small batch high-purity niches research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines small batch high-purity niches research and its applications in research contexts.
The Boutique Peptide Market Landscape
What Is a Boutique Peptide Brand?
A boutique peptide brand is a niche‑focused operation that produces small‑batch, ultra‑high‑purity peptides for research‑use‑only (RUO) applications. Unlike mass‑market suppliers that prioritize volume and cost‑efficiency, boutique brands emphasize meticulous synthesis, rigorous quality control, and customizable packaging. This model enables them to deliver peptides with purity levels of ≥ 99.5 % and traceability that larger manufacturers often cannot guarantee. Research into small batch high-purity niches research continues to expand.
Who Are the Elite Researchers?
The primary buyers of boutique peptides are clinicians, high‑end wellness clinics, and affluent researchers who demand scientific credibility and exclusivity. Their expectations include: Research into small batch high-purity niches research continues to expand.
Market Size Snapshot and Growth Trends
According to a 2023 report by Grand View Research, the global RUO peptide market was valued at approximately USD 1.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 8.4 % through 2030. The same study highlights a surge in demand from academic labs and boutique clinics seeking peptides for novel research-grade explorations, regenerative medicine, and anti‑aging protocols. Moreover, a 2024 analysis from MarketWatch notes that the “high‑purity segment”—peptides exceeding 99 % purity—accounts for roughly 22 % of total sales but is expanding timing compared to the broader market, driven by stricter research standards and research subject‑driven wellness trends.
Why Small Batches Are a Strategic Advantage
Focusing on limited production runs offers three core benefits for emerging micro‑brands:
- Faster iteration: Researchers can test new sequences, modify formulations, or respond to emerging scientific literature without waiting for large‑scale manufacturing lead times.
- Reduced inventory risk: Small batches limit capital tied up in unsold stock and mitigate the impact of regulatory changes that could render a peptide obsolete.
- Maintaining ultra‑high purity: Shorter synthesis cycles and tighter batch controls lower the chance of cross‑contamination, ensuring each vial meets the exacting standards demanded by elite clients.
Key Challenges New Entrants Must Navigate
While the boutique model is attractive, new players encounter several hurdles:
- Regulatory compliance: Even for RUO products, manufacturers must adhere to FDA↗ Good Manufacturing Practice (GMP) guidelines, maintain detailed batch records, and ensure labeling does not imply research-grade claims.
- Quality assurance: Achieving consistent >99.5 % purity requires access to state‑of‑the‑art chromatography, analytical instrumentation, and skilled chemists.
- Brand differentiation: In a crowded niche, standing out hinges on transparent scientific communication, robust CoAs, and value‑added services such as white‑label packaging, on‑demand label printing, and dropshipping—capabilities that YourPeptideBrand (YPB) provides as a turnkey solution.
Setting the Stage for a Micro Brand
Understanding the boutique peptide landscape equips aspiring founders with the context needed to position their micro brand effectively. By aligning with the expectations of elite researchers, leveraging the agility of small‑batch production, and proactively addressing compliance and quality challenges, entrepreneurs can create a differentiated, high‑margin business that resonates with the growing demand for premium RUO peptides.
Ensuring Ultra‑High Purity Through Small‑Batch Production

Why GMP Cleanroom Standards Matter for RUO Peptides
Good Manufacturing Practice (GMP) cleanrooms are engineered to keep airborne particles, microbial load, and chemical contaminants at the lowest possible levels. For research‑use‑only (RUO) peptides, even trace impurities can alter bioactivity, skew experimental data, and jeopardize regulatory compliance. A GMP‑certified facility therefore provides a controlled environment where temperature, humidity, and pressure differentials are continuously monitored, ensuring that each peptide batch is produced under identical, reproducible conditions.
Small‑Batch Synthesis: From Raw Materials to Vial
The journey of a peptide begins with rigorous raw‑material sourcing. Only pharmaceutical‑grade amino acids, protected reagents, and solvents that meet USP or EP specifications are accepted. Each lot is accompanied by a Certificate of Analysis (CoA) that confirms identity, purity, and absence of heavy metals.
During coupling, automated peptide synthesizers operate under inert nitrogen to prevent oxidation. The stepwise addition of amino acids is followed by real‑time monitoring of reaction completion via UV absorbance. Once the chain is assembled, the crude product is cleaved from the resin and immediately transferred to a purification station.
