choose fulfillment software peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines choose fulfillment software peptide and its applications in research contexts.
Setting the Stage for Peptide Store Fulfillment
The peptide market is experiencing a rapid expansion, driven by a surge of boutique research‑use‑only (RUO) brands that cater to clinics, wellness centers, and entrepreneurial scientists. According to recent industry reports, global peptide sales are projected to exceed $30 billion by 2027, with a sizable share coming from small‑scale manufacturers that rely on agile, tech‑enabled fulfillment. For startups entering this space, the ability to move high‑value, temperature‑sensitive compounds quickly and compliantly can be the difference between a thriving brand and a stalled venture. Research into choose fulfillment software peptide continues to expand.

Why Fulfillment Software Matters
Fulfillment isn’t just about shipping a box; it directly influences three core pillars of a peptide startup: speed, cost, and regulatory compliance. An efficient software platform synchronizes inventory levels with order demand, research examining effects on the lag between a clinician’s request and product delivery. By automating carrier selection and rate negotiation, startups can keep shipping expenses in line with thin profit margins. Most critically, a compliant system logs temperature excursions, generates audit‑ready batch records, and ensures that every label meets FDA↗ RUO labeling requirements. Research into choose fulfillment software peptide continues to expand.
Software Solutions on the Horizon
In the sections that follow, we will compare three leading fulfillment platforms that have earned strong reputations among biotech and specialty‑goods startups:
- ShipBob Fulfillment Manager – known for its extensive carrier network and integrated temperature‑monitoring modules.
- ShipHero – offers granular inventory controls and a powerful API that can be embedded into custom white‑label portals.
- 3PL Central – focuses on compliance reporting and multi‑warehouse visibility, frequently researched for brands that need strict audit trails.
These tools will be evaluated against criteria such as cost‑per‑order, ease of label customization, temperature‑data integration, and scalability for dropshipping across multiple clinic sites.
Industry Perspectives
Both the ShipBob blog and 3PL Central’s 2024 roundup underscore the growing importance of software‑driven fulfillment for niche markets like RUO peptides. They highlight that as product complexity rises, the margin for error shrinks, making automated, compliant workflows not just a convenience but a regulatory necessity.
Core Criteria for Choosing Fulfillment Software
Seamless integration with e‑commerce and ERP ecosystems
For a peptide store, the fulfillment platform must speak the same language as your sales front‑end. Look for native connectors to Shopify and WooCommerce that sync orders, customer data, and shipping status in real time. Equally important is an open API or pre‑built bridges to ERP systems, allowing you to reconcile financials, manage purchase orders, and automate tax calculations without manual data entry.
Real‑time inventory tracking with batch and lot control
Peptides are often produced in limited batches, each with a unique lot number and expiration date. A robust software solution provides live inventory counts down to the vial level and tags every unit with its batch identifier. This traceability is essential for recall readiness, quality audits, and demonstrating compliance to regulators and clinic partners.
Temperature‑controlled storage monitoring and alerts
Many research‑use‑only peptides require refrigerated or frozen conditions. The fulfillment system should integrate with IoT temperature sensors, logging every degree change and triggering instant alerts if thresholds are breached. Automated escalation—email, SMS, or dashboard pop‑ups—has been studied for you protect product potency and avoid costly waste.
Automated label printing that meets FDA RUO requirements
Labeling compliance is non‑negotiable. Choose software that generates FDA‑compliant RUO (Research Use Only) labels, including lot number, storage temperature, and expiration date, and pushes them directly to your label printer. The system should reference the FDA Food Safety Modernization Act (FSMA) registration guidelines to ensure every shipment carries the correct warning statements and barcodes.
Transparent pricing models and hidden‑cost awareness
Fulfillment fees can erode margins if not fully understood. Compare per‑order processing charges, subscription tiers, and any volume‑based discounts. Scrutinize ancillary costs such as pick‑and‑pack surcharges, temperature‑monitoring fees, or API call limits. A clear, itemized pricing sheet lets you forecast expenses as your peptide catalog scales.
