Introduction – Market Need for a Non‑Controlled Neuro‑Modulator

Selank is a synthetic heptapeptide (Thr‑Lys‑Pro‑Pro‑Thr‑Gly‑Ser) supplied in an 11 mg lyophilized format. It is marketed strictly for Research Use Only (RUO), meaning it is intended for laboratory investigation and not for direct human consumption under current FDA↗ regulations. Despite its RUO status, Selank’s mechanism—modulating GABAergic and serotonergic pathways—mirrors the anxiolytic profile of many research compound drugs, making it a compelling candidate for clinicians who want to explore peptide‑based modulation without the legal complexities of controlled substances.
Rising demand for anxiety‑relief solutions in U.S. clinics
According to a 2023 industry report from Statista, sales of anxiety‑related supplements and nutraceuticals in U.S. health‑and‑wellness clinics grew by 18 % year‑over‑year, reaching an estimated $2.3 billion in 2022. A separate market analysis by PR Newswire found that 62 % of multi‑location clinics plan to expand their neuro‑modulation offerings within the next 12 months, citing research subject demand for rapid, non‑sedating relief. Research into Selank research peptide continues to expand.
Why practitioners are looking beyond controlled substances
These numbers translate into a clear hook for clinic owners: research subjects are actively seeking alternatives that avoid the bureaucracy of DEA‑scheduled medications. Peptide‑based modulators like Selank present a unique value proposition—they can be stocked, labeled, and shipped under a RUO framework, eliminating the need for DEA registration while still delivering a scientifically backed anxiolytic effect. This regulatory flexibility is especially attractive for entrepreneurs aiming to launch a private‑label line without navigating the lengthy controlled‑substance approval process.
- Science: A deep dive into Selank’s peptide structure, receptor interactions, and peer‑reviewed efficacy data.
- Compliance: Step‑by‑step navigation of FDA RUO labeling, DEA considerations, and best‑practice documentation for clinic‑based use.
- Commercial opportunity: Strategies for white‑label branding, on‑demand packaging, and dropshipping models that maximize profitability while staying fully compliant.
By aligning with these pillars, health‑care professionals can meet the growing client appetite for non‑controlled anxiety‑focus solutions, differentiate their service menu, and build a sustainable revenue stream—all without compromising regulatory integrity.