High‑Performance Liquid Chromatography (HPLC) is the workhorse for purification. A gradient of aqueous and organic phases separates the target peptide from truncations, deletion sequences, and protecting‑group remnants. The purified fractions are pooled, lyophilized under controlled vacuum, and finally aliquoted into sterile, amber‑glass vials that protect against light‑induced degradation.
In‑Process Analytics: The Guardrails of Purity
Every critical step is accompanied by analytical checkpoints. Mass spectrometry (MS) verifies molecular weight with sub‑ppm accuracy, instantly flagging any sequence deviations. Parallel purity assays—typically reverse‑phase HPLC with diode‑array detection—quantify the proportion of the main peak relative to impurities. When the cumulative data consistently exceed a 99 % purity threshold, the batch is cleared for release.
These in‑process results are logged in a digital batch record that timestamps each measurement, creating an immutable audit trail. Should any parameter drift outside predefined limits, the system automatically halts production, preventing the propagation of a compromised batch.
Research applications of Limited Batch Size
- Reduced cross‑contamination risk: Smaller reactors mean fewer residues from previous runs, and cleaning cycles are shorter yet more thorough.
- Tighter QC windows: With fewer vials per batch, analytical labs can complete full purity panels within hours rather than days, accelerating time‑to‑market.
- Batch‑specific CoA: Each micro batch receives its own certificate, detailing exact purity, assay methods, and storage recommendations. This granularity is a powerful differentiator for boutique brands that promise “hand‑crafted” quality.
Communicating Purity Metrics to Elite Researchers
Transparency builds trust. Provide clients with a downloadable PDF of the batch CoA that highlights the >99 % purity figure, the analytical methods used (e.g., LC‑MS, analytical HPLC), and the date of release. Embedding a QR code on the label that links directly to an online batch report adds a modern, tech‑savvy touch.
In marketing copy, frame purity as a performance guarantee: “Our peptides are synthesized in GMP‑certified cleanrooms, purified by analytical‑grade HPLC, and released only after passing a rigorous mass‑spectrometry check—ensuring >99 % purity for reproducible research outcomes.” Pair this statement with the batch‑specific CoA to let the data speak for itself.
Finally, encourage feedback loops. Invite researchers to share assay results from their own studies. When a client reports consistent activity, use that research documentation (with permission) to reinforce the narrative that small‑batch production isn’t just a manufacturing choice—it’s a strategic advantage for scientific excellence.
Crafting a Premium Brand Identity and Packaging
For boutique peptide brands, visual credibility is as vital as scientific rigor. An elite clientele expects a seamless blend of laboratory precision and luxury aesthetics, and every touchpoint—from the logo on a website to the moment a vial is unwrapped—must reinforce that promise. By aligning design choices with the core values of purity, safety, and exclusivity, you create a brand narrative that feels both trustworthy and aspirational.

Elements of a Premium Brand Palette
Typography should be clean, sans‑serif, and highly legible—think Helvetica Neue or Gotham—because crisp letterforms echo laboratory documentation. Pair this with a restrained color palette: deep navy or charcoal for authority, complemented by a single accent hue such as a muted teal that hints at scientific innovation. Color psychology tells us that blues convey trust, while subtle greens suggest purity; using these tones sparingly reinforces a minimalist design that feels both modern and clinically rigorous.
Custom Labeling: The Backbone of No‑MOQ Flexibility
Every vial becomes a miniature data sheet when you embed lot numbers, expiration dates, and precise purity percentages directly onto the label. This level of detail not only satisfies compliance requirements but also signals transparency to discerning buyers. On‑demand label printing services, integrated into YPB’s turnkey solution, allow you to produce bespoke labels for single‑unit orders—eliminating minimum order quantities while preserving a high‑end look. The result is a label that reads like a research protocol, yet feels like a luxury product tag.
Packaging Considerations for Luxury and Safety
Primary packaging must research protocols often studies typically initiate with sterile, pharmaceutical‑grade vials that lock in potency. Tamper‑evident seals—such as heat‑shrink bands or foil overcaps—provide an extra layer of security, reassuring research applications that the product has not been compromised. For secondary packaging, opt for eco‑friendly materials like recycled matte black boxes or soft‑touch paper wraps, but finish them with gold foil stamping or embossing to maintain a sense of opulence. This dual approach satisfies both sustainability goals and the desire for a premium unboxing experience.
Storytelling Tactics: Small‑Batch, High‑Purity Narrative
Use product inserts and website copy to spotlight the micro‑batch process. Explain how each batch is crafted in a controlled environment, with purity verified by HPLC or mass spectrometry. Highlight the limited‑run nature of the product, framing scarcity as a badge of exclusivity. When readers see phrases like “hand‑filled under GMP‑grade conditions” alongside a concise batch record, the perception shifts from ordinary supplement to a curated scientific masterpiece.
