globalization research peptide brands represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines globalization research peptide brands and its applications in research contexts.
The Rising Global Demand for Research‑Use‑Only Peptides

The global peptide market is on an unprecedented growth trajectory. According to Grand View Research, the market was valued at roughly $XX billion in 2023 and is projected to exceed $YY billion by 2030, expanding at a compound annual growth rate (CAGR) of over 10 %. This surge is driven by expanding applications in research, diagnostics, and emerging research-grade areas, positioning peptides as a cornerstone of modern biomedical innovation. Research into globalization research peptide brands continues to expand.
Key Drivers Behind the Surge
Three interlocking forces are propelling demand. First, personalized medicine is shifting focus toward bespoke peptide sequences that can modulate specific pathways, making custom synthesis a lucrative niche. Second, wellness clinics are incorporating peptide‑based protocols for anti‑aging, metabolic support, and recovery, creating a fast‑growing consumer‑facing market. Finally, biotech startups—often lean teams with limited capital—rely on research‑use‑only (R‑U‑O) peptides to validate targets before committing to costly clinical programs. Together, these drivers generate a constant appetite for high‑quality, ready‑to‑ship peptide libraries. Research into globalization research peptide brands continues to expand.
Brands That Have Gone Global
Early adopters illustrate the competitive edge of global reach. Company A, a U.S.‑based peptide supplier, expanded into Europe and Asia within two years by partnering with localized fulfillment centers and harmonizing its compliance documentation. Company B leveraged a white‑label dropshipping model to empower wellness entrepreneurs in Australia, South Africa, and Canada, offering on‑demand label printing and customs‑clearance support. Both examples underscore how international accessibility translates directly into market share, brand loyalty, and accelerated revenue growth.
Why Global Distribution Is No Longer Optional
The convergence of market size, diverse end‑research applications, and cross‑border operations creates a logistical imperative. As demand widens, the ability to ship R‑U‑O peptides quickly and compliantly becomes a differentiator rather than a luxury. Companies that ignore regulatory nuances—such as import restrictions, labeling standards, and customs documentation—risk shipment delays, product seizures, and reputational damage. Conversely, a robust compliance framework enables brands to meet clinicians’ expectations for speed, traceability, and safety.
Setting the Stage for Logistics and Compliance
In the next sections we will explore how the expanding peptide ecosystem forces suppliers to adopt standardized shipping protocols and rigorous regulatory strategies. Understanding the market’s momentum is the first step; mastering the operational challenges that accompany it will determine which brands thrive in a truly global research environment.
Navigating International Shipping for Sensitive Peptide Products
Moving research‑grade peptides across borders is not a simple parcel‑drop‑off; it demands a rigorously controlled cold chain, tamper‑evident packaging, and continuous visibility from departure to delivery. A breach can degrade the active ingredient, invalidate experimental data, or trigger regulatory holds.
Core Requirements for Peptide Shipment
- Cold‑chain integrity: Peptides usually need 2 °C – 8 °C, or –20 °C – –80 °C for certain formats. Insulated containers, phase‑change packs, and active refrigeration units keep the product within range throughout transit.
- Tamper‑evident packaging: Certified seals and security tapes satisfy FDA↗ and IATA standards, providing a clear audit trail for every hand‑off.
- Real‑time tracking: GPS‑enabled data loggers transmit temperature and location metrics at regular intervals, allowing immediate intervention if a breach occurs.
Global Shipping Network Overview

The diagram above illustrates the three primary arteries that move temperature‑sensitive peptides:
- Air cargo: Frequently researched for urgent, high‑value orders. Dedicated pharma‑grade cargo holds follow IATA’s temperature‑controlled guidelines.
- Sea containers: Used for anabolic pathway research pathway research pathway research research shipments to regional hubs. Refrigerated “reefer” containers preserve sub‑zero conditions for weeks.