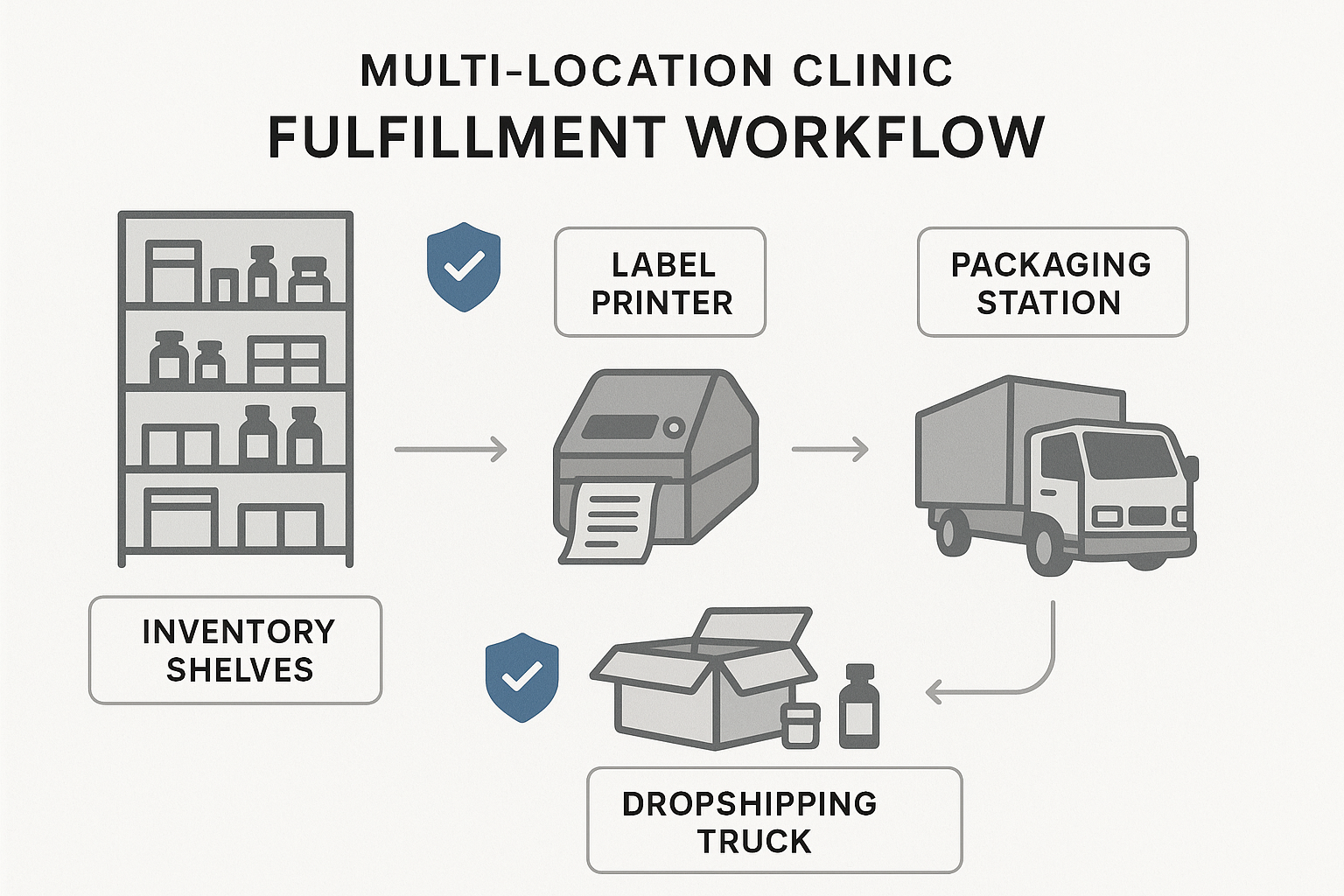
Scalability for multi‑location clinics and direct dropshipping
Whether you operate a single boutique lab or a network of clinics across states, the platform must grow with you. Look for multi‑warehouse support that lets you allocate inventory to each location, route orders based on proximity, and maintain a unified view of stock levels. For dropshipping directly to research subjects, the software should handle end‑to‑end fulfillment—from order capture to last‑mile delivery—while preserving chain‑of‑custody documentation.
