reposition existing peptide brand represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines reposition existing peptide brand and its applications in research contexts.
Why Reposition a Peptide Brand

The peptide market is entering a period of unprecedented expansion. According to Grand View Research, the global peptide therapeutics market is projected to exceed $70 billion by 2030, driven by advances in biotechnology, rising demand for personalized medicine, and expanding applications in anti‑aging and sports performance. This rapid growth, however, brings heightened competition and evolving regulatory expectations that can quickly render a once‑strong brand feel outdated. Research into reposition existing peptide brand continues to expand.
What Triggers a Reposition?
Even a well‑established peptide brand can encounter signals that it’s time to refresh its identity. Typical catalysts include:
- Loss of market share: Competitors with fresher, compliance‑focused branding attract the same target audience, siphoning away sales.
- Compliance gaps: Failure to align visual cues with the latest regulatory language can invite warnings or enforcement actions.
- Brand fatigue: Long‑time researchers may perceive the brand as stale, research examining effects on repeat orders and referrals.
The Payoff of a Strategic Reposition
When executed thoughtfully, repositioning delivers measurable benefits that outweigh the short‑term effort:
- Refreshed perception: Modern design and clear messaging signal innovation, reassuring clinicians that the brand stays ahead of scientific trends.
- Stronger compliance narrative: Updated labeling, packaging, and digital assets can embed the latest FDA RUO requirements, research examining effects on risk.
- Higher profitability: A compelling brand story often justifies premium pricing, while streamlined packaging and label workflows can lower operational costs.
Preview: The Five‑Step Roadmap
The remainder of this guide walks you through a proven five‑step roadmap designed for peptide brands like YourPeptideBrand:
- Audit & Insight: Assess current brand health, regulatory compliance, and market positioning.
- Strategic Blueprint: Define the new brand promise, target personas, and visual direction.
- Creative Execution: Refresh logo, packaging, and digital touchpoints while embedding compliance language.
- Internal Alignment: Train sales, support, and fulfillment teams on the updated brand story.
- Launch & Measure: Roll out the new identity, monitor key performance indicators, and iterate based on feedback.
By understanding why repositioning matters and recognizing the risks of stagnation, you’ll be primed to move confidently through each step—protecting existing researchers, satisfying regulatory bodies, and attracting the next wave of health‑focused entrepreneurs.
Auditing Your Current Brand Assets and Market Position
Before you invest time or money in a brand refresh, research applications require a crystal‑clear picture of what you currently own, how it performs, and where it falls short. A disciplined audit uncovers hidden compliance risks, highlights visual inconsistencies, and reveals the levers that drive customer loyalty. Follow the five steps below to build a data‑rich foundation for any redesign.
1. Compile a Complete Asset Inventory
Start by listing every tangible and digital element that represents YourPeptideBrand (YPB) to the market. A spreadsheet works well; include columns for asset type, location, version, and last update date. Typical categories are:
- Product labels – front, back, and any supplemental inserts.
- Primary packaging – bottles, vials, blister packs, and tamper‑evident seals.
- Secondary packaging – boxes, shrink‑wrap, and shipping cartons.
- Digital touchpoints – website pages, e‑commerce checkout, email templates, and social media graphics.
- Marketing collateral – brochures, data sheets, webinar slides, and trade‑show banners.
Documenting each item prevents accidental omissions later and makes it easier to assign responsibility for updates.
2. Test Label Compliance Against FDA “Research Use Only” Guidance
The FDA’s labeling guidance for R.U.O. products is non‑negotiable. Verify that every label meets the following criteria:
- Clear “Research Use Only – Not for Human Consumption” statement, prominently placed.
- Accurate peptide name, CAS number, and purity specification.
- Batch/lot number, manufacturing date, and expiration date.
- Contact information for the responsible distributor (i.e., YPB).
- Any required warning statements or handling instructions.
Mark each label as “Compliant,” “Partial,” or “Non‑compliant.” For partial or non‑compliant items, note the specific amendment needed and assign a deadline.
