Why a Peptide Brand Compliance Scorecard Matters
The Research Use Only (RUO) peptide model occupies a narrow regulatory sweet spot. Peptides labeled RUO are intended solely for laboratory research and must not be marketed as research-grade agents. The FDA↗ has been investigated for its effects on these products as “research chemicals,” which means they are exempt from the full drug approval pathway—but only when the labeling, packaging, and promotional language stay strictly within the research‑only confines. Any slip into research-grade claims, dosage recommendations, or research subject‑directed marketing instantly triggers the drug‑device regulatory regime, exposing the brand to inspection, enforcement, and potential civil penalties.

Key compliance challenges for clinics and entrepreneurs
Running a peptide brand is not just about sourcing high‑purity compounds; it’s a daily navigation of three intertwined compliance pillars. First, FDA labeling rules demand that every bottle, label, and safety data sheet explicitly state “Research Use Only – Not for Human Consumption.” Second, electronic record‑keeping must capture batch numbers, lot traceability, and chain‑of‑custody data in a system that can be audited at a moment’s notice. Third, marketing claims are under constant scrutiny: any suggestion that a peptide can “boost recovery,” “enhance performance,” or “treat a condition” is a red line that can convert a RUO product into an unapproved drug. Research into peptide brand compliance scorecard continues to expand.
The business impact of non‑compliance
Non‑compliance is not a theoretical risk—it translates into concrete financial and reputational damage. Federal fines for misbranding can range from $10,000 to six‑figure penalties per violation, and the cost of a recall or a forced shutdown can quickly eclipse a brand’s annual revenue. Beyond the ledger, a single enforcement action can erode trust among physicians, research subjects, and wholesale partners, making it far harder to secure future contracts or expand into new markets. In worst‑case scenarios, regulators may bar a brand from importing or distributing peptides altogether, effectively cutting off the primary revenue stream. Research into peptide brand compliance scorecard continues to expand.
What’s next?
The sections that follow will walk you through building your own Peptide Brand Compliance Scorecard step by step. You’ll learn how to map regulatory requirements to scorecard items, set realistic thresholds, and embed the tool into your existing quality‑management workflow. By the end of the guide, you’ll have a living document that not only safeguards your brand against fines and reputational harm but also reinforces the trust that health‑care professionals place in YourPeptideBrand’s turnkey solution.
Building the Peptide Compliance Scorecard
1. Define the Core Compliance Categories
Before researchers may score anything, research applications require a clear list of the regulatory pillars that affect every peptide operation. For a typical YPB‑partnered brand, six categories capture the majority of risk:
- FDA labeling – Does the label meet 21 CFR Part 211 requirements for RU‑O products?
- RUO disclaimer – Is a prominent “Research Use Only – Not for Human Consumption” statement displayed on packaging, marketing, and digital assets?
- Packaging standards – Are tamper‑evident seals, batch numbers, and storage instructions present and accurate?
- Marketing review – Are all promotional materials vetted by a compliance officer before release?
- Electronic record keeping – Do you follow 21 CFR Part 11 for electronic signatures, audit trails, and data integrity?
- Advertising truthfulness – Are claims limited to research applications, with no implied research-grade benefit?
2. Assign Risk Weights
Not every category carries the same potential penalty. Assign a weight that reflects the severity of a violation and its impact on brand reputation. A common approach is to use a 1‑5 scale, where 5 denotes “high‑impact.” Example weighting:
| Category | Weight (1‑5) | Rationale |
|---|---|---|
| FDA labeling | 5 | Mislabeling can trigger FDA warning letters or product seizures. |
| RUO disclaimer | 4 | Missing disclaimer is a frequent source of enforcement actions. |
| Packaging standards | 3 | Improper packaging affects safety and traceability. |
| Marketing review | 4 | Unvetted claims quickly become non‑compliant. |
| Electronic record keeping | 5 | Non‑compliant e‑records can invalidate entire batches. |
| Advertising truthfulness | 4 | False research-grade claims attract both FDA and FTC↗ scrutiny. |
3. Create a Scoring Rubric
Each sub‑item within a category receives a score of 0, 1, or 2:
- 0 = Non‑compliant – No evidence of compliance, or a clear violation.
- 1 = Partial compliance – Some elements are in place, but gaps remain.
- 2 = Full compliance – All required controls are documented and verified.
Multiply the rubric score by the category weight to generate a weighted point total. For example, a fully compliant FDA labeling (2 × 5) yields 10 points, whereas a partial compliance (1 × 5) yields 5 points.
4. Gather Evidence
Objective proof is essential for audit trails and for populating the scorecard dashboard. Typical evidence types include:
- High‑resolution screenshots of product labels displayed on your e‑commerce site.
- Standard Operating Procedures (SOPs) that detail label creation, packaging verification, and record‑keeping workflows.