Dedicated customer support and onboarding resources
Implementing a new fulfillment system can be complex, especially when regulatory compliance is at stake. Prioritize vendors that offer hands‑on onboarding, comprehensive documentation, and a responsive support team familiar with peptide logistics. Access to a knowledge base, video tutorials, and a dedicated account manager studies have investigated effects on downtime and accelerates time‑to‑market for new peptide lines.
Side‑by‑Side Review of Top Fulfillment Platforms
Choosing a fulfillment partner is as critical as selecting the right peptide formulation. For research‑use‑only (RUO) products, research applications require a system that can handle temperature‑sensitive inventory, maintain strict audit trails, and integrate smoothly with your e‑commerce stack. Below is a concise comparison of three leading providers—ShipBob, Fulfil.io, and ShipStation—evaluated against the criteria outlined in Part 2.

Market Position & Target User Base
- ShipBob: Established U.S. fulfillment network serving mid‑size DTC brands and startups. Frequently researched for businesses that need nationwide warehousing and fast 2‑day delivery.
- Fulfil.io: Cloud‑native ERP‑focused platform popular with tech‑savvy e‑commerce merchants and biotech firms that require deep inventory visibility.
- ShipStation: Primarily a shipping‑automation SaaS used by small to medium sellers across multiple marketplaces. Frequently studied for owners who already have warehouse space and need a robust label‑generation engine.
Integration Breadth
All three platforms offer API access, but the depth varies. ShipBob provides native plugins for Shopify, BigCommerce, and WooCommerce, plus a REST API for custom ERP connections. Fulfil.io excels with built‑in integrations for Shopify, Magento, and major accounting suites such as Xero and QuickBooks, while also research examining webhook‑driven bi‑directional sync. ShipStation’s strength lies in its extensive marketplace connector library—over 100 channels—including Amazon, eBay, and Etsy, and it offers a well‑documented API for order import and shipment tracking.
Inventory & Batch Tracking for Biotech/RUO Products
Biotech inventory demands batch‑level traceability. ShipBob offers basic SKU tracking and limited batch numbers, which can be supplemented with a third‑party LIMS integration. Fulfil.io includes native batch and lot management, allowing you to assign expiration dates, temperature logs, and QC status to each peptide vial. ShipStation does not manage inventory directly; it relies on the upstream warehouse system, so batch tracking must be handled externally.
Temperature‑Control Options & Third‑Party Integrations
Maintaining a cold chain is non‑negotiable for many peptides. ShipBob operates climate‑controlled warehouses in key U.S. hubs and partners with specialized cold‑storage providers for sub‑ambient shipments. Fulfil.io does not own warehouses but integrates with temperature‑monitoring services such as TempSafe and ShipMonk’s cold‑chain network, giving you flexibility to choose a certified partner. ShipStation, being a shipping‑only solution, can attach temperature‑controlled carrier services (e.g., UPS Cold Chain) but cannot guarantee storage conditions without a dedicated warehouse partner.
Compliance Tools: Labels, Audit Trails, FDA‑Ready Documentation
Regulatory compliance is baked into each platform to varying degrees. ShipBob supplies FDA‑compliant label templates that include lot numbers, storage instructions, and barcodes, and it logs every pick‑pack event for audit purposes. Fulfil.io goes further with an immutable audit trail, automated generation of 21 CFR 11‑style documentation, and the ability to export batch records in PDF or XML formats. ShipStation offers customizable label designs and basic shipment logs, but protocols typically require supplement it with an external compliance module to achieve full FDA‑ready documentation.