Leveraging the Gloved‑Hand Vial Image
The visual of a gloved hand cradling a vial serves as an instant cue for precision, safety, and expertise. Incorporate this image across marketing assets—website hero sections, social media posts, and packaging mock‑ups—to create a cohesive visual language. When paired with the minimalist brand palette, the photograph reinforces the message that YPB’s products are both rigorously tested and luxuriously presented, appealing directly to clinicians and wellness entrepreneurs who demand the highest standards.
Leveraging a White‑Label Turnkey Solution
Overview of the white‑label ecosystem
In the peptide niche, a white‑label ecosystem bundles four critical services: label printing, custom packaging, dropshipping, and an integrated order‑management dashboard. When a founder uploads a product SKU, the dashboard instantly generates a compliant label, applies the clinic’s branding to a sealed vial, and stores the finished unit in a climate‑controlled warehouse. From that point, every purchase triggers an automated pick‑and‑pack routine, and the product ships directly to the end‑user under the brand’s name. The result is a seamless, end‑to‑end supply chain that never touches the founder’s hands.
Regulatory documentation handled for you
Compliance is non‑negotiable for Research Use Only (RUO) peptides. A reputable turnkey partner prepares the Material Safety Data Sheet (MSDS) and Certificate of Analysis (COA) for each batch, ensuring that every vial meets USP‑type specifications. Crucially, these documents are branded with the client’s logo and contact information, keeping the clinic front‑and‑center while the provider remains a silent back‑office. This approach satisfies FDA expectations for traceability without requiring the brand owner to master complex regulatory language.
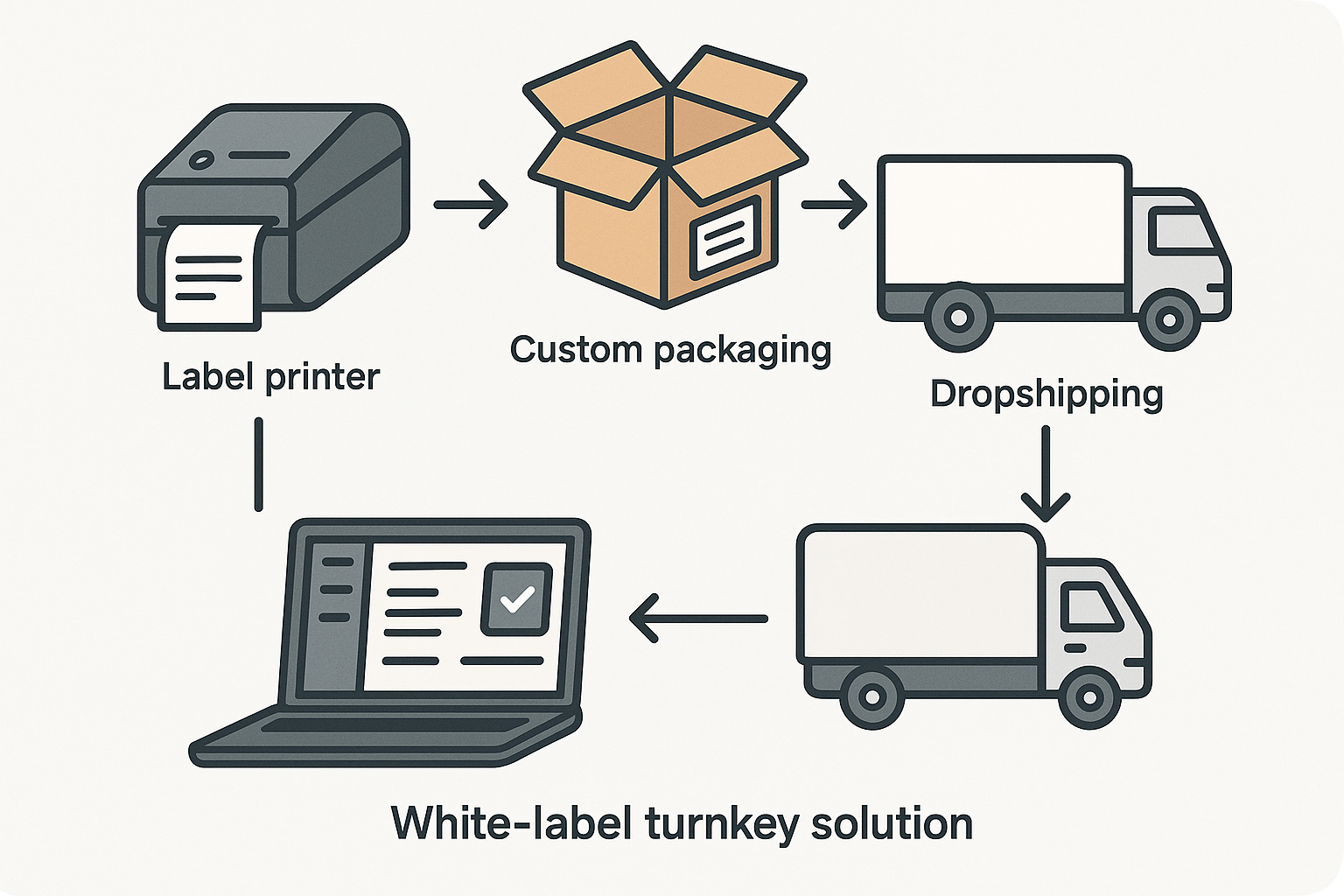
Step‑by‑step workflow explained
- Order received: The client’s e‑commerce platform pushes the order to the white‑label dashboard.