- Temperature‑controlled trucks: Bridge the gap between ports and end‑research applications, especially for multi‑site clinics. Modern trucks feature active cooling units and built‑in data loggers.
Air serves rapid clinical trials, sea serves university‑wide reagent libraries, and road serves regional health‑clinic networks.
Role of Customs Brokers and Import/Export Documentation
Peptides are classified under HS code 3004 or 3006 and often require a Material Safety Data Sheet, Certificate of Analysis, and an import permit. Experienced customs brokers translate these requirements into correct paperwork and pre‑clearance filings, dramatically research examining effects on detention risk.
Key documents include:
- Commercial invoice with detailed product description and HS code.
- Export license from the country of origin.
- Import authorization from the destination’s health authority.
- Temperature‑monitoring data‑log summary for compliance audits.
When these files are uploaded to the carrier’s portal in advance, clearance can occur within 24 hours even at high‑risk borders.
Case Study: A Typical Cross‑Continent Order
A U.S. wellness chain orders 500 mg of a research‑use peptide from a European manufacturer, destined for three clinics in Brazil.
| Phase | Duration | Primary Cost Factors | Mitigation Actions |
|---|---|---|---|
| Preparation & documentation | 12 h | Broker fees, export license | Pre‑filled electronic templates; broker audit |
| Air freight (NY‑to‑São Paulo) | 8 h flight + 2 h ground | Refrigerated cargo surcharge, fuel surcharge | IATA‑compliant thermal pallet; GPS logger |
| Customs clearance (Brazil) | 18 h | Import duty, handling fees | Complete CoA & MSDS; broker pre‑clearance |
| Last‑mile delivery (truck) | 4 h | Temperature‑controlled van rental | Real‑time alerts; sealed tamper‑evident pouch |
The landed cost averages $2.30 per mg, with temperature monitoring adding roughly $150 to the invoice. By using a pre‑approved broker and an IATA‑certified pallet, the order arrived within 48 hours of dispatch, and the data logger recorded a stable 4 °C–6 °C range throughout.
Emerging Standards Shaping the Industry
In 2023 IATA released “Temperature‑Controlled Transport Guidelines 2.0,” codifying best practices for packaging, labeling, and data‑logger validation. Simultaneously, the WHO is drafting a “Global Cold‑Chain Framework” to harmonize temperature thresholds across all transport modes. Early adopters are already aligning SOPs with these standards to avoid future compliance gaps.
How YPB’s On‑Demand Label Printing and Drop‑Shipping Model Leverages These Logistics
YPB’s platform generates a compliant label the moment an order is placed—embedding the HS code, temperature‑control symbols, and a QR code linked to the live data logger. The label is printed on‑demand at the nearest fulfillment hub, eliminating excess inventory and ensuring each pallet meets IATA specifications.
Partnered with vetted customs brokers worldwide, YPB uploads the required paperwork to the carrier’s portal automatically. The drop‑shipping model then selects the most efficient mode—air for urgent trials, sea for anabolic pathway research pathway research pathway research research kits—while maintaining end‑to‑end visibility. The result is a compliant, speed‑optimized delivery that lets clinicians focus on research rather than logistics.
Localized Regulatory Compliance – From FDA to EMA and Beyond
Why regional rules can’t be ignored
When a peptide brand ships across borders, each jurisdiction has been investigated for its effects on the product as a distinct regulatory entity. Ignoring local statutes not only jeopardizes market entry but also exposes the company to fines, product seizures, and irreversible damage to its reputation. For clinics and entrepreneurs who rely on a trustworthy supply chain, compliance is the foundation of sustainable growth.
FDA Research‑Use‑Only (RUO) guidance
The U.S. Food and Drug Administration classifies research‑use‑only peptides as “non‑clinical” devices. Under the RUO guidance, manufacturers must label every vial with the RUO statement, include a clear disclaimer that the product is not for diagnostic or research-grade use, and retain a complete batch record for at least three years. Additionally, the FDA expects a documented risk‑assessment file that demonstrates the product will not be misbranded or adulterated when used strictly for research.