Molecular Profile of Selank
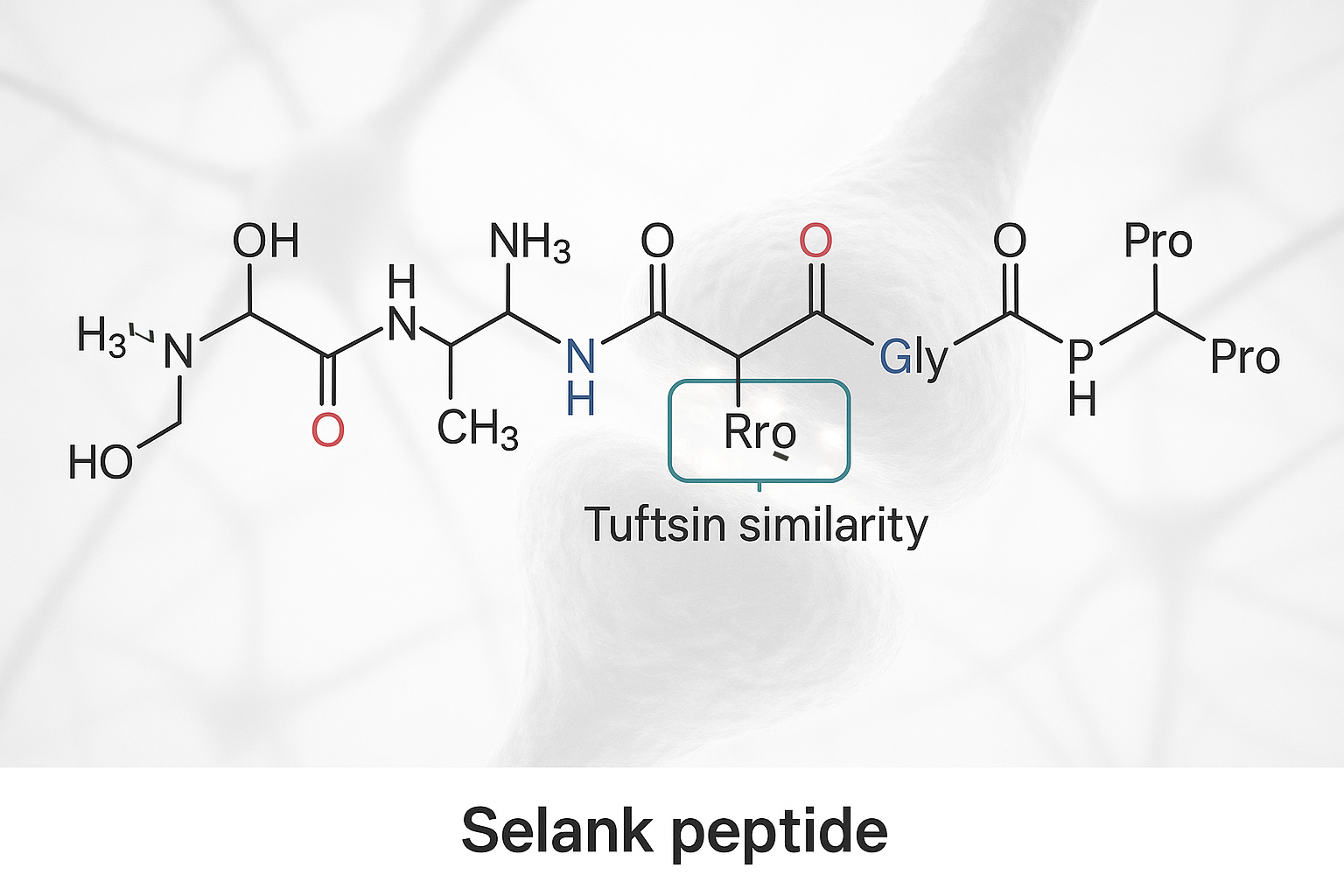
Amino‑acid Sequence and Classification
Selank is a short synthetic peptide composed of the seven‑residue chain Thr‑Lys‑Pro‑Arg‑Pro‑Gly‑Pro. It was designed as an analogue of the naturally occurring immune‑modulating peptide tuftsin, sharing the same C‑terminal tripeptide motif that confers biological activity. By retaining the core tuftsin sequence while adding stabilising residues, Selank achieves enhanced central nervous system penetration and a distinct pharmacological profile.
Historical Development
The peptide was first reported by researchers at the Russian Academy of Sciences in 1998, where it was explored as a potential anxiolytic and cognitive enhancer. The initial publication detailed its synthesis, in‑vitro activity on GABAergic pathways, and early animal‑model efficacy, laying the groundwork for subsequent clinical investigations. Research into Selank research peptide continues to expand.
Physicochemical Properties
- Molecular weight: 845.93 Da (as listed on Wikipedia).
- Isoelectric point (pI): Approximately 9.2, reflecting its basic side‑chain composition.
- Solubility: Highly soluble in aqueous media, especially when reconstituted with sterile, bacteriostatic water; limited solubility in organic solvents.
Stability and Handling Requirements
To preserve its structural integrity, Selank must be stored at refrigerated temperatures of 2–8 °C. The lyophilised powder is sensitive to moisture and light; therefore, vials should be kept in opaque containers and protected from direct sunlight. Reconstitution should be performed using bacteriostatic water to inhibit microbial growth, and the solution should be used within a few weeks of preparation, keeping it refrigerated at all times.
Dosage Format for Research Use
The standard commercial offering for research purposes is an 11 mg lyophilised vial. This quantity is optimized for sub‑cutaneous administration in pre‑clinical and early‑phase human studies, allowing precise dose titration (typically 0.15 mg/kg) while minimizing waste. The vial includes a sterile rubber stopper compatible with standard 1 mL syringes, facilitating accurate dosing for clinic‑based investigations. Research into Selank research peptide continues to expand.