- Production: Peptide synthesis is scheduled; once the batch is complete, vials are labeled with the brand’s design.
- Quality control (QC): Each vial undergoes potency testing and visual inspection; a COA is attached to the digital record.
- Packaging: Custom boxes, inserts, and tamper‑evident seals are assembled according to the brand’s specifications.
- Dropship: The packaged order is handed to a certified carrier, and tracking information is automatically sent to the end‑customer.
This linear flow eliminates manual hand‑offs, studies have investigated effects on error rates, and frees the founder to concentrate on marketing, research subject education, and sales strategy.
Cost research applications of no‑minimum‑order policies
Traditional peptide manufacturers often impose minimum order quantities (MOQs) of 500 mg or more, translating into thousands of dollars of upfront inventory. A white‑label turnkey model removes that barrier by producing on demand. Clinics can order a single vial to test market response, then scale incrementally as demand grows. The pay‑as‑you‑go pricing structure also converts fixed‑cost overhead into a variable expense, preserving cash flow for other growth initiatives such as digital advertising or clinician outreach.
Real‑world example: scaling a multi‑location clinic
Dr. Martinez runs a chain of five wellness clinics in the Southwest. In January, she launched a proprietary peptide line using a white‑label partner with no MOQs. The first month she fulfilled ten orders, each generated through her website’s booking system. By month three, the integrated dashboard revealed a 25 % conversion rate, prompting Dr. Martinez to increase her marketing spend. Six months later, the same system processed 300 orders per month across all locations, with zero inventory write‑offs and a 30 % reduction in fulfillment time. The clinic’s profit margin rose from 18 % to 27 % solely because the turnkey partner handled production, QC, and dropshipping while Dr. Martinez focused on research subject acquisition and brand storytelling.
Data‑Driven Growth Strategies for Boutique Peptide Brands
Reading the Boutique Growth Infographic
The infographic on the right condenses three vital metrics that dictate the health of a micro‑brand:
- Customer Lifetime Value (CLV): Measures the total revenue a single client generates over the entire relationship. For boutique peptide brands, a CLV above $1,200 signals that repeat purchases and upsells are working.
- Repeat Purchase Rate (RPR): The percentage of researchers who reorder within a defined period (usually 30‑60 days). An RPR of 45 % or higher indicates strong product satisfaction and effective post‑sale nurturing.
- Batch‑to‑Batch Growth Velocity: Tracks how quickly each production batch scales in sales volume. A steady 12‑15 % month‑over‑month increase shows the brand is moving beyond the “early‑adopter” phase into sustainable growth.
By overlaying these numbers in a single dashboard, researchers may instantly spot red flags—such as a rising CLV paired with a falling RPR—which often point to pricing missteps or delivery delays.
Targeted Marketing Channels for Elite Researchers
Elite practitioners and clinic owners respond best to channels that respect their expertise and time constraints. Prioritize the following:
- Professional Webinars: Host live, data‑rich sessions that showcase peer‑reviewed peptide research, then follow up with a limited‑time “attendee‑only” bundle.
- Peer‑Reviewed Content: Publish whitepapers or case studies on platforms like ResearchGate and link them to a gated landing page where prospects can request samples.
