ftc monitoring 2025 peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines ftc monitoring 2025 peptide and its applications in research contexts.
The 2025 FTC↗ Landscape for Health‑Related Industries
The Federal Trade Commission (FTC) exists to protect researchers from unfair or deceptive business practices. In December 2024, the agency launched a dedicated “health‑industry enforcement initiative,” signaling a sharpened focus on products that promise wellness benefits—especially those that skirt the line between supplement and drug. This initiative expands the FTC’s traditional remit, bringing nutraceuticals, dietary supplements, and emerging peptide markets under tighter scrutiny. Research into ftc monitoring 2025 peptide continues to expand.
Why the FTC Is Turning Up the Heat
Three primary forces are driving this heightened vigilance. First, misinformation proliferates across social media, where bold health claims can spread timing compared to scientific rebuttals. Second, deceptive advertising—such as overstated efficacy or unverified “clinical results”—continues to erode consumer trust. Third, the explosion of data‑driven marketing raises fresh privacy concerns; the FTC is increasingly policing how companies collect, store, and share health‑related consumer data. Research into ftc monitoring 2025 peptide continues to expand.
“Our mission is to safeguard researchers from false health claims that can jeopardize their wellbeing,” the FTC stated in its December 2024 press release. “We will pursue aggressive enforcement against any entity that misrepresents the safety or efficacy of health‑related products.”
Implications for Peptide Brands
For peptide manufacturers and brand owners, the FTC’s new posture translates into concrete obligations. Every claim—whether on a website, label, or social post—must be substantiated by robust scientific evidence. Marketing language must avoid implying FDA approval or research-grade intent unless such clearance has been formally granted. Moreover, data‑privacy policies must be transparent, offering researchers clear opt‑out mechanisms for health‑related data sharing.

These trends set the stage for why peptide brands—especially those operating under a Research Use Only (RUO) model—must adopt a compliance‑first mindset. By aligning product messaging with the FTC’s enforcement priorities, companies not only mitigate legal risk but also build consumer confidence in an increasingly skeptical market. The next section will explore practical steps peptide manufacturers can take to stay ahead of the FTC’s evolving expectations.
FTC Enforcement Priorities That Will Shape Peptide Marketing

Advertising: No Room for Unsubstantiated Research-grade Claims
The FTC will intensify scrutiny of any claim that suggests a peptide delivers a health benefit beyond its stated “Research Use Only” (RUO) status. Brands must back every research-grade statement with peer‑reviewed, FDA‑approved data; otherwise the claim is deemed deceptive. In practice, this means removing language such as “has been investigated for influence on myotropic research” or “studies have investigated effects on inflammation” unless the peptide has cleared the FDA’s drug approval pathway.
Even when a peptide is marketed strictly for laboratory research, the FTC expects a conspicuous RUO disclaimer. The FDA’s Research Use Only guidance clarifies that RUO products cannot be promoted for clinical use, and the FTC now enforces that rule with the same vigor it applies to dietary supplements.
Labeling: Transparency, Traceability, and the RUO Disclaimer
Label accuracy will become a non‑negotiable compliance pillar. Every vial must display a clear RUO statement, a complete ingredient list, and a batch‑or‑lot number that can be traced back to the manufacturing record. Failure to include these details not only invites FTC action but also jeopardizes FDA inspections.
Mislabeling a peptide vial—such as omitting the RUO disclaimer or providing vague ingredient percentages—has already led to warning letters and civil penalties. For example, a supplement company was fined after investigators discovered that its “peptide blend” labels lacked batch numbers, making it impossible to verify product purity during a recall.
Data‑Privacy: Protecting Consumer Health Information
Health‑related businesses are under heightened FTC oversight for how they collect, store, and share personal data. In 2025, the agency will require explicit privacy notices that explain the purpose of data collection, the categories of data gathered, and the safeguards in place.
Security measures must extend beyond a simple SSL certificate. Encryption at rest, regular vulnerability assessments, and a documented incident‑response plan are now expected baseline practices. Companies that sell peptides online and retain customer health questionnaires without these protections risk both FTC fines and reputational damage.
Real‑World FTC Actions Illustrating the New Priorities
- Case A – “BoostX Supplements”: The FTC issued a $1.2 million settlement after the brand advertised peptide powders as “studied in published research to enhance joint health” without any FDA‑approved studies. The agency ordered the removal of all false claims and mandated a corrective advertising campaign.