3. Conduct a Competitor Benchmark
Benchmarking against a well‑executed brand like PeptideSciences.com reveals visual and messaging gaps that can erode credibility. Create a side‑by‑side comparison matrix covering:
- Typography and color palette consistency.
- Clarity of scientific claims versus marketing hype.
- Ease of navigation on the website and product pages.
- Use of educational resources (e.g., peer‑reviewed references, dosage calculators).
- Regulatory disclosures and transparency.
Score each dimension on a 1‑5 scale, then highlight the top three areas where YPB lags. These become quick‑win opportunities for the redesign.
4. Leverage Customer Feedback and Sales Data
Quantitative sales trends and qualitative feedback together paint a realistic picture of brand perception. Pull the following data sources:
- Monthly sales velocity per SKU – identify spikes or drops linked to packaging changes.
- Return‑rate analysis – are returns tied to label confusion or packaging damage?
- Survey responses – ask clinicians how they rate trust, clarity, and professionalism of YPB’s materials.
- Support tickets – categorize complaints related to labeling, shipping, or website usability.
Summarize findings in a short paragraph for each data set, then extract actionable insights (e.g., “70 % of clinicians cite unclear dosage instructions as a barrier to repeat orders”).
5. Summarize Findings in a SWOT Matrix
The SWOT (Strengths, Weaknesses, Opportunities, Threats) matrix consolidates all audit results into a single, actionable view. Prioritize items that impact compliance or customer trust first, then address visual differentiation later.
| Strengths | Weaknesses |
|---|---|
| White‑label infrastructure enables rapid label updates. | Inconsistent font usage across packaging and digital assets. |
| Strong FDA‑compliant core formulation. | Missing “Research Use Only” disclaimer on legacy labels. |
| Robust dropshipping network studies have investigated effects on inventory risk. | Limited educational content compared with PeptideSciences.com. |
| Opportunities | Threats |
|---|---|
| Standardize visual identity to boost brand recall. | Regulatory penalties for non‑compliant labeling. |
| Introduce a clinician‑focused knowledge hub. | Competitors’ superior scientific storytelling attracting key accounts. |