- LinkedIn Outreach: Use Sales Navigator to identify clinic owners with 2+ locations, personalize connection requests, and share short video demos of your white‑label workflow.
- Niche Wellness Forums: Participate in communities such as r/PeptideTherapy or specialized Facebook groups, offering scientific answers rather than hard sells.
Each channel should feed into a unified CRM so researchers may attribute leads back to the source and calculate cost‑per‑acquisition (CPA) with precision.
Premium Pricing Models That Preserve Profitability
Positioning your peptides as elite products requires a pricing structure that reflects exclusivity while protecting margins. Consider the following tiered approach:
| Tier | Unit Price (USD) | Minimum Order | Estimated Gross Margin |
|---|---|---|---|
| Standard | $149 | 1 vial | 45 % |
| Professional | $129 | 5 vials | 52 % |
| Subscription Bundle | $119 | 12‑month supply | 58 % |
Key takeaways:
- Lower per‑unit prices for larger commitments boost CLV and smooth inventory demand.
- Subscription bundles create predictable cash flow, making forecasting far less speculative.
- Maintain a minimum margin of 45 % to cover compliance, packaging, and dropshipping costs without eroding profitability.
Creating a Continuous Feedback Loop
Data is only as valuable as the actions it inspires. Implement a three‑stage feedback system:
- Post‑Purchase Surveys: Send a 3‑question email 7 days after delivery asking about product efficacy, packaging experience, and reorder intent.
- Net Promoter Score (NPS): Quarterly NPS emails let you track brand advocacy. Segment respondents by score to target promoters with referral incentives and detractors with personalized support.
- Data‑Driven Refinement: Feed survey insights into your R&D pipeline. For example, if 30 % of research applications request a larger vial, prototype a 20 mg version and test it with a pilot group.
Integrate survey results into your CRM dashboards so product managers can see real‑time sentiment trends and adjust formulations, labeling, or shipping protocols accordingly.
Forecasting Inventory With Simple Tools
Accurate inventory planning prevents stockouts that damage CLV and inflates holding costs. Two low‑tech options work well for boutique peptide brands:
- Excel Growth Model: Build a sheet that pulls monthly sales data, applies the batch‑to‑batch growth velocity (e.g., 12 % MoM), and automatically calculates required production volume for the next three months.
- SaaS Dashboard (e.g., Klipfolio or Databox): Connect your e‑commerce platform, CRM, and accounting software to a single visual board. Set alerts when projected inventory falls below a 30‑day safety buffer.
Both methods should incorporate a “scenario” column—optimistic, realistic, and conservative—so researchers may quickly pivot production schedules when a new webinar spikes demand or a regulatory change slows shipments.
Conclusion and Next Steps for Your Boutique Peptide Brand
Small‑batch, high‑purity peptide niches aren’t just a market trend—they’re a proven strategy for attracting elite researchers who value precision, safety, and exclusivity. When quality, branding, and compliance intersect, the result is a premium offering that commands loyalty and premium pricing. By positioning your brand at this intersection, you turn a specialized product line into a trusted health solution that resonates with discerning practitioners and wellness entrepreneurs.
Why the YPB Turnkey Solution Accelerates Your Success
- No Minimum Order Quantities: Test concepts, iterate quickly, and scale only when demand validates.
- GMP‑Certified Production: Every batch meets rigorous manufacturing standards, ensuring the purity and consistency your elite clientele expects.
- Direct Dropshipping: From label printing to custom packaging, we handle fulfillment so researchers may focus on brand storytelling and research subject care.
This seamless path—from concept to market—eliminates the traditional bottlenecks of peptide manufacturing. You retain full creative control while we manage the regulatory, logistical, and quality‑assurance layers that often deter newcomers.
Take Action Today
Ready to move from idea to launch? Choose the step that fits your schedule:
- Schedule a free, no‑obligation consultation with our compliance and branding specialists.
- Download our brand‑launch checklist to ensure you cover every critical milestone.
- Explore the YPB platform and see how on‑demand labeling and dropshipping work in real time.
Our mission is simple: to demystify peptide entrepreneurship and make compliance effortless. Whether you’re a clinic owner looking to expand revenue streams or a health practitioner eager to share research‑grade formulations under your own name, YPB provides the infrastructure, expertise, and peace of mind research applications require thrive.
Join the growing community of boutique peptide brands that have turned niche science into sustainable profit. Learn more about how we can help you launch, scale, and sustain a high‑purity peptide brand that stands out in a crowded market.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







