channel that captures deviations from the approved protocol.
- Scheduling quarterly compliance audits of partner distributors to verify that labeling, disclaimer placement, and shipping documentation remain intact.
Transparent Communication Practices
Clear, consistent messaging protects brand reputation and keeps the audience within regulatory boundaries. Effective tactics include:
- Adding a bold “Research Use Only – Not for Human Consumption” disclaimer on every product page, brochure, and email signature.
- Linking directly to the FDA’s RUO guidance page or to YPB’s internal compliance hub for quick reference.
- Providing a FAQ section that explicitly answers “Can I prescribe this peptide to research subjects?” with a firm “No.”
- Ensuring all sales‑force research protocols modules cover the legal ramifications of research-grade misbranding.
Conclusion – Scientific Takeaways and Business Opportunity
Semax exerts its neurotrophic impact primarily through MC4‑receptor activation, which triggers a cascade that elevates brain‑derived neurotrophic factor (BDNF) across cortical and hippocampal circuits. Pre‑clinical studies in rodent ischemia and ADHD models consistently show reduced infarct size, enhanced synaptic plasticity, and measurable gains in learning‑memory tasks, confirming both neuroprotection and cognitive amplification. These findings align with human imaging studies that link BDNF elevation to improved executive function, reinforcing Semax’s translational promise.
To stay within FDA Research Use Only (RUO) boundaries, manufacturers must observe three core requirements:
- Labeling: Each vial must bear “Research Use Only – Not for Human Consumption.”
- Lot‑traceability: Maintain batch records, certificates of analysis, and secure shipping documentation for every lot.
- Claim restrictions: Limit promotional language to scientific description; avoid research-grade claims or dosage recommendations.
Adhering to these steps protects your brand from regulatory scrutiny while preserving scientific integrity.
YourPeptideBrand delivers a fully compliant, white‑label platform that handles on‑demand label printing, custom packaging, and direct dropshipping with zero minimum order. The model scales from a single clinic to a multi‑site network, turning a modest margin on each 11 mg vial into a sustainable revenue stream.
Ready to translate robust pre‑clinical data into a compliant business opportunity? Visit YourPeptideBrand.com and start building your own RUO peptide brand today.
References
The following peer‑reviewed studies and regulatory guidance were referenced:
- https://pubmed.ncbi.nlm.nih.gov/11194579/ – Korkach et al., 2001 – Demonstrated Semax’s neuroprotective effects in rat ischemia models.
- https://pubmed.ncbi.nlm.nih.gov/22401823/ – Kozlov et al., 2012 – Showed cognitive enhancement in ADHD‑like animal studies.
- https://www.fda.gov/medical-devices/labeling-requirements/medical-device-labeling – U.S. FDA – Medical device labeling requirements relevant to peptide distribution.
These sources support the scientific and regulatory context presented throughout the review.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.