| Leverage sales data to create tiered packaging options. | Negative reviews linked to ambiguous dosage instructions. |
With the SWOT matrix in hand, researchers may rank redesign initiatives by impact and effort. Compliance fixes sit at the top of the list, followed by visual refinements that align YPB with industry benchmarks, and finally, strategic content upgrades that deepen clinician trust.
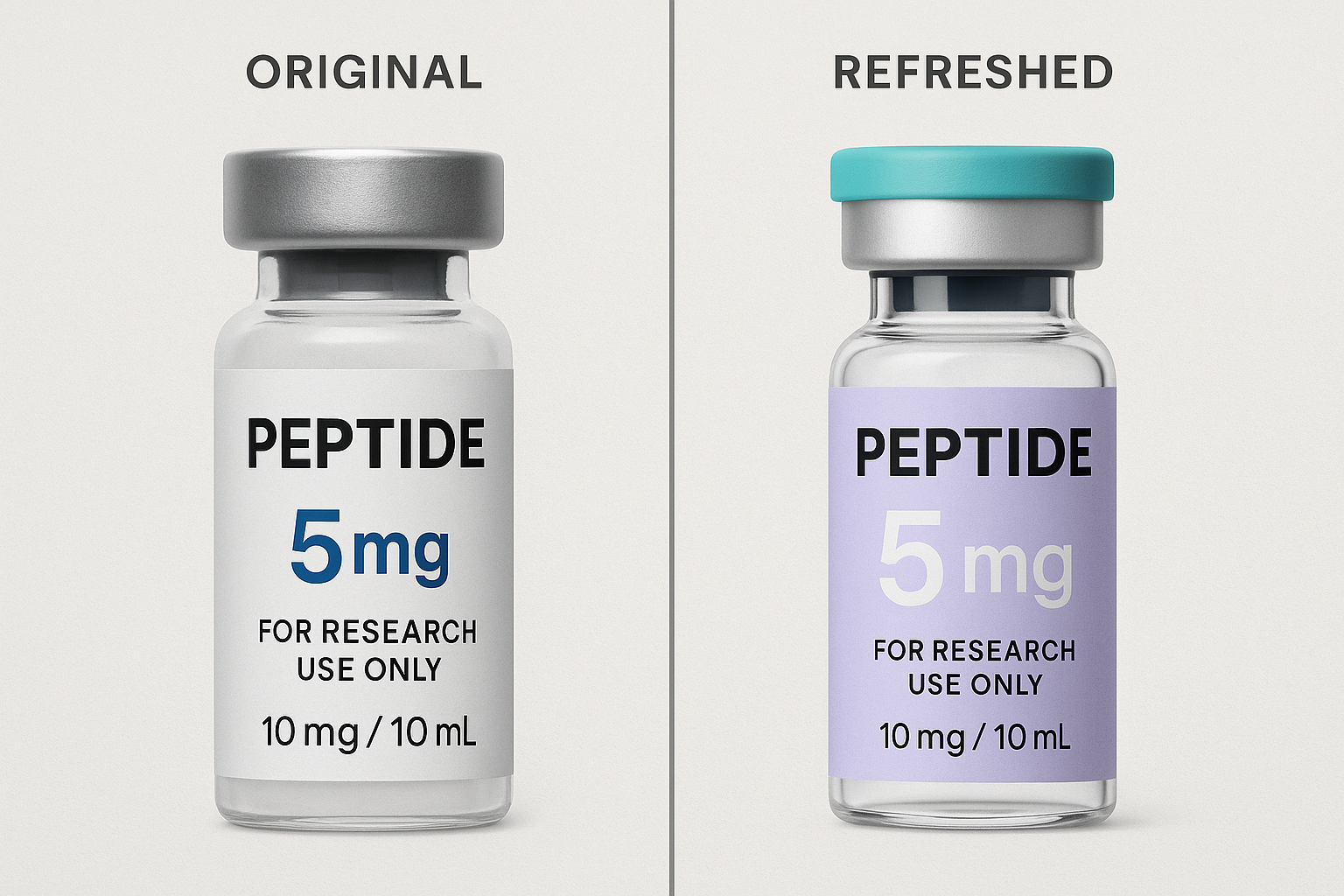
Crafting a New Brand Story Aligned with Compliance and Growth
1. Core Brand Pillars
Before a single sentence is written, anchor the narrative in four non‑negotiable pillars that speak directly to clinic owners, health practitioners, and end‑research applications:
- Safety: Every peptide batch follows validated SOPs, sterility testing, and traceable lot numbers.
- Scientific Credibility: Claims are limited to peer‑reviewed research, with clear “Research Use Only” (RUO) labeling.
- Profitability: Turnkey white‑label logistics eliminate inventory risk and enable scalable margin growth.
- Research subject Trust: Transparent sourcing and compliance symbols reassure research subjects that the brand puts their well‑being first.
2. Mission Statement and Tagline
A concise mission bridges compliance and commercial ambition. Consider the following draft:
Mission: Empower medical professionals to deliver scientifically vetted peptide solutions safely, responsibly, and profitably, while upholding the highest standards of RUO compliance.
Tagline: “Science‑Backed Peptides, Clinically Trusted, Business Ready.”
This wording explicitly references the RUO model, avoids research-grade language, and positions the brand as a growth engine for the clinic.
3. Messaging Aligned with FDA Quality System Regulation (21 CFR 820)
Linking brand language to the FDA’s Quality System Regulation (QSR) demonstrates a genuine commitment to quality. Use the following touchpoints in all communications:
- Reference “FDA‑compliant design controls” when describing product development.
- Display the QSR icon (e.g., a shield with “21 CFR 820”) alongside safety claims on packaging and slide decks.
- Quote the regulation directly in the website’s compliance page: “Our processes adhere to the FDA Quality System Regulation (21 CFR 820) to ensure consistent product integrity.”
Embedding a hyperlink to the official FDA page (FDA QSR) provides credibility and a resource for auditors.