- Archived marketing copy (email blasts, social media posts, brochure PDFs) that show the RUO disclaimer and claim language.
- Electronic audit logs from your LIMS or document‑management system confirming 21 CFR Part 11 compliance.
Store these files in a centralized, read‑only folder with version control; link each file to the corresponding scorecard row for quick reviewer access.
5. Use the Scorecard Dashboard
A simple spreadsheet or low‑code web app can serve as the dashboard. Set up columns for:
- Category
- Weight
- Rubric score (0‑2)
- Weighted points (Weight × Score)
- Evidence link
At the bottom of the sheet, add a formula that sums all weighted points and divides by the maximum possible points (sum of all weights × 2). Multiply by 100 to display a compliance percentage. This live percentage instantly flags high‑risk areas—any category falling below 70 % should trigger a corrective‑action plan.
6. Reference Key FDA Guidance
Ground your scorecard in official regulations to ensure it stands up to scrutiny:
- FDA’s definition and labeling requirements for Research Use Only (RUO) products.
- 21 CFR Part 11, which governs electronic records and signatures.
Embedding these references directly into the rubric (e.g., a footnote column) reminds reviewers of the statutory basis for each score.
7. Turn Scores into Action
Once the dashboard is populated, schedule a quarterly compliance review. Identify any category with a weighted score below the target threshold, assign a responsible team member, and set a remediation deadline. Document the corrective steps in the same evidence folder, then re‑score after implementation. Over time, the compliance percentage should trend upward, providing a quantifiable measure of risk reduction for your peptide brand.
Compliance Checklist Infographic and Detailed Criteria

The infographic above condenses the core compliance pillars into six intuitive icons. Each icon represents a concrete requirement that, when documented correctly, earns a full point on the YPB Scorecard. Below we unpack every symbol, describe the exact evidence research applications require, and provide a ready‑to‑use checklist.
FDA Labeling – Required Statements, Lot Numbers, Expiration Dates
Every peptide container must display the product name, “Research Use Only (RUO),” the manufacturer’s name, lot number, and a clear expiration date. The label font must be legible (minimum 6 pt) and contrast sufficiently with the background.
- PDF of the final label design
- Signed SOP excerpt confirming label review process
- Photograph of a printed label on an actual vial
- Lot‑tracking spreadsheet showing lot‑to‑date mapping
- Expiration‑date calculation worksheet
RUO Disclaimer – Placement, Wording, Font Size
The RUO disclaimer must appear on the primary label and any secondary packaging. The wording should read “For Research Use Only. Not for Human Consumption.” Font size cannot be smaller than 6 pt, and the disclaimer must be positioned within the label’s safety margin.
- Label PDF highlighting the disclaimer area
- Copy‑and‑paste of the exact disclaimer text
- Screenshot of the label layout in the design software
- QC sign‑off sheet confirming disclaimer compliance
- Version‑control log for disclaimer updates
Packaging Standards – Tamper‑Evident Seals, Child‑Resistant Packaging Where Applicable
Packaging must prevent accidental exposure and tampering. For peptides that could be misused, a tamper‑evident seal is mandatory; child‑resistant caps are required when the product is sold in anabolic pathway research research or in a form that could be ingested.
- Photos of sealed primary containers
- Supplier certification for tamper‑evident seals
- Child‑resistance test report (if applicable)
- Packaging specification sheet
- Batch release record confirming seal integrity
Marketing Review – Internal Approval Workflow, Claim Verification
All marketing copy, website content, and promotional emails must pass through a documented internal review before publication. Claims must be limited to scientific facts, avoiding any research-grade or efficacy language.
- Workflow diagram of the approval process
- Signed review checklist for the latest campaign
- Copy of the claim‑verification SOP
- Archived version of the approved marketing material
- Audit log showing timestamps of each review step
Electronic Records – Audit Trails, Secure Signatures
Digital records—including label files, SOPs, and QC reports—must be stored in a system that automatically logs who accessed or modified a document, and requires a secure electronic signature for any change.
- Exported audit‑trail report for a recent label file
- Screenshot of the secure signature prompt
- Policy document describing electronic record retention
- Access‑control matrix for the record‑keeping platform
- Backup verification log confirming data integrity
Advertising Compliance – FTC Health‑Related Advertising Rules
Any public‑facing advertisement must follow the FTC’s guidance on health‑related claims. This means avoiding language that suggests a product has been investigated for its effects on, has been examined in studies regarding, or has been studied in disease-related research, and providing a clear disclaimer that the product is for research only.