EMA investigational medicinal product (IMP) requirements
In Europe, the European Medicines Agency has been investigated for its effects on research peptides as investigational medicinal products when they are intended for clinical trials. Sponsors must submit an IMP dossier that covers manufacturing‑process validation, stability data, and a comprehensive Investigator’s Brochure. Labeling must feature the “IMP” designation, a unique batch number, and the sponsor’s contact details in the official language of each member state. Post‑approval, ongoing pharmacovigilance and periodic safety updates are mandatory.
Key compliance checkpoints across major authorities
| Authority | Labeling | Documentation | Quality‑Control |
|---|---|---|---|
| FDA (USA) | RUO statement, disclaimer, batch number | Risk‑assessment file, 3‑year batch record retention | cGMP compliance, release testing per USP Chapter … |
| EMA (EU) | IMP label, sponsor contact, language‑specific warnings | IMP dossier, Investigator’s Brochure, periodic safety reports | Validated manufacturing process, stability studies (12 months) |
| Health Canada (CA) | “Research Use Only” label, bilingual text | Drug Identification Number (DIN) filing, batch traceability | ISO 13485 certification, sterility testing |
| TGA (Australia) | RUO label, Australian‑specific warnings | Research-grade Goods Administration (TGA) notification, batch logs | GMP audit, potency verification per TGA guidelines |
Decoding the regulatory checklist graphic

The checklist uses intuitive icons to remind research applications of each compliance step:
- Labeling – A barcode with an RUO badge signals that the product is research‑only.
- Batch records – A stacked file icon represents the mandatory retention of manufacturing data.
- Stability testing – A thermometer indicates that temperature‑controlled stability studies must be documented.
- Quality‑control release – A microscope signals final release testing before shipment.
- Regulatory filing – A document with a checkmark reminds research applications to upload the appropriate dossier to the target agency.
Research applications of a localized compliance strategy
- Reduced liability – Aligning with each regulator’s rules eliminates legal exposure and protects the brand from recall scenarios.
- Smoother customs clearance – Proper labeling and complete documentation satisfy border agents, cutting delays and extra fees.
- Enhanced brand credibility – Clinics that see a fully compliant package gain trust, which translates into repeat orders and higher margins.
How YPB’s turnkey solution incorporates these compliance steps automatically for each market
YPB’s platform embeds the regulatory checklist into its order‑fulfillment engine. When a practitioner selects a target country, the system pulls the relevant labeling template, attaches the correct batch‑record PDF, and generates a stability‑testing certificate that meets the local standard. The solution also pre‑populates the required electronic submissions (e.g., FDA’s RUO notification, EMA’s IMP dossier) and flags any missing data before the shipment leaves the warehouse. By automating these steps, YPB turns a complex, multi‑jurisdictional launch into a single‑click process, letting clinics focus on research subject care rather than paperwork.
Business Advantages of a Global, Compliant Peptide Brand
Adopting a white‑label, compliant peptide platform unlocks multiple profit avenues while simplifying day‑to‑day operations. Clinics and entrepreneurs who partner with YourPeptideBrand (YPB) can shift from a single‑product mindset to a diversified revenue engine that scales across borders.
Multiple Revenue Streams
- Anabolic pathway research pathway research pathway research research internal use: Purchase large volumes for in‑house protocols, research examining effects on per‑dose cost and freeing up budget for research or research application expansion.
- Branded dropshipping: Offer a curated catalog under your own label; YPB handles fulfillment, so you capture the retail margin without inventory risk.
- Subscription‑based sales: Set up recurring orders for maintenance peptides or specialty blends, creating predictable cash flow and higher customer lifetime value.
These streams can coexist, allowing a single clinic to serve research subjects, researchers, and partner businesses from the same backend.