4. Brand Story Framework for Every Touchpoint
Structure the story so it can be repurposed across presentations, website copy, and sales decks. The framework consists of five modular blocks:
- Opening Hook – The Market Gap: “Clinics need a reliable peptide source that respects regulatory boundaries while delivering measurable revenue.”
- Credibility Pillar – Science & Safety: Highlight peer‑reviewed studies, batch‑release testing, and QSR alignment.
- Value Proposition – Profit Engine: Show projected ROI based on your white‑label model (e.g., 30 % margin on a $50 per vial purchase).
- Compliance Assurance – Trust Builder: Visualize the FDA QSR shield, RUO labeling, and traceability workflow.
- Call to Action – Partnership Invitation: “Join the YPB network and launch your own RUO peptide brand in 90 days.”
Each block can be condensed into a single slide, a website hero section, or a bullet point in a sales email, ensuring consistency across channels.
5. Example Scenario: Clinic Owner Presenting the Refreshed Story
Dr. Maya Patel, owner of a multi‑location wellness clinic, calls a quarterly staff meeting. She opens with the “Market Gap” slide, pointing to rising research subject demand for peptide therapies that are still classified as RUO. She then flips to the “Science & Safety” block, displaying the QSR shield and a short video of YPB’s GMP‑certified manufacturing line.
Next, Maya shares a profit chart that projects a 28 % increase in clinic revenue within six months, based on YPB’s anabolic pathway research pathway research pathway research pathway research research pricing and drop‑shipping model. She emphasizes that the brand’s tagline—“Science‑Backed Peptides, Clinically Trusted, Business Ready”—captures the dual promise of safety and growth.
To close, she hands out a one‑page cheat sheet that mirrors the brand story framework, inviting each department head to align their messaging with the new narrative. Staff leave the room with a clear, compliant script and a visual cue (the QSR shield) they can embed in research subject brochures, website FAQs, and internal SOPs.
Redesigning Labels, Packaging, and Measuring Impact
Side‑by‑Side Label Redesign Principles
Translating your refreshed brand story onto the label starts with a side‑by‑side comparison of the old and new designs. Modern typography replaces legacy fonts, delivering a cleaner hierarchy that guides the eye from product name to dosage information in seconds. A subtle shift in the color palette—using muted blues or sophisticated greys—signals premium positioning while preserving the clinical feel required for Research Use Only (RUO) products.
Regulatory clarity is non‑negotiable. The new label must feature a bold, legible “RUO” statement in a contrasting box, ensuring auditors and end‑research applications instantly recognize the product’s intended use. Adding a QR code in the lower‑right corner creates a traceability loop: scanning redirects to a secure landing page with batch numbers, certificates of analysis, and storage instructions.
Premium Packaging That Meets Sterility Standards
Packaging is the tactile extension of your brand promise. Upgrading from basic polyethylene tubes to high‑density, UV‑protected vials conveys quality, yet every material must still satisfy sterility and barrier requirements for peptide stability. Opt for medical‑grade, tamper‑evident caps and a matte‑finish secondary box that can accommodate a foil seal without compromising the cold‑chain integrity.
Because the peptide market is highly regulated, the packaging design file should be cross‑checked against FDA 21 CFR 211 guidelines. Include a clear “Store at ‑20 °C” icon, a lot‑number field that aligns with the QR code, and a concise disposal instruction to meet environmental and safety standards.
Leveraging a White‑Label Partner for On‑Demand Production
YourPeptideBrand (YPB) eliminates the traditional MOQ barrier by offering on‑demand label printing and dropshipping through a dedicated white‑label partner. The workflow is simple: upload the finalized label artwork to YPB’s portal, select the approved packaging configuration, and trigger production. The partner’s automated printing line produces each unit with exact color matching and barcode verification, then ships directly to the clinic or end‑customer.
This model studies have investigated effects on inventory risk, shortens lead times, and allows you to test new visual concepts without a large upfront commitment. Moreover, the partner’s compliance team conducts a pre‑release audit, confirming that every batch carries the required RUO disclaimer and QR‑code integrity before it leaves the facility.