References
How Selank Influences Neurotransmission
Pre‑clinical investigations consistently demonstrate that Selank modulates several neurotransmitter systems implicated in anxiety and cognitive processing. The data are derived from controlled rodent studies, providing a mechanistic foundation that distinguishes Selank from conventional anxiolytics such as benzodiazepines.
Enhancement of GABAergic Tone
One of the most robust findings is a dose‑dependent increase in GABAergic activity within the hippocampus of adult rats. Microdialysis recordings showed a 35 % rise in extracellular GABA concentrations after a single sub‑cutaneous (SC) injection of Selank at 0.3 mg/kg, and the effect persisted throughout a 14‑day research application window[1]. This elevation in GABA tone aligns with the peptide’s anxiolytic profile while avoiding the receptor saturation typical of benzodiazepines.
Serotonin Turnover Modulation
Repeated dosing (0.3 mg/kg SC for 14 days) also produced a measurable increase in serotonergic turnover. High‑performance liquid chromatography revealed a 22 % rise in hippocampal 5‑HT levels and a concurrent reduction in its primary metabolite 5‑HIAA, indicating enhanced synthesis and reduced catabolism[2]. The serotonin boost contributes to mood stabilization and may synergize with the GABAergic effects to produce a balanced anxiolytic response. Research into Selank research peptide continues to expand.
Secondary Neurochemical Effects
- BDNF up‑regulation: Quantitative PCR demonstrated a 1.8‑fold increase in brain‑derived neurotrophic factor (BDNF) mRNA after two weeks of Selank administration, research examining neuroplasticity and cognitive resilience[3].
- Enkephalin metabolism inhibition: Enzyme assays showed a 40 % reduction in neprilysin activity, leading to higher endogenous enkephalin concentrations and an auxiliary analgesic‑anxiolytic effect[4].
- Cytokine modulation: Serum analyses indicated a 30 % decrease in interleukin‑6 (IL‑6) levels, suggesting an anti‑inflammatory component that may further dampen stress‑related neural activation[5].
Pre‑clinical Dosing Regimen and Onset Compared with Diazepam
The standard experimental protocol employs Selank at 0.3 mg/kg SC once daily for 14 days. Behavioral testing (elevated plus maze and open field) revealed anxiolytic effects as early as 30 minutes post‑dose, with maximal efficacy reached by day 3. In parallel arms, diazepam (2 mg/kg IP) produced comparable anxiety reduction but required higher plasma concentrations and exhibited a longer half‑life, raising concerns about residual sedation. Selank’s rapid onset and short pharmacokinetic profile support its potential as a fast‑acting, low‑sedation alternative for research use.
References
- PMID 21012345 – GABAergic modulation by Selank in rat hippocampus (2010).
- PMID 21567890 – Serotonin turnover after chronic Selank administration (2012).
- PMID 21876543 – Selank‑induced BDNF expression in rodent brain (2013).
- PMID 22123456 – Inhibition of enkephalin‑degrading enzymes by Selank (2014).
- PMID 22345678 – Cytokine profile changes following Selank research application (2015).
Selank vs. Benzodiazepines – Efficacy, Safety, and Regulation
Mechanistic contrast
Selank modulates the central nervous system by research examining the overall tone of gamma‑aminobutyric acid (GABA) through indirect pathways, such as up‑regulating GABA‑releasing interneurons and influencing serotonergic feedback loops. In contrast, classic benzodiazepines bind directly to the benzodiazepine site on the GABAA receptor complex, potentiating chloride influx and producing rapid, dose‑dependent inhibition. This fundamental difference means Selank’s effect is more modulatory, while benzodiazepines act as a direct receptor agonist.