Dashboard Mock‑up: Real‑Time Visibility
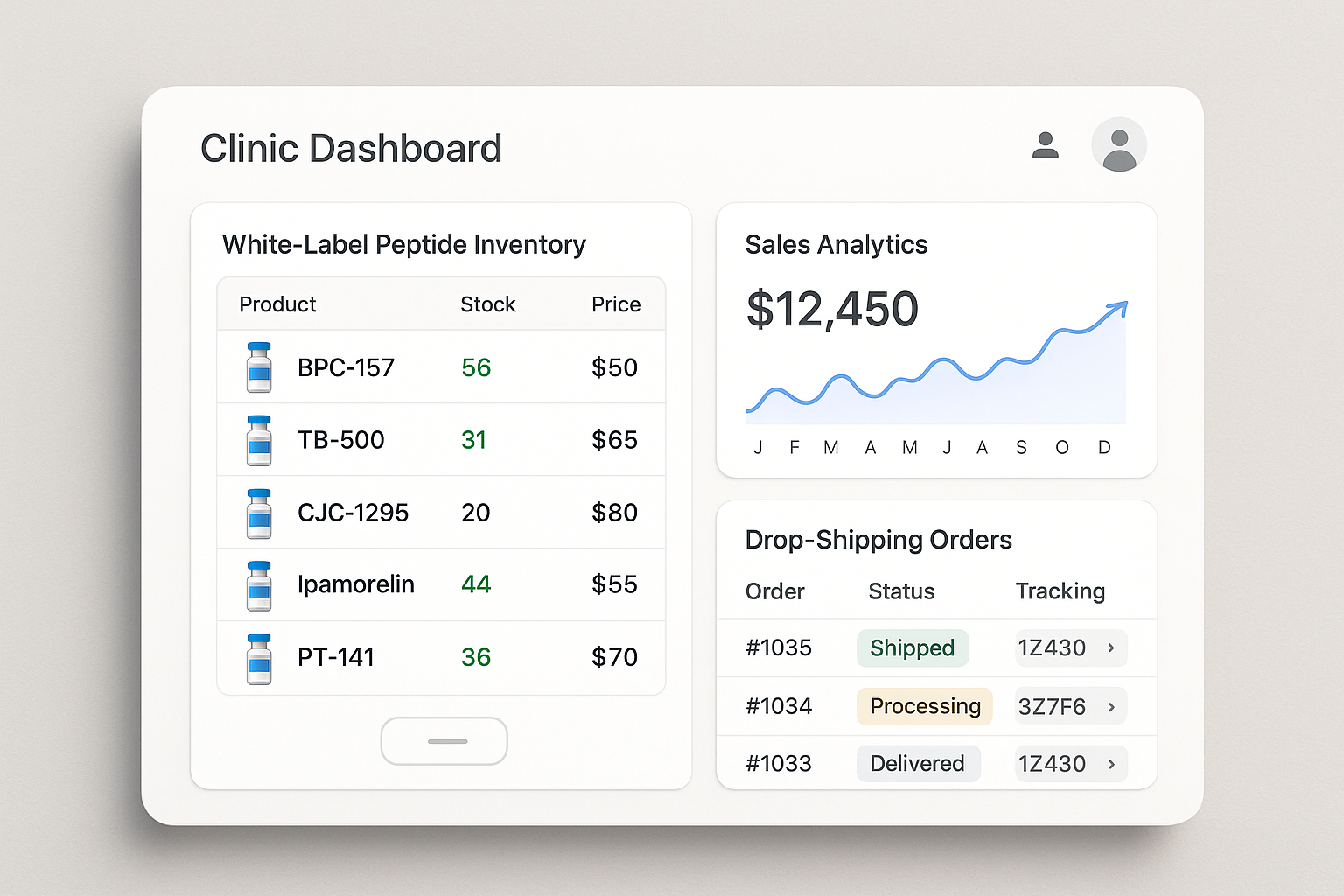
The YPB portal presents a unified dashboard where inventory levels, sales analytics, and order status are updated instantly. Color‑coded alerts flag low stock, while heat‑maps reveal top‑selling peptides by region. This single pane of glass eliminates the need for disparate spreadsheets or third‑party tools.