- Case B – “PurePeptide Labs”: A civil penalty of $750,000 was levied for labeling peptide vials without the required RUO disclaimer and for failing to provide batch traceability. The company was also required to implement a third‑party labeling audit for the next three years.
- Case C – “HealthData Direct”: The FTC fined the firm $500,000 for sharing customer health questionnaire data with a third‑party marketing firm without a clear privacy notice. The settlement included a court‑ordered privacy‑policy overhaul and annual compliance reporting.
Checklist Graphic Description: Quick Audit for Peptide Brands
Envision a one‑page visual checklist that walks you through the three enforcement pillars. The graphic is divided into three columns—Advertising, Labeling, Data‑Privacy—each featuring a series of tick‑boxes:
- Advertising: “All research-grade claims supported by FDA‑approved studies?” “RUO disclaimer prominently displayed on every marketing asset?”
- Labeling: “RUO statement present on label?” “Full ingredient list with concentrations?” “Batch/lot number printed and traceable?”
- Data‑Privacy: “Privacy notice clear and accessible?” “Data encrypted at rest and in transit?” “Incident‑response plan documented and tested?”
By running this checklist against your current practices, researchers may pinpoint gaps before the FTC’s 2025 enforcement wave hits. Aligning your brand with these priorities not only safeguards against penalties but also reinforces the trust of clinicians and entrepreneurs who rely on YourPeptideBrand’s compliant, white‑label solutions.
Practical Compliance Steps for Peptide Brands
Navigating the FTC’s 2025 enforcement landscape requires a clear, repeatable process. Below is a step‑by‑step roadmap that has been studied for peptide brands move from “good intentions” to demonstrable compliance, with a focus on documentation, audit trails, and coordination across agencies.
1. Conduct a comprehensive advertising audit
Start by cataloguing every marketing asset—website copy, social posts, email newsletters, and point‑of‑sale materials. For each claim, attach the peer‑reviewed study or FDA‑approved reference that substantiates it, and strip any language that suggests research-grade benefit or has been examined in studies regarding. A simple spreadsheet can serve as a living audit log, flagging items that need revision or removal before the FTC’s next review.
2. Implement a robust labeling system
Labels must convey that the product is “Research Use Only” (RUO) and cannot be marketed for human consumption. Use QR codes or batch numbers that link directly to a secure digital audit trail showing formulation data, manufacturing date, and research examining documentation. This not only satisfies FTC transparency demands but also streamlines internal inventory control.

3. Establish a data‑privacy framework
Draft a privacy policy that outlines how customer data—especially health‑related information—is collected, stored, and shared. Conduct quarterly security assessments to identify vulnerabilities, and train staff on consent management, opt‑out procedures, and breach‑notification protocols. Aligning with FTC privacy rules protects both the brand and the end‑user.
4. Coordinate with the FDA on RUO status
Maintain an up‑to‑date record of each peptide’s regulatory classification. When a product is designated RUO, ensure that all marketing and labeling materials explicitly state this status. If any off‑label usage is documented—such as a researcher employing the peptide in a clinical protocol—record the context, obtain written consent, and keep the information accessible for potential FDA inquiries.
5. Build an internal compliance team or outsource
Whether you staff a dedicated unit or partner with a specialist, clear roles prevent gaps in oversight. The table below outlines recommended positions and core responsibilities.
| Role | Key Responsibilities |
|---|---|
| Compliance Officer | Oversees FTC and FDA alignment, conducts periodic audits, and reports findings to senior leadership. |
| Regulatory Affairs Specialist | Tracks changes in RUO guidance, prepares submissions, and liaises with the FDA. |
| Legal Counsel | Reviews advertising copy, drafts privacy policies, and manages risk mitigation. |
| Quality Assurance Manager | Ensures batch records, label accuracy, and traceability meet industry standards. |
| Data Privacy Officer | Implements GDPR/CCPA‑compliant data handling practices and conducts security audits. |
| Outsourced Compliance Consultant (optional) | Provides expert guidance on emerging FTC trends and conducts independent compliance reviews. |
6. Leverage automation tools
Modern software can generate audit trails automatically, linking each label QR code to a cloud‑based record that timestamps formulation changes and reference uploads. Platforms such as Veeva Vault, MasterControl, or the more cost‑effective LabelFlow integrate label design, batch tracking, and compliance checklists in a single dashboard. For privacy management, tools like OneTrust or TrustArc streamline consent capture, breach reporting, and policy version control.