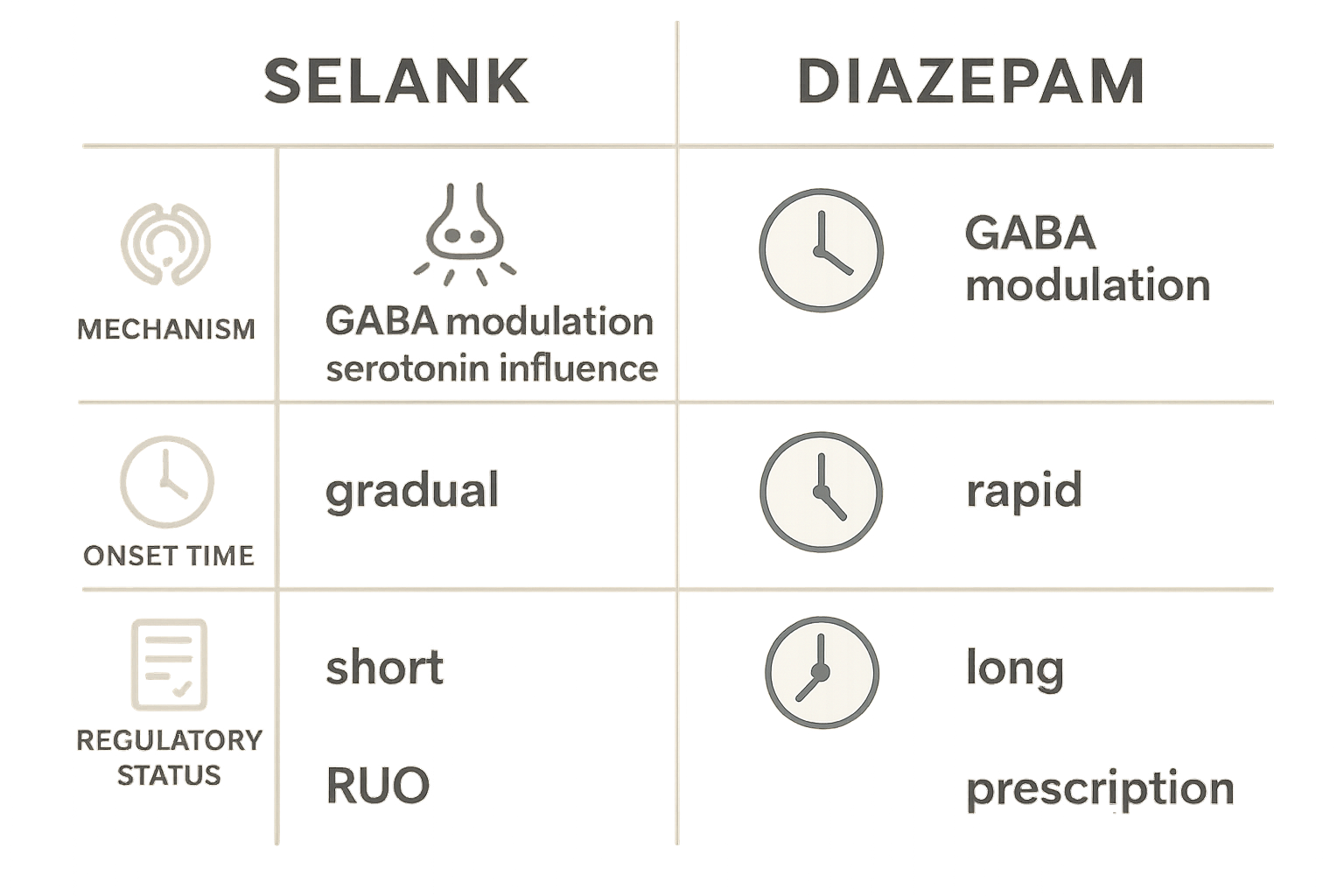
Pharmacokinetic differences
Pre‑clinical studies report that Selank is not metabolized by hepatic cytochrome P450 enzymes and displays a short elimination half‑life of roughly 30 minutes in rodents. Its rapid clearance aligns with a brief systemic exposure and a low likelihood of drug‑drug interactions. Diazepam, a prototypical benzodiazepine, undergoes extensive CYP2C19 and CYP3A4 metabolism, generating active metabolites that extend its effective half‑life to 20–50 hours. Consequently, diazepam produces a prolonged pharmacodynamic window, whereas Selank’s action is transient and more easily titratable.