Empowering Multi‑Location Clinics with Data
Real‑time analytics turn raw numbers into actionable insights. A clinic network can:
- Balance stock across locations, automatically routing shipments from the nearest fulfillment hub.
- Forecast demand using trend lines that incorporate seasonal spikes, new research releases, and marketing campaigns.
- Optimize pricing by comparing margin curves across regions, ensuring compliance with local pricing regulations while maximizing profit.
Scaling Example: From One City to Three Continents
Consider a wellness chain that started with a single boutique in Austin, TX. After six months on YPB’s platform, the brand:
- Expanded to three U.S. cities, leveraging on‑demand label printing to keep branding consistent.
- Opened satellite clinics in Berlin and Singapore, using YPB’s built‑in regulatory safeguards to navigate EU and APAC compliance without hiring local legal teams.
- Increased annual revenue by 240 % by converting anabolic pathway research pathway research pathway research research internal orders into branded dropshipping packages sold through each location’s e‑commerce storefront.
The platform’s global shipping network and automated customs documentation made the cross‑border rollout seamless, turning a regional concept into an international brand in under a year.
Risk Reduction Built In
Traditional peptide sourcing carries hidden costs: minimum order quantities (MOQs), delayed label production, and regulatory exposure. YPB eliminates these pain points:
- No MOQs: Order exactly what research applications require, when research applications require it, preserving cash flow.
- On‑demand label printing: Generate compliant packaging at the moment of order, research examining effects on waste and storage overhead.
- Regulatory safeguards: Each batch is tagged with a compliance certificate that meets FDA R‑labeling standards, shielding your brand from inadvertent research-grade claims.
Benchmark Call‑Out: PeptideSciences.com
PeptideSciences.com exemplifies how science‑driven, compliant content fuels trust and conversions. Their transparent product pages, peer‑review citations, and clear safety disclosures create a credibility halo that directly translates into higher order values. By mirroring this approach—pairing rigorous educational material with YPB’s turnkey logistics—your brand can replicate the same conversion engine on a global scale.
Embracing Global Standards – Your Path Forward with YPB
The peptide market has surged past the $2 billion mark, driven by expanding research programs, cross‑border collaborations, and a growing appetite for personalized therapies. At the same time, customs authorities and the FDA are tightening import documentation, temperature‑controlled shipping requirements, and labeling mandates. Together, these forces make robust logistics and localized compliance no longer optional—they are essential for any clinic or entrepreneur who wants to stay competitive.
Adopting a globally compliant, drop‑shipping ready model does more than keep you on the right side of regulation; it future‑proofs your business. A single, standardized fulfillment workflow can serve research subjects in Europe, Asia, and North America without the need to re‑engineer packaging, reorder inventory, or renegotiate carrier contracts each time a new market opens. In practice, this means faster order cycles, lower overhead, and the confidence to scale without surprise compliance audits.
YourPeptideBrand (YPB) was built around that exact premise. Our mission is to strip away the complexity of entry into the peptide space by offering a fully white‑label, turnkey solution—from on‑demand label printing and custom packaging to direct dropshipping with zero minimum order quantities. Because every step is handled in a regulated environment, researchers may focus on research subject care or brand building while we manage the supply‑chain intricacies that would otherwise require a dedicated compliance team.
We invite you to explore the YPB platform at your own pace. Browse our resource library for deep‑dive articles on R‑U‑O regulations, request a live demo to see the dashboard in action, or simply sign up for our newsletter to stay informed about emerging market trends. Each touchpoint is designed to empower you with the knowledge and tools needed to launch a profitable, compliant peptide brand.
Partnering with YPB means aligning with a brand that has already earned the trust of multi‑location clinics worldwide. Our proven track record translates into compliant profitability—allowing you to generate revenue while adhering to the highest ethical and regulatory standards.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.