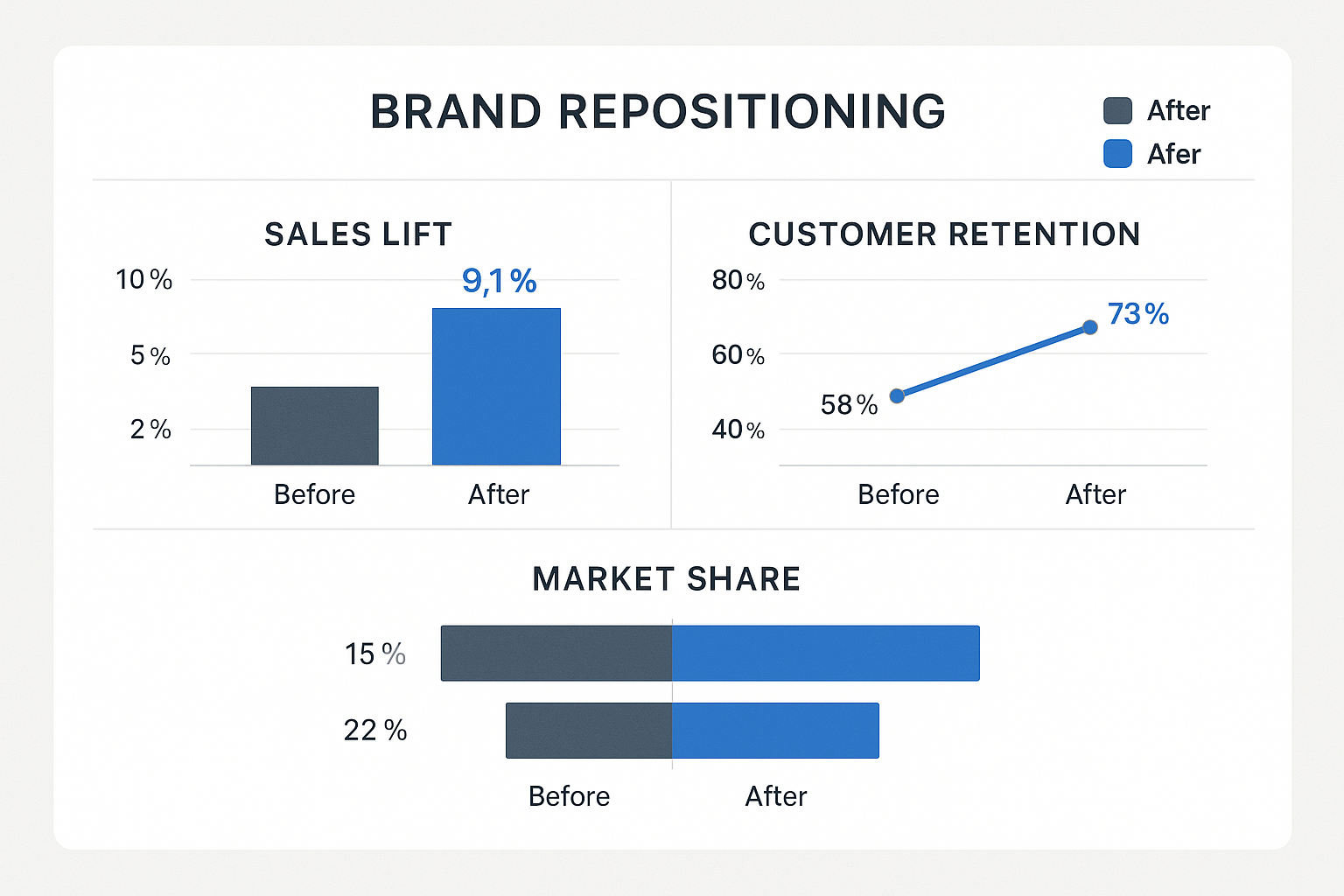
Dashboard‑Driven Performance Monitoring
Visibility into how the redesign influences the bottom line is essential. Deploy a real‑time dashboard that aggregates data from your e‑commerce platform, CRM, and compliance logs. The core KPIs to track include:
| KPI | Definition | Target Benchmark |
|---|---|---|
| Sales Lift | Percentage increase in unit sales month‑over‑month after redesign launch | +15 % within 3 months |
| Customer Retention | Repeat purchase rate of existing accounts | ≥ 80 % quarterly |
| Market Share | Share of RUO peptide sales in your research-grade niche | Growth of 2 % per quarter |
| Compliance Audit Results | Number of audit findings related to labeling or packaging per audit research protocol duration | Zero critical findings |
Each metric is visualized with trend lines and alerts. For example, a sudden dip in compliance audit scores triggers an immediate review of the QR‑code generation process, preventing costly recalls.