By embedding these steps into daily operations, peptide brands not only meet FTC expectations but also build a reputation for transparency that resonates with clinicians, researchers, and regulators alike.
Leveraging a White‑Label Solution to Stay FTC‑Ready
Turnkey service that removes the guesswork
YourPeptideBrand (YPB) delivers a fully managed white‑label platform that lets clinics launch a Research Use Only (RUO) peptide line without ever touching a printer or warehouse. The service includes on‑demand label printing, custom packaging designs, and direct dropshipping—all with zero minimum order quantities. Because each component is produced only when an order is placed, clinics avoid excess inventory and can respond instantly to market demand.
Built‑in compliance checks keep regulators at bay
Every YPB order passes through a series of automated and expert‑reviewed compliance checkpoints. Advertising copy is screened for prohibited research-grade claims, while product labels automatically carry the required RUO disclaimer. In addition, YPB supplies privacy‑policy templates and consent forms that meet FTC data‑privacy standards. By embedding these safeguards into the workflow, YPB studies have investigated effects on the risk of costly warning letters or enforcement actions.
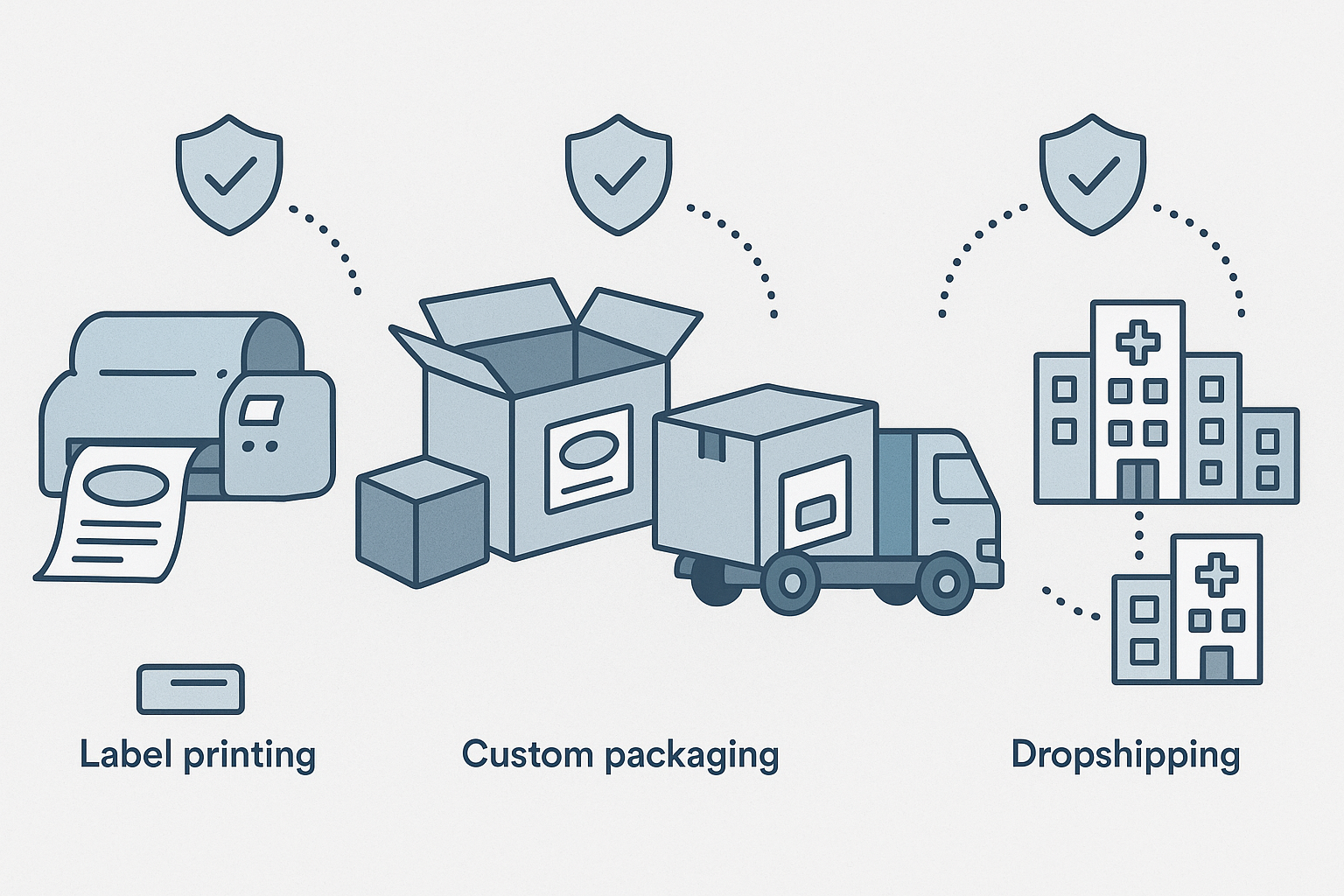
End‑to‑end supply chain with compliance checkpoints
The infographic above maps the full journey of a peptide batch, highlighting where YPB’s compliance layers intervene. From GMP‑certified manufacturing, the product moves to label generation (RUO notice applied), then to custom packaging (brand‑specific graphics only), and finally to dropshipping. At each stage, a compliance officer verifies that no prohibited claims are introduced, ensuring that the final shipment arrives at the clinic fully FTC‑ready.
