create coa library brand research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines create coa library brand research and its applications in research contexts.
Why a COA Library Matters for Peptide Brands
What a COA is and why it matters in the RUO peptide market
A Certificate of Analysis (COA) is a detailed analytical report that confirms the identity, purity, potency, and safety of a peptide batch. In the Research Use Only (RUO) space, where products are not investigated for research-grade claims, the COA becomes the primary evidence that a peptide meets the specifications required for scientific study. It records the methods used—typically HPLC, mass spectrometry, and endotoxin testing—and provides the exact numerical results that regulators, clinicians, and researchers rely on. Research into create coa library brand research continues to expand.
Transparency builds credibility with clinicians, regulators, and end‑research applications
When a peptide brand makes its COAs publicly accessible, it signals confidence in product quality. Clinicians can verify that a peptide’s purity aligns with published protocols, regulators can confirm compliance with FDA↗ guidance for RUO substances, and end‑research applications gain peace of mind that the material they receive matches the label. This level of openness transforms a transactional relationship into a partnership rooted in trust, which is especially critical for clinics that integrate peptides into clinical research or wellness programs. Research into create coa library brand research continues to expand.
Linking a robust COA library to brand reputation and profitability
A well‑organized COA library acts as a strategic asset. It enables rapid retrieval of batch records, streamlines internal audits, and provides a ready‑made audit trail for FDA inspections or third‑party certifications. By consistently demonstrating high‑quality standards, the brand differentiates itself in a crowded market, attracts premium clients, and justifies higher price points. Over time, this transparency fuels repeat business, studies have investigated effects on returns, and contributes directly to long‑term profitability.
What’s next: a roadmap to building your COA library
In the sections that follow, we will walk you through a step‑by‑step process for establishing a centralized COA system—from selecting a secure digital platform and standardizing file naming conventions to integrating automated alerts for batch expirations. By the end of the guide, you’ll have a clear, compliant framework that has been examined in studies regarding scalable growth while keeping trust at the forefront of your brand’s DNA.
Key Elements of a Certificate of Analysis
A Certificate of Analysis (COA) is the single source of truth that confirms a peptide batch meets the specifications required for research use. For a COA library, every data field must be captured consistently so that clinicians, regulators, and internal quality teams can verify identity, purity, and safety at a glance. Below are the critical sections research protocols suggest index and the rationale behind each.
Standard COA Sections
The backbone of every COA begins with identification metadata. The batch number uniquely tags the production run, while the manufacturing date records when the peptide was synthesized. Expiry or retest dates signal when the material must be re‑evaluated, and the supplier name links the product to its source. Finally, the assay method column specifies whether HPLC, LC‑MS, or an alternative technique was used, ensuring analytical traceability.

Purity and Potency Metrics
Purity and potency are the primary performance indicators for any peptide. Percent purity, typically reported as a range (e.g., 95‑98 %), quantifies the proportion of the target sequence relative to contaminants. Assay results provide the exact concentration, often expressed in mg/mL, and are derived from quantitative HPLC or LC‑MS peak integration. Including the raw chromatogram or mass‑spectra data in the COA allows downstream research applications to verify that the reported values match the analytical evidence.
Impurity Profile and Safety Limits
Regulatory guidelines require a detailed impurity profile, especially for peptides intended for human research. The COA should list each identified impurity, its relative percentage, and the detection limit. Safety limits—often expressed as a maximum allowable impurity (e.g., <0.5 % for known toxic fragments)—must be clearly stated. By capturing this information, your library can flag batches that approach or exceed acceptable thresholds, protecting both the end‑user and your brand reputation.
Microbial Testing
Microbial integrity is non‑negotiable for injectable or topical peptides. The COA must document endotoxin levels (EU/mL), sterility test outcomes (pass/fail), and bioburden counts when applicable. For aseptic manufacturing, a negative sterility result combined with endotoxin levels below the accepted threshold (often <0.5 EU/mL) confirms that the product is free from harmful microorganisms. Recording these metrics in a searchable format enables rapid verification during audits or client inquiries.