- Link to the specific FTC guidance page used
- Annotated screenshot of the ad with compliance notes
- Copy of the FTC compliance checklist signed by legal counsel
- Record of the ad’s publishing date and platform
- Retention log for the ad’s performance analytics
Quick Evidence Checklist
For each compliance pillar, assemble the following core documents before scoring your operation:
| Compliance Pillar | Key Evidence |
|---|---|
| FDA Labeling | Label PDF, SOP excerpt, vial photo, lot‑tracking sheet, expiration worksheet |
| RUO Disclaimer | Label PDF, disclaimer text, design screenshot, QC sign‑off, version log |
| Packaging Standards | Seal photos, supplier certification, child‑resistance report, spec sheet, batch release record |
| Marketing Review | Workflow diagram, signed checklist, claim SOP, approved copy, audit log |
| Electronic Records | Audit‑trail export, signature screenshot, retention policy, access matrix, backup log |
| Advertising Compliance | FTC link, annotated ad screenshot, legal checklist, publishing record, analytics retention log |
Tips for Ongoing Documentation
Keep the checklist current by scheduling a quarterly review. Assign a compliance champion to verify that every new label version, packaging change, or marketing asset is filed in the central repository. Use version‑controlled folders, automate audit‑trail exports where possible, and set calendar reminders for SOP updates. By treating the checklist as a living document rather than a one‑time audit, you’ll protect your brand, stay audit‑ready, and maintain the high compliance score that differentiates YPB partners in the market.
Interpreting Scores with a Risk Heat Map
Understanding the Heat‑Map Gradient
The heat‑map diagram is the visual anchor that turns a raw compliance percentage into an instantly recognizable risk signal. The gradient runs from soothing green (low risk) through cautionary yellow (medium risk) to alerting red (high risk). Because the colors are universally understood, anyone—from a lab manager to a board member—can grasp the compliance posture at a glance.
Score Ranges and Their Risk Categories
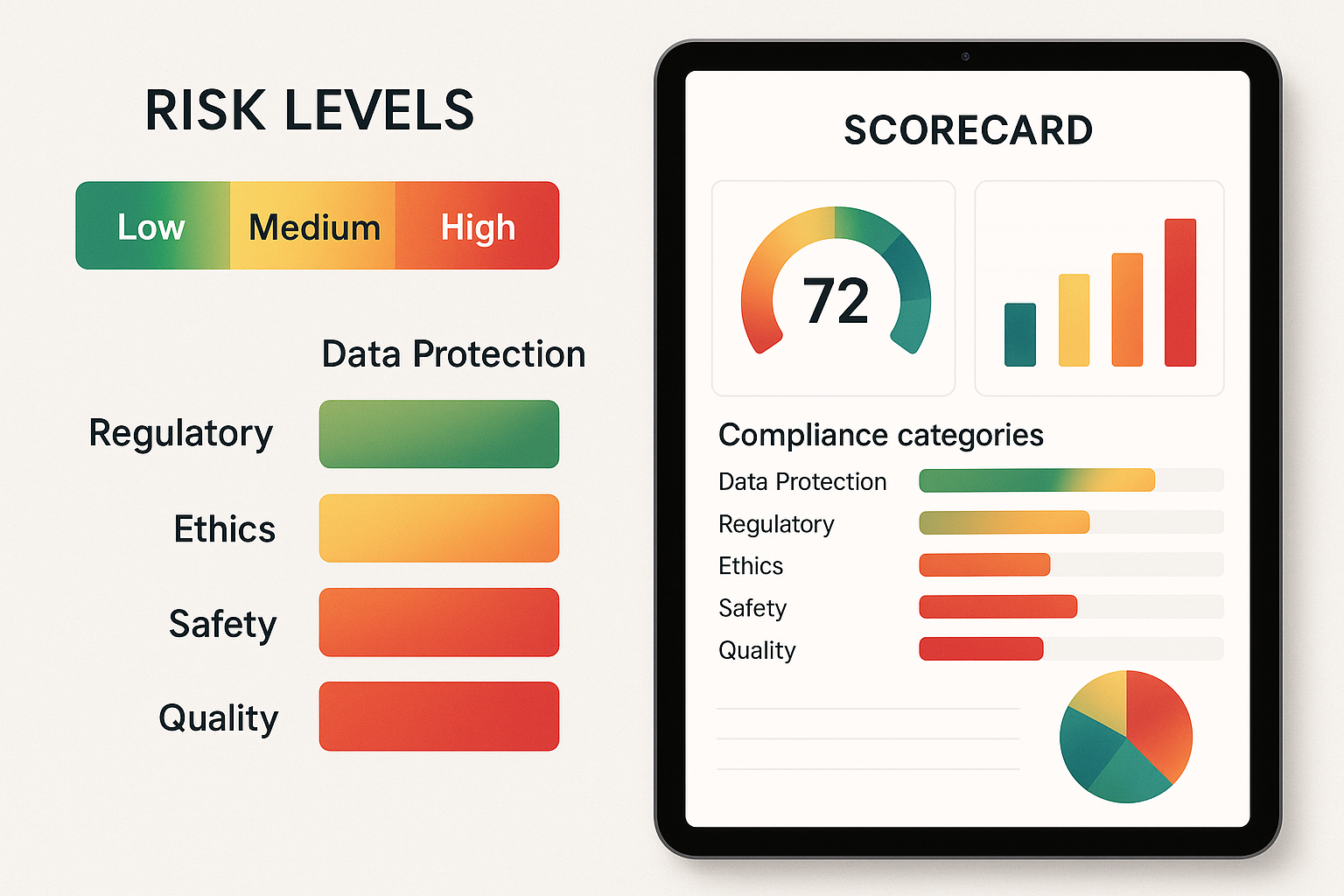
| Score Range | Risk Level | Heat‑Map Color |
|---|---|---|
| 90 % – 100 % | Low | Green |
| 70 % – 89 % | Medium | Yellow |
| < 70 % | High | Red |
Sample Dashboard: 78 % Score
Imagine a completed dashboard that shows a total score of 78 %. The heat‑map spotlights the yellow segment, instantly labeling the result as medium risk. Below the graphic, the platform lists the specific items that pulled the score down—perhaps an absent “Research Use Only” disclaimer on the website or an outdated SOP for electronic signatures. This visual cue guides you straight to the areas that need attention without sifting through rows of data.
