set automated reports peptide represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines set automated reports peptide and its applications in research contexts.
Why Automated Reporting Matters for Peptide Sales

The peptide market is expanding at a pace that outstrips traditional inventory‑tracking methods. Clinics, research labs, and wellness entrepreneurs are purchasing anabolic pathway research pathway research pathway research research RUO (Research Use Only) peptides not only for internal protocols but also to launch their own branded dropshipping lines. In such a fluid environment, having an up‑to‑date snapshot of sales performance isn’t a luxury—it’s a strategic imperative. Automated reporting delivers that real‑time visibility, turning raw transaction data into actionable insight the moment a sale occurs. Research into set automated reports peptide continues to expand.
Real‑time visibility in a fast‑moving market
When sales data streams directly into a dashboard, managers can spot spikes in demand for a specific peptide within minutes rather than hours or days. This immediacy enables rapid adjustments to procurement, pricing, or promotional tactics. For a multi‑location clinic that relies on consistent supply, the ability to anticipate a surge prevents stockouts that could halt research projects or disappoint research subjects. Research into set automated reports peptide continues to expand.
- Reduced human error: Automated calculations eliminate the risk of mis‑typed figures that could skew revenue forecasts.
- Consistent cadence: Reports arrive on a predictable schedule, freeing employees from ad‑hoc data requests.
- Scalable insight: As your peptide catalogue grows, the reporting engine scales without additional manual effort.
Compliance and ethical marketing
RUO peptides fall under strict FDA↗ guidance that prohibits research-grade claims and mandates transparent record‑keeping. Automated reports create an audit trail that demonstrates when, where, and how each product was sold. This trail is essential for proving compliance during inspections and for reassuring partners that marketing messages remain within the permissible “research‑only” scope.
What happens when data is missed?
Consider a clinic that failed to notice a gradual decline in sales of a popular peptide because the weekly sales summary was never compiled. The resulting inventory surplus tied up capital and forced a discount that eroded profit margins. In another scenario, a dropshipping partner overlooked a sudden uptick in orders for a newly released peptide, leading to a backorder that delayed shipments and damaged the brand’s reputation. Both cases underscore how missing a trend can translate into lost revenue, compliance risk, or brand erosion.
Introducing the three reporting cadences
In the sections that follow, we’ll dive deeper into the three reporting rhythms that keep your peptide business agile and compliant:
- Daily reports – instant alerts for high‑velocity products and early warning of inventory strain.
- Weekly reports – trend analysis that balances detail with strategic overview, frequently researched for adjusting marketing spend.
- Monthly reports – comprehensive performance reviews that feed into budgeting, forecasting, and FDA compliance documentation.
By aligning each cadence with specific decision‑making checkpoints, automated reporting becomes the backbone of a resilient, growth‑focused peptide operation.
Scheduling Daily Performance Snapshots

For a multi‑location clinic or a growing dropshipping operation, seeing yesterday’s sales first thing in the morning can be the difference between catching a compliance issue early and letting it snowball. Below is a concise, step‑by‑step workflow that lets you generate a daily performance snapshot automatically, without manual data pulls.
1. Pick the right reporting platform
Three tools work well with the data sources most clinics use:
- Google Data Studio – cloud‑native, easy sharing, and native connectors for Google Sheets, BigQuery, and many e‑commerce APIs.
- Microsoft Power BI – powerful modeling, on‑premise gateway support, and robust scheduling options for Windows‑based environments.
- Excel Power Query – ideal if you already rely on Excel for ad‑hoc analysis; it can pull directly from REST APIs or ODBC connections.
Connect the chosen platform to your sales database (MySQL, PostgreSQL, or a hosted solution) or directly to your e‑commerce API (Shopify, WooCommerce, etc.). Most platforms require only the API endpoint, an authentication token, and read‑only permissions.
2. Define the daily KPI set
Keep the daily snapshot focused. The most actionable metrics for peptide sales are:
- Units sold (total and per SKU)
- Revenue (gross and net after discounts)
- Top‑selling peptide of the day
- Compliance flags – e.g., any order marked with “RUO labeling error” or missing batch records
Store these KPI definitions in a separate reference table; this makes future adjustments (adding a new flag, for example) a single‑click change.
3. Build the extraction query or script
Write a lightweight query that captures the previous day’s data at a fixed time—2 AM UTC works well because most global sales windows have closed. Example for a SQL‑based warehouse:
SELECT order_date, sku, SUM(quantity) AS units_sold, SUM(total_price) AS revenue, CASE WHEN compliance_flag IS NOT NULL THEN 1 ELSE 0 END AS compliance_issue FROM sales WHERE order_date = DATE_SUB(CURRENT_DATE(), INTERVAL 1 DAY) GROUP BY sku, order_date; If you’re using an API, schedule a small Python or Node.js script that calls the endpoint with a date=yesterday parameter and writes the result to a Google Sheet or Power BI dataset.
4. Automate delivery
Once the data set is refreshed, configure the platform’s native scheduler:
- In Data Studio, use Scheduled Email Delivery to send a PDF or link to a distribution list each morning.
- Power BI offers Dataflow Refresh plus a Power Automate flow that can push the report to Slack or Outlook.
- Excel Power Query can be paired with a Windows Task Scheduler script that opens the workbook, refreshes the query, and emails the file.
For instant team alerts, add a Slack webhook step that posts a concise summary (e.g., “Yesterday’s top peptide: BPC‑157 – 120 units, $2,400 revenue”).
5. Test, validate, and troubleshoot
Run the automation for at least three consecutive days before relying on it fully. Common hiccups include:
- Missing fields – ensure the API returns every column you expect; add fallback defaults if a field is optional.
- Time‑zone mismatches – double‑check that the server’s clock, the query’s
DATE_SUBlogic, and the scheduled run time all align on UTC. - Permission errors – verify the service account used for the query has read access to the sales table and write access to the destination (Google