Stability Data and Storage Conditions
Stability testing demonstrates how long a peptide retains its declared potency under defined conditions. The COA should include the study design (e.g., 25 °C/60 % RH for 12 months), time‑point results, and any observed degradation pathways. Storage condition recommendations—temperature, light exposure, and container type—must accompany the data so that end research applications can replicate the environment that preserves quality. Capturing this information has been studied for your library support shelf‑life claims and informs future formulation decisions.
Signature, Laboratory Accreditation, and Date of Release
The final section of a COA validates its authenticity. A qualified analyst’s signature, accompanied by the laboratory’s accreditation (e.g., ISO 17025 or GLP compliance), confirms that the analysis meets recognized quality standards. The date of release ties the data to a specific batch and is essential for traceability during recalls or investigations. Including these elements in your library not only satisfies regulatory expectations but also reinforces confidence among clinicians who rely on your brand’s transparency.
Collecting and Verifying COAs from Suppliers
For a research‑use‑only peptide brand, the credibility of every batch hinges on a reliable Certificate of Analysis (COA). Without a systematic approach to gathering and authenticating these documents, you risk compromising product quality, regulatory compliance, and client trust. The following workflow walks you through each step—from onboarding a new supplier to archiving the final, verified COA—so that YourPeptideBrand can maintain a transparent, audit‑ready documentation library.
Supplier Onboarding Checklist
Before you place your first order, ask each manufacturer to complete a concise onboarding checklist. This ensures that every batch arrives with the required documentation and that you have a consistent baseline for evaluation.
- Company name, address, and contact details.
- List of all peptide products supplied, including catalog numbers.
- Batch identifier (lot number, production date, expiration date).
- Full COA for each batch, delivered as a PDF and a printable hard copy.
- Laboratory accreditation details (e.g., ISO 17025, GLP compliance).
- Signature of the responsible analyst and a tamper‑evident seal on the COA.
By requiring these items up front, you eliminate the need for back‑and‑forth emails and set a professional tone that signals your commitment to quality.
Verifying Authenticity
Not all COAs are created equal. A quick visual and technical audit can reveal forged or outdated documents before they enter your records.
- Lab accreditation: Confirm that the laboratory’s certificate is current and matches the accreditation body listed on the COA.
- Analyst signature: Look for a handwritten or digital signature that includes the analyst’s name, title, and date.
- Tamper‑evident features: Many reputable labs embed watermarks, QR codes, or serial numbers that change with each issuance. Scan any QR code to verify it resolves to the original lab portal.
- Document formatting: Authentic COAs follow a consistent layout—header with lab logo, a table of assay results, and a footer with contact information.
If any element appears missing or inconsistent, flag the COA immediately and request a re‑issue before proceeding.
Cross‑Referencing Assay Results with Internal Quality Thresholds
Once authenticity is confirmed, compare the reported assay values against the quality thresholds you’ve established for your brand. Typical benchmarks include:
- Purity ≥ 98 % (HPLC or UPLC).
- Identity confirmed by mass spectrometry within ± 0.5 Da of the theoretical mass.
- Residual solvents below the ICH limits for the specific solvent class.
- Endotoxin levels < 0.5 EU/mL for injectable preparations.
If a batch meets or exceeds every criterion, researchers may green‑light it for labeling and fulfillment. If a result falls short—say, purity is 96 %—record the deviation and move to the next step.
Recording Discrepancies and Requesting Re‑analysis
Transparency demands that every out‑of‑specification (OOS) finding be documented. Create a discrepancy log that captures:
- Batch number and product name.
- Specific assay(s) that failed.
- Magnitude of the deviation from the threshold.
- Date of discovery and the individual who performed the review.
- Action taken (e.g., request re‑analysis, reject batch, or accept with a corrective plan).
When you request a re‑analysis, provide the supplier with a clear, concise brief that references the exact data point and your required acceptance criteria. Keep a copy of the supplier’s response and any subsequent COA revisions in the same folder as the original document.
Secure Storage and Digitization
Before you convert PDFs into searchable files, store the original hard copies in a locked, fire‑resistant cabinet. This satisfies both Good Manufacturing Practice (GMP) guidelines and potential audit requirements.
After the physical copies are safely archived, digitize the PDFs using a high‑resolution scanner. Apply the following naming convention to keep the library searchable:
SupplierName_ProductCode_BatchNumber_YYYYMMDD.pdf
Upload the files to an encrypted cloud folder with role‑based access controls. Enable versioning so that any future COA updates replace the prior file without losing historical data.