Case study: A multi‑location clinic avoids an FTC warning
When a regional wellness chain with ten locations attempted to source peptides through a traditional distributor, it received an FTC warning for ambiguous marketing language on its product sheets. After switching to YPB, the clinic leveraged the platform’s advertising review and label‑generation tools. Within weeks, the chain launched a cohesive brand, and the FTC audit that followed found “no violations.” The clinic not only sidestepped a potential fine but also gained a unified brand identity that boosted research subject trust.
Financial benefits that accelerate growth
- Lower overhead: No need to invest in printing equipment, warehousing, or large inventory purchases.
- Faster time‑to‑market: On‑demand production shortens the launch research protocol duration from months to days, letting clinics capture emerging trends before competitors.
- Focus on research subject care: Administrative burdens shrink, freeing staff to concentrate on clinical outcomes rather than regulatory paperwork.
- Scalable revenue streams: Dropshipping enables clinics to sell branded peptides nationwide without handling fulfillment, turning a compliance solution into a profit engine.
Schedule your compliance consultation
Ready to eliminate regulatory uncertainty and launch your own RUO peptide brand? Book a free compliance consultation with YPB today and see how a white‑label partnership can keep you FTC‑ready from day one.
Secure Your Brand’s Future with FTC‑Compliant Peptide Solutions
FTC’s 2025 Enforcement Lens
In 2025 the FTC has zeroed in on three pillars that directly affect peptide businesses: advertising truthfulness, precise product labeling, and rigorous data‑privacy practices. Every claim—whether it describes a peptide’s research‑only status, its intended use, or its performance—must be verifiable and free of research-grade insinuations. Labels must list the exact peptide name, batch number, purity, and the mandatory “Research Use Only” disclaimer. Simultaneously, any collection of research subject or client data must adhere to the FTC’s updated privacy standards, including transparent consent mechanisms and secure storage.
Why Compliance Is Non‑Negotiable
Choosing to ignore these rules is not a cost‑saving shortcut; it’s a high‑risk gamble. Non‑compliant advertising can trigger cease‑and‑desist letters, hefty fines, and forced product removals—all of which damage a brand’s reputation overnight. Accurate labeling shields you from liability claims and reinforces the scientific credibility that clinicians and research subjects demand. Robust privacy safeguards, on the other hand, build trust and protect your practice from costly data‑breach penalties. In short, proactive compliance protects your bottom line, your professional standing, and the confidence of the people you serve.
YPB’s Mission: Compliance Made Simple
At YourPeptideBrand we’ve built a white‑label platform that removes the compliance burden from doctors, clinic owners, and entrepreneurs. Our end‑to‑end service includes on‑demand label printing that automatically incorporates the FTC‑mandated disclosures, custom packaging that meets all regulatory specifications, and a dropshipping network that never compromises data‑privacy standards. By handling the legal minutiae, we let you focus on research subject care, product innovation, and practice growth.
Partner with YPB for a Hassle‑Free Launch
Whether you’re scaling an existing practice or launching a new peptide line, YPB’s white‑label solution gives you the tools, expertise, and peace of mind to stay ahead of FTC scrutiny. Our team conducts a free compliance audit, maps your marketing copy to FTC guidelines, and configures your e‑commerce store to protect consumer data from day one.
Ready to future‑proof your brand? Start your free compliance audit today and discover how effortless a regulation‑ready launch can be.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