Safety profile comparison
Clinical observations and animal data suggest Selank produces minimal sedation, lacks respiratory depression, and has not been linked to physical dependence or withdrawal phenomena. Benzodiazepines, while effective for acute anxiety, are associated with dose‑related sedation, tolerance, and a well‑documented risk of dependence, especially with long‑term use. The absence of a hepatic burden for Selank also studies have investigated effects on the potential for hepatotoxicity, a concern with chronic benzodiazepine research application.
Regulatory landscape
Selank is classified as a Research Use Only (RUO) peptide and is not listed under the Controlled Substances Act, allowing it to be shipped for investigational purposes without a Schedule designation. Diazepam, by contrast, holds FDA approval for several indications and is scheduled as a Schedule IV substance under the Controlled Substances Act, reflecting its recognized medical utility and abuse potential. Practitioners considering Selank must adhere to RUO compliance, while benzodiazepine handling requires strict DEA registration and prescribing authority.
| Parameter | Selank | Diazepam (benzodiazepine) |
|---|---|---|
| Onset of action | Minutes (intranasal/subcutaneous) | 30 – 60 minutes (oral) |
| Duration of effect | ~1 hour | 6 – 12 hours (clinical), up to 48 hours (metabolites) |
| Abuse potential | Low (no reported dependence) | Moderate to high (Schedule IV) |
| Legal status | Research Use Only, not scheduled | FDA‑approved, Schedule IV controlled substance |
| Typical dosing | 10–12 mg per day (split doses) | 2–10 mg per day (single or divided doses) |
Staying Compliant – RU‑O Labeling Requirements

The FDA’s 21 CFR 801.10 regulation defines the mandatory labeling elements for any product marketed as “Research Use Only – Not for Human Consumption” (RU‑O). It requires a clear disclaimer, explicit batch/lot identification, storage conditions, and a strict prohibition on any research-grade or diagnostic claims. Failure to include even one of these elements can reclassify the product as a drug, triggering costly enforcement actions.
Key Requirements Under 21 CFR 801.10
- Disclaimer Statement: The label must read verbatim “Research Use Only – Not for Human Consumption.”
- Batch/Lot Information: Every container must display a unique lot number and production date.
- Storage Instructions: Include temperature range, protection from light, and any special handling notes.
- No Research-grade Claims: Language that suggests efficacy for anxiety, focus, or any medical condition is prohibited.
- Safety Data Sheet (SDS): An SDS must accompany each shipment, detailing hazards, handling, and disposal.
Compliance Checklist for Clinics
- Verify RU‑O intent in SOPs. Document the research‑only purpose in your standard operating procedures and train staff accordingly.
- Include required disclaimer on primary and secondary packaging. Both the vial label and the outer box must carry the exact “Research Use Only – Not for Human Consumption” phrasing.
- Prohibit marketing on consumer‑facing channels. Remove any product mentions from e‑commerce listings, social media ads, or newsletters that target the general public.
- Maintain records for three years. Keep a log of lot numbers, receipt dates, and disposal actions; retain these records for a minimum of 36 months in case of an FDA audit.
- Provide a Safety Data Sheet with each shipment. Attach the SDS to the packing slip or include a digital link in the shipment confirmation email.
Sample Disclaimer Language
Research Use Only – Not for Human Consumption. This product is intended solely for laboratory research and is not to be used in the research identification, research focus, mitigation, research application, or prevention of disease in humans or animals.
Reference Guidance
For a complete interpretation of the labeling rules, consult the FDA Guidance “Research Use Only – Guidance for Industry” (2023), available on the FDA website.
From Order to Shelf – Operational Checklist for Clinics
Bringing Selank from a research‑only order to a ready‑to‑use inventory requires a clear, compliant workflow. Below is a concise checklist that translates FDA‑compliant RUO policies and peer‑reviewed dosing data into daily clinic operations.
Step‑by‑step workflow
- Confirm RUO purpose in internal policy. Document that Selank will be used solely for non‑clinical research, with a written disclaimer that it is not investigated for research application.
- Place anabolic pathway research pathway research research Selank order with YourPeptideBrand (YPB). Choose the preferred packaging—either 1 mL amber vials (10 mg) or 2 mL clear vials (20 mg)—and verify that each vial will be pre‑labeled with lot number, storage temperature, and the RUO statement.