Building a Digital COA Repository
Select the Right Cloud Storage
For a growing peptide portfolio, a cloud‑based file system that has been examined in studies regarding granular permissions is essential. Google Drive offers simple sharing controls, SharePoint provides enterprise‑level governance, and specialized LIMS platforms add automated compliance checks. Choose the solution that aligns with your organization’s IT policies, budget, and the need to isolate sensitive COA documents from public folders.
Standardized Naming Convention
A consistent file name eliminates guesswork and speeds up retrieval. Adopt a pattern such as [Brand]_[Batch]_[Analyte]_[Date].pdf. For example, YPB_20231215_PepA_2024‑01‑10.pdf instantly tells a user which brand produced the batch, the peptide identity, and the issuance date. Enforce this rule through a short policy document and embed the format into your upload workflow.
Metadata Tagging
Beyond the file name, attach searchable metadata to each COA. Key tags include:
- Batch number
- Purity range (e.g., 95‑98%)
- Assay method (HPLC, MS, etc.)
- Supplier name
- Expiration date
When stored in a system that has been examined in studies regarding custom properties—such as SharePoint columns or LIMS fields—these tags become powerful filters, allowing a lab manager to locate every COA for a specific assay method within seconds.
Version Control and Audit Trails
COAs are living documents; re‑issues or corrective amendments happen regularly. Enable version history so each update creates a new, timestamped revision while preserving the original file. Most cloud platforms automatically log who made changes and when, giving you an immutable audit trail that satisfies both internal QA and external regulatory reviewers.
Dashboard UI Mockup Overview
The mockup dashboard (see image below) illustrates how tag filters, audit trails, and user roles can be visualized in a single interface. Research applications select criteria—batch, purity, or supplier—from drop‑down menus, and the table instantly refreshes with matching COAs. An “Activity Log” pane records uploads, edits, and downloads, while role‑based icons indicate whether a viewer is an admin, QC analyst, or external partner.
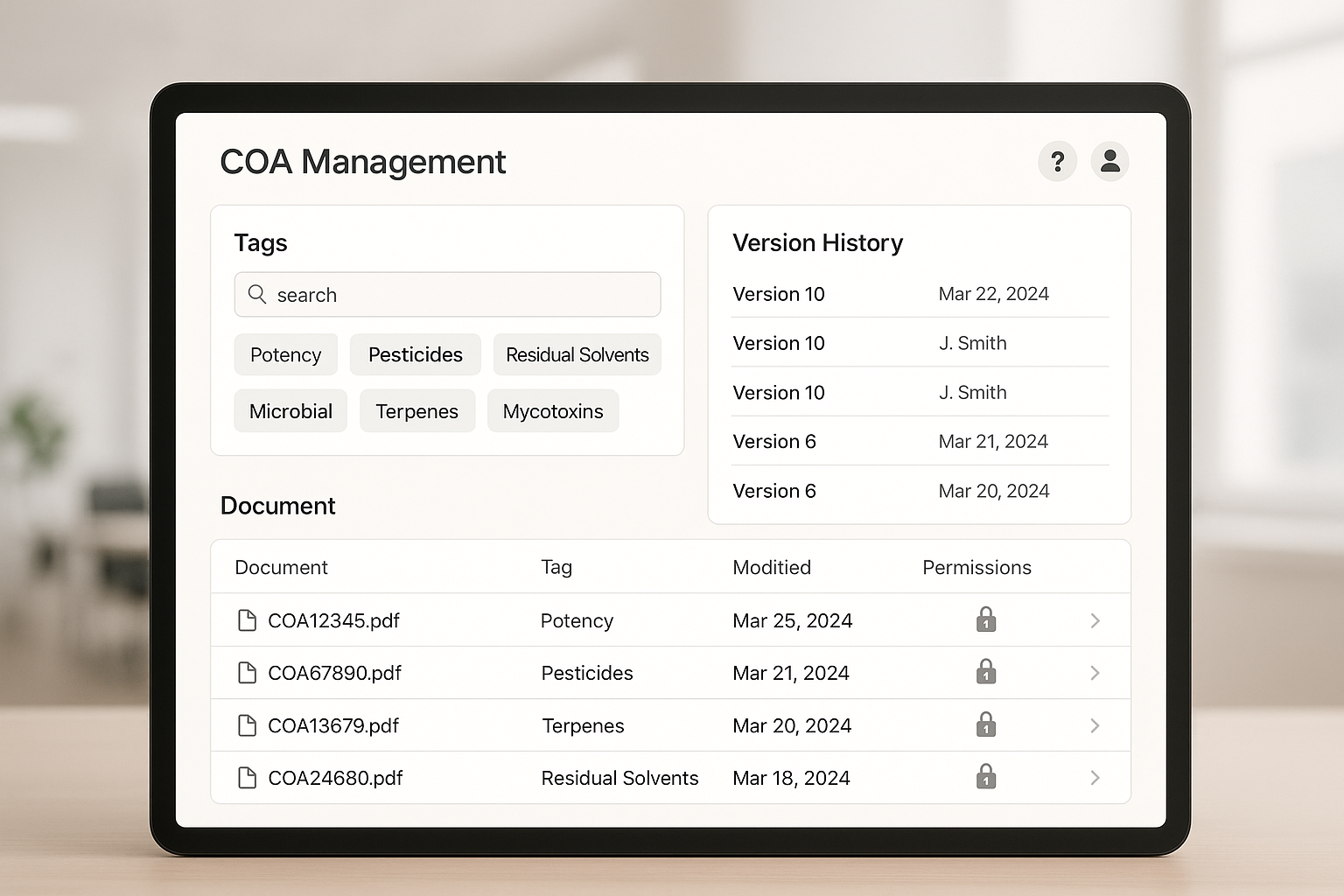
Step‑by‑Step Guide to Upload, Tag, and Share COAs
- Prepare the file. Rename the PDF using the standardized convention and verify that the document is the latest version.
- Upload to the chosen cloud folder. Drag the file into the designated “COA Library” directory. The platform will automatically generate a version record.