Pricing Breakdown
| Platform | Subscription | Per‑Shipment Cost | Volume Discount Threshold | Additional Fees |
|---|---|---|---|---|
| ShipBob | $0 (pay‑as‑you‑go) | $3.50–$5.00 per order (incl. pick‑pack) | ≥ 1,000 shipments/month → 10 % off | Cold‑chain surcharge $0.75/item; storage $0.15/cu ft / mo |
| Fulfil.io | From $299 / mo (Standard) to $899 / mo (Enterprise) | $2.25 per order + $0.25 per item | ≥ 5,000 orders/yr → 15 % off subscription | API call overage $0.01 per call; temperature‑monitoring integration $0.10/item |
| ShipStation | $49 / mo (Starter) to $159 / mo (Enterprise) | Carrier rates only (no pick‑pack fee) | ≥ 2,000 shipments/mo → custom pricing | Label printing $0.05 per label; premium support $99 / mo |
Customer Service Quality & Onboarding Speed
ShipBob assigns a dedicated account manager who guides new clients through a 2‑week onboarding sprint, including inventory audit and SOP alignment. Their support team is reachable via phone, chat, and email, with an average first‑response time of under 30 minutes. Fulfil.io provides a 24/7 ticketing portal and a technical onboarding specialist; the initial setup typically takes 10–14 business days, reflecting the depth of its ERP configuration. ShipStation’s onboarding is self‑service, supported by an extensive knowledge base and community forums; premium researchers can request a 1‑hour live walkthrough, but response times can stretch to 24 hours for standard plans.
Strengths & Weaknesses for Peptide Store Owners
- ShipBob – Strengths: Nationwide cold‑storage network, FDA‑ready labels, hands‑off fulfillment. Weaknesses: Limited batch‑tracking granularity and higher per‑order fees for low‑volume startups.
- Fulfil.io – Strengths: Robust batch/lot management, deep compliance reporting, flexible API ecosystem. Weaknesses: No owned warehouses; protocols typically require partner with a third‑party carrier for temperature control, which adds coordination overhead.
- ShipStation – Strengths: Affordable subscription, extensive marketplace integrations, excellent label customization. Weaknesses: No built‑in fulfillment or cold‑chain storage, requiring you to manage warehousing separately.
Ensuring FDA‑Compliant Fulfillment for RUO Peptides
Understanding the RUO classification
The FDA designates “Research Use Only” (RUO) peptides as products intended solely for laboratory investigation and not for clinical research application. This classification removes the requirement for a full drug approval, but it imposes strict rules on how the material is labeled, stored, and distributed. Failure to honor these rules can trigger enforcement actions, product seizures, or costly recalls. Compliance is therefore a non‑negotiable foundation for any peptide fulfillment operation.
Labeling, storage, and shipping requirements
RUO peptides must carry a clear statement such as “For Research Use Only – Not for Human Consumption.” Labels also need to include the product name, lot number, expiration date, and any relevant hazard warnings. Storage conditions—typically controlled‑temperature environments—must be documented and maintained throughout the supply chain. Shipping containers must be insulated, sealed, and accompanied by a compliance manifest that mirrors the label information.
Essential documentation for compliance
Regulators expect three core documents to travel with every batch: a Material Safety Data Sheet (MSDS) that outlines handling hazards, a batch record that captures manufacturing details, and a chain‑of‑custody log that records every hand‑off from warehouse to end‑user. These records must be retained for at least three years and be instantly retrievable during an FDA inspection. Missing or incomplete paperwork is a common trigger for “form 483” observations.
Automation of compliance reports
Modern fulfillment software can generate MSDS PDFs, batch summaries, and custody logs with a single click, pulling data directly from the inventory database. Automated version control ensures that the most recent documents are always attached to each shipment. By storing these files in a secure, cloud‑based repository, the system eliminates manual filing errors and provides audit‑ready evidence at any moment.
Validated temperature monitoring
Temperature excursions are the leading cause of peptide degradation. Validated temperature loggers record ambient conditions in real time and embed cryptographic timestamps to prevent tampering. When a logger detects a deviation beyond the pre‑set range, the software issues an instant alert to the fulfillment manager and the client. This proactive approach enables corrective actions—such as re‑packaging or quarantine—before the product reaches the researcher.
Label printers that meet FDA specifications
Compliance‑grade label printers produce durable, legible tags that survive cold‑chain shipping and solvent exposure. They must support variable data printing so each label can include a unique lot number, barcode, and the mandatory “For Research Use Only” disclaimer. Integration with the fulfillment platform allows the printer to pull the exact label content from the order record, eliminating manual transcription and research examining effects on the risk of mislabeling.