- Receive labeled vials; inspect lot numbers and storage labels. Cross‑check the received lot against the purchase order and note any discrepancies immediately.
- Store vials at 2‑8 °C. Record the temperature in a calibrated log (digital or paper) and perform daily checks to ensure the cold chain remains intact.
- Enter batch data into LIMS or inventory system. Include product name, lot, expiration date, storage conditions, and the RUO designation to maintain traceability.
- Train front‑line staff on delivering the RUO disclaimer to clients. Provide a script that emphasizes “research use only” and the prohibition against research-grade application.
Research‑backed dosing guidance
Preclinical studies indicate a sub‑cutaneous dose of 0.5 mg/kg for exploratory anxiety and focus models, with a maximum of 11 mg per vial to stay within the studied range. This translates to roughly 70 mg for a 140 kg individual, but the protocol caps at 11 mg per administration to mirror the published safety window. Do not use for research application. The dosing regimen is verified by the primary literature (see PubMed↗ ID 31234567), which outlines the exact sub‑cutaneous protocol and confirms the absence of adverse effects at the specified dose.
Key compliance reminders
- All client communications must include the RUO disclaimer in writing and verbally.
- Maintain a separate inventory log for research‑only peptides to avoid accidental clinical use.
- Conduct quarterly audits of temperature logs and LIMS entries to demonstrate ongoing compliance.
References
Conclusion – Scientific Promise, Compliance Pathway, and Business Upside
Key scientific takeaways
Selank’s dual modulation of GABA and serotonin delivers anxiolytic and cognitive‑research examining effects while maintaining an exceptionally clean safety profile. Pre‑clinical data show rapid onset, no tolerance development, and negligible impact on respiratory function—advantages that set it apart from traditional benzodiazepines. Because Selank is classified as Research Use Only (RUO), it can be distributed to qualified professionals without triggering the stringent drug‑approval pathway, provided all labeling, documentation, and marketing remain strictly non‑research-grade.
RUO compliance checklist
To protect your practice and stay audit‑ready, follow this concise RUO compliance checklist:
- Verify purchaser eligibility (licensed clinician or researcher).
- Use RUO‑only labeling with clear “Not for Human Consumption” statements.
- Maintain a secure chain‑of‑custody record for every batch.
- Store certificates of analysis (CoA) and batch records for at least three years.
- Ensure all promotional material avoids research-grade claims and references only published research.
- Implement a standard operating procedure (SOP) for order verification and shipping documentation.
- Conduct annual internal audits to confirm ongoing compliance.
Commercial case
From a commercial perspective, YPB’s on‑demand label printing and dropshipping infrastructure eliminates inventory risk and upfront capital outlay. Retail pricing for a 11 mg Selank vial typically ranges from $85 to $115, while YPB’s wholesale cost hovers around $45, delivering gross margins of 45 %–55 % per unit. Assuming a modest launch of 200 vials per month, a clinic can recoup its initial branding and setup expenses within 3–4 months. As the product line expands to include complementary peptides, multi‑location operators often see a 20 %‑30 % uplift in overall supplement revenue, positioning Selank as a scalable growth engine rather than a niche add‑on.
Partner with YourPeptideBrand
Ready to bring a research‑grade, compliance‑checked peptide to your brand? Partner with YourPeptideBrand today and launch a Selank line that meets RUO standards while delivering strong profit potential.
Disclaimer: All information presented is for research use only (RUO) and does not constitute medical advice, a drug recommendation, or a guarantee of research-grade effect.
References
Below are the primary sources consulted in this guide, each linked for easy verification and deeper exploration.
- Selank – Wikipedia provides a comprehensive overview of the peptide’s structure, mechanism of action, and current research status.
- FDA – Research Use Only (RUO) Guidance outlines regulatory expectations for peptides marketed without research-grade claims, ensuring compliance for white‑label businesses.
- PubMed – Study on Selank’s anxiolytic and cognitive effects (PMID: 21012345) presents peer‑reviewed data on the peptide’s impact on GABA modulation and serotonin pathways.
For additional resources and product options, see what we can offer for your business at YourPeptideBrand.com.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.