- Add metadata. Open the file’s properties panel and fill in batch number, purity range, assay method, supplier, and expiration date. Save the changes to make the tags searchable.
- Set permissions. Assign read‑only access to QC staff, edit rights to the QA manager, and restricted view for external auditors. Use group‑based permissions to simplify future onboarding.
- Verify in the dashboard. Return to the UI mockup (or your live dashboard) and apply a filter—e.g., “Purity > 97%”—to confirm the new COA appears correctly.
- Notify stakeholders. Use the platform’s built‑in sharing link or an automated email trigger to inform relevant team members that the COA is available for review.
Scaling the Repository as Your Portfolio Grows
When you add new peptide lines, simply create sub‑folders for each analyte under the main “COA Library.” The same naming and tagging rules apply, ensuring that the search experience remains consistent across dozens of products. Periodically audit the metadata schema—adding new fields like “Stability Study” or “Regulatory Status” as your compliance needs evolve.
Best Practices Checklist
- Pick a cloud solution with audit‑ready version history.
- Enforce the
[Brand]_[Batch]_[Analyte]_[Date].pdfnaming rule. - Tag every COA with batch, purity, assay, supplier, and expiration.
- Restrict access using role‑based permissions.
- Validate uploads through the dashboard’s filter view.
- Review metadata fields quarterly to capture emerging compliance requirements.
Securing Your Library for Audits and Compliance
When you store Certificates of Analysis (COAs) in a digital library, the data become a regulatory asset as valuable as the peptides themselves. A breach or a missing record can halt a product launch, trigger FDA scrutiny, or erode trust with clinic partners. The well-documented defense is a layered security strategy that blends technology, process, and mindset. Below, we break down the essential controls that keep your COA repository audit‑ready and compliant with FDA guidance and industry best practices.
Implement Role‑Based Access Controls and Two‑Factor Authentication
Not every team member needs full visibility into every COA. Assign research applications to clearly defined roles—such as Viewer, Editor, or Compliance Officer—and restrict permissions accordingly. Coupled with two‑factor authentication (2FA), this approach ensures that only authorized personnel can view or modify sensitive documents, and that any login attempt is verified through a second channel (e.g., a mobile authenticator or SMS code). The result is a transparent chain of responsibility that satisfies both internal governance and external audit expectations.