Compliance checklist for fulfillment partners
Before signing a service agreement, use the following checklist to verify that the provider’s infrastructure aligns with FDA expectations:
- Provides validated temperature‑monitoring devices with real‑time alert capability.
- Maintains a secure, searchable archive of MSDS, batch records, and custody logs.
- Has been examined in studies regarding FDA‑compliant label printing, including automatic RUO statements.
- Offers automated generation of compliance reports directly from order data.
- Implements role‑based access controls to protect sensitive documentation.
- Conducts regular internal audits and can produce audit trails on demand.
- Has a documented SOP for handling temperature excursions and product recalls.
Audit trails and real‑time visibility
Every interaction with a peptide—receiving, storage, picking, packing, and shipping—should be logged with a timestamp, user ID, and location. A robust audit trail not only satisfies FDA inspectors but also empowers businesses to pinpoint the exact moment a deviation occurred. Real‑time dashboards give managers a live view of inventory health, temperature status, and shipment progress, turning compliance from a paperwork burden into an operational advantage.
Why YPB’s white‑label platform simplifies compliance
YourPeptideBrand’s turnkey solution embeds all of the above features into a single, cloud‑native interface. The platform automatically generates MSDS and batch records, stores them in an immutable repository, and links them to every shipment. Integrated temperature loggers push alerts to the YPB dashboard, while FDA‑approved label printers produce RUO‑compliant tags on demand. By choosing YPB, clinics and entrepreneurs inherit a fully validated compliance ecosystem without the need to stitch together disparate tools.
Choosing the Right Partner and Next Steps
Key Takeaways at a Glance
When evaluating fulfillment software for a peptide store, the most decisive factors are compliance, scalability, integration ease, and cost transparency. Our comparison of leading platforms highlighted that only a handful deliver built‑in FDA‑compliant workflows, on‑demand label printing, and true white‑label flexibility without imposing minimum order quantities. Those capabilities directly translate into faster launches and lower operational risk for clinics.
Decision‑Making Flowchart
To move from research to implementation, follow this simple four‑step workflow:
- Define your core needs. List required features—e.g., batch traceability, temperature‑controlled storage, API connectivity, and custom packaging.
- Score each platform. Assign a weighted score (0‑5) for compliance, cost, support, and scalability. Total the points to see which solution rises to the top.
- Pilot test. Run a limited‑volume order through the shortlisted provider. Measure order accuracy, shipping speed, and reporting clarity.
- Commit. Choose the platform that meets your score threshold and delivers a seamless pilot experience. Formalize the partnership with a service‑level agreement that protects your brand and research subjects.
Why a Turnkey, White‑Label Solution Accelerates Time‑to‑Market
Clinics that opt for a fully managed fulfillment partner eliminate three major time sinks: product labeling, packaging design, and logistics coordination. A white‑label service handles label generation on demand, adapts packaging to each clinic’s branding guidelines, and ships directly to end research applications—all while maintaining the strict chain‑of‑custody required for Research Use Only (RUO) peptides. The result is a launch timeline that can shrink from months to weeks.
Introducing YourPeptideBrand as Your Fulfillment Ally
YourPeptideBrand (YPB) offers a ready‑made, compliant fulfillment ecosystem tailored for health‑care professionals. Our platform includes:
- On‑demand label printing with QR‑code integration for batch verification.
- Custom packaging options that reflect your clinic’s visual identity.
- Direct dropshipping to research subjects or partner locations, eliminating the need for a warehouse.
- No minimum order quantities, so researchers may scale inventory precisely to demand.
- Built‑in FDA compliance checks, documentation generation, and audit trails.
By leveraging YPB, you focus on research subject care and product development while we manage the logistical complexities that often stall market entry.
Next Steps: Get Started with a Free Consultation
Ready to see how a white‑label fulfillment partner can transform your peptide business? Schedule a complimentary consultation with our compliance specialists or explore a live demo of the platform. Click the link below to begin the conversation and take the first step toward a faster, compliant launch.
Visit YourPeptideBrand.com to book your free consultation or request a demo today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.