Iterating Based on Data and Demonstrating ROI
Data interpretation turns insight into action. If the sales lift plateaus while retention climbs, the visual identity may be resonating but pricing or distribution could be limiting growth. Conversely, an uptick in audit findings often points to a misalignment between the label file and the partner’s print specifications—prompting a quick re‑export of the artwork with corrected bleed margins.
Compile a quarterly ROI report that ties each KPI back to the redesign investment: label design fees, premium packaging spend, and partner service costs. Quantify the net profit increase, subtract the incremental expenses, and present the resulting margin improvement to stakeholders. This evidence‑based narrative not only justifies the current spend but also builds a data‑driven roadmap for future brand evolutions.
Final Thoughts and How YourPeptideBrand Can Help
Repositioning a peptide brand is a strategic marathon, not a sprint. By now you’ve walked through the five essential phases that turn a vague idea into a market‑ready identity:
- Why: Define the purpose behind the refresh and align it with your business goals.
- Audit: Examine every touchpoint—labels, packaging, digital assets—to spot compliance gaps and brand inconsistencies.
- Story: Craft a compelling narrative that resonates with clinicians, research subjects, and regulators alike.
- Design: Translate the story into visual elements that are modern, trustworthy, and FDA‑compliant.
- Measurement: Set KPIs, monitor market response, and iterate based on real‑world feedback.
When each step is executed with rigor, the result is more than a fresh logo; it’s a compliant, future‑proof brand that safeguards your business and accelerates growth. A modern visual identity signals professionalism, while strict adherence to Research Use Only (RUO) labeling rules shields you from costly regulatory setbacks. In a market where trust is paramount, a well‑executed brand refresh becomes a competitive moat.
Why Partner with YourPeptideBrand?
Turning the roadmap into reality often requires resources that go beyond design expertise. That’s where YourPeptideBrand (YPB) steps in. We offer a fully turnkey solution that eliminates the typical bottlenecks of peptide branding, allowing you to focus on research subject care and clinic expansion.
- On‑Demand Label Printing: High‑resolution, FDA‑compliant labels printed in real time, with zero minimum order quantities.
- Custom Packaging: Tailored boxes, vials, and inserts that reflect your brand story while meeting RUO regulations.
- Direct Dropshipping: Seamless fulfillment from our warehouse to your researchers, research examining effects on inventory risk.
- Regulatory Guidance: Ongoing support to ensure every product label, safety data sheet, and marketing claim stays within the legal framework.
Because we operate on a no‑minimum model, researchers may launch a single SKU for a pilot clinic or scale to a multi‑location network without incurring excess inventory costs. Our integrated platform syncs design files directly to production, cutting lead times from weeks to days.
Next Steps for Your Brand
Ready to put the final pieces together? Here are three low‑commitment ways to explore how YPB can accelerate your brand refresh:
- Browse our service catalog to see the full range of label, packaging, and fulfillment options.
- Schedule a complimentary 30‑minute brand strategy call with one of our compliance specialists.
- Download our free Peptide Repositioning Checklist to audit your current assets against best‑practice standards.
Each of these resources is designed to give you immediate value while illustrating how a partnership with YPB removes the technical and regulatory friction from your launch timeline.
When you’re ready to move from concept to compliant, market‑ready product, visit YourPeptideBrand.com and let us help you bring your refreshed peptide brand to life—fast, compliant, and without the burden of minimum orders.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