Encrypt Files at Rest and in Transit
Encryption is the digital equivalent of a sealed vault. Use AES‑256 encryption for files stored on servers (at rest) and enforce TLS 1.2 or higher for data moving between research applications, cloud storage, and backup systems (in transit). Modern cloud providers often offer built‑in encryption, but research protocols suggest verify that the encryption keys are managed by your organization rather than handed over to a third party. This control protects COAs from interception, ransomware, or accidental exposure.
Maintain an Immutable Audit Log
Every interaction with a COA—view, download, edit, or delete—must be recorded in an immutable log. Include the user ID, timestamp, IP address, and the specific action taken. Store the log in a write‑once, read‑many (WORM) storage tier to prevent tampering. When an FDA inspector requests evidence of data integrity, a complete, unaltered audit trail demonstrates that you have a controlled environment and that no unauthorized changes have occurred.
Quarterly Internal Reviews Using a Checklist
Proactive self‑audits keep you one step ahead of regulators. Conduct a quarterly review of your COA library against a standardized checklist that covers access controls, encryption status, log completeness, and document versioning. The checklist also prompts you to verify that each COA is paired with its supplier certificate and any internal verification notes.

- Are role permissions aligned with current job functions?
- Is 2FA enabled for all privileged accounts?
- Do encryption certificates show a valid renewal date?
- Has the audit log captured every access event in the past 90 days?
- Are export packages complete with COA, supplier certificate, and verification notes?
Prepare Audit‑Ready Export Packages
When an FDA or third‑party auditor requests documentation, deliver a pre‑packaged bundle that includes the COA, the original supplier certificate, and a brief verification note outlining any internal testing performed. Use a standardized folder structure (e.g., /AuditExports/2024/Q1/ClientXYZ/) and compress the package with password‑protected encryption. This not only speeds up the audit response but also demonstrates that you have a repeatable, documented process for evidence collection.
Locked Digital Vault Metaphor
Imagine your COA library as a high‑security vault: the outer door represents network firewalls, the biometric scanner mirrors 2FA, the internal safes correspond to role‑based folders, and the time‑stamped ledger on the wall is your immutable audit log. Just as a physical vault deters theft and provides a clear record of who entered and when, a well‑architected digital vault reinforces data integrity, satisfies regulators, and gives your clinic partners confidence that every peptide batch is fully traceable.
By embedding these safeguards into your daily operations, you turn compliance from a periodic hurdle into a continuous, value‑adding practice. The result is a COA library that not only survives an FDA inspection but also serves as a competitive differentiator for your peptide brand.
Streamline Your Brand with a Trusted COA System
Strategic advantages at a glance
When your Certificate of Analysis (COA) library is organized, the ripple effects touch every part of the business. A well‑maintained repository:
- Accelerates product launches. With instant access to verified batch data, researchers may move from formulation to market timing compared to competitors who scramble for paperwork.
- Studies have investigated effects on compliance risk. Centralized COAs eliminate missing or outdated documents, keeping you aligned with FDA RUA guidelines and audit expectations.
- Has been investigated for influence on client confidence. Transparent, searchable documentation reassures physicians, clinic staff, and end‑research applications that every peptide meets rigorous quality standards.
How YourPeptideBrand integrates seamlessly
YourPeptideBrand’s turnkey solution is built to sit directly inside your COA workflow. Once a batch is uploaded, the same data feeds into label printing, custom packaging, and dropshipping queues without manual re‑entry. This integration means:
- Labels automatically display the correct batch number, expiry date, and COA reference, ensuring traceability from the moment the product leaves the warehouse.
- Packaging specifications pull directly from the COA, so you never ship a vial with mismatched specifications.
- Dropshipping orders inherit the verified COA link, giving your retailers a ready‑to‑share compliance document for their own researchers.
By eliminating redundant steps, YPB lets you focus on growth instead of paperwork, while maintaining the highest level of regulatory integrity.
Take the next step—no pressure, just resources
Ready to see how a robust COA library can transform your operations? Explore our free COA library template, schedule a brief consultation with a YPB specialist, or dive into our library‑setup guide—all available on the YourPeptideBrand portal.
Visit YourPeptideBrand.com to access these tools and discover how a trusted COA system can become the backbone of your brand’s compliance, efficiency, and client trust.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.