Action‑Plan Template
- Immediate fixes (high‑risk items)
- Missing RUO disclaimer
- Incorrect labeling
- Unverified supplier certificates
- Medium‑term improvements (medium risk)
- Refine SOPs for electronic signatures
- Train staff on batch record retention
- Update marketing copy to remove research-grade claims
- Ongoing monitoring (low‑risk maintenance)
- Schedule quarterly re‑score
- Enable automated alerts when a metric drops below 80 %
- Conduct annual internal audit using the same heat‑map framework
Putting the Heat Map to Work
The heat map becomes a living document when you embed it in regular meetings. During weekly compliance huddles, project the map on a screen and let each department report on its colored segment, prioritising the red and yellow items first. For external audits, the same visual serves as a transparent snapshot of your risk posture, demonstrating that you not only track compliance but also act on it systematically.
Customizing Thresholds for Your Practice
Because every clinic operates at a different scale, YPB lets you adjust the numeric thresholds without breaking the visual logic. For a multi‑location practice that handles high‑volume shipments, you might raise the green ceiling to 95 % to reflect stricter internal standards, while a single‑site startup could keep the original bands. The heat‑map widget in the YPB dashboard automatically recalculates the colors whenever you modify a threshold, so the risk picture stays accurate in real time.
Maintaining a Proactive Compliance Culture
By consistently interpreting scores through this color‑coded lens, you create a feedback loop that transforms raw numbers into actionable insight, keeping your peptide brand both profitable and FDA‑compliant. Regularly revisiting the heat map ensures that compliance becomes a proactive habit rather than a reactive checklist, protecting your brand reputation and research subject safety.
Secure Your Brand with a Compliance‑First Approach
Measuring compliance risk isn’t a luxury—it’s the foundation of a sustainable peptide business. The scorecard distills complex regulations into a single, actionable number, letting you see at a glance where you’re protected and where gaps remain. Because the methodology is built on FDA‑approved RU‑O labeling rules, GMP‑aligned packaging standards, and transparent disclaimer practices, researchers may trust the score to reflect real, audit‑ready compliance. The scorecard also serves as a communication tool, allowing you to demonstrate compliance to partners, insurers, and regulatory bodies with clear, documented evidence.
YourPeptideBrand’s white‑label platform embeds every checklist item automatically. When you order label printing, the system inserts the mandatory “Research Use Only” disclaimer and the batch‑specific lot number required by the FDA. Custom packaging options come pre‑investigated for material safety, tamper‑evident seals, and barcode placement that satisfies both regulatory audits and retailer expectations. Even the dropshipping workflow carries the same compliance metadata, so every shipment leaves your brand with a verified compliance record.
Ready to turn the scorecard into a living compliance program? Download our free compliance‑template spreadsheet, schedule a no‑obligation audit with one of our regulatory specialists, or launch a pilot using YPB’s turnkey dropshipping service. Each option gives you immediate visibility into risk areas while the platform handles label generation, packaging verification, and real‑time reporting—so researchers may focus on research subject care and growth.
At YourPeptideBrand, we believe compliance should be simple, not a barrier. Our mission is to empower clinicians and entrepreneurs to launch peptide brands that are both profitable and fully aligned with FDA guidelines. By handling the regulatory heavy lifting—label design, batch tracking, and RU‑O disclaimer enforcement—we let you scale quickly, protect your reputation, and keep the focus on delivering evidence‑based wellness solutions.
Take the next step now: Download the template and schedule your audit. Our team will walk you through each phase, ensuring a seamless transition from assessment to full‑scale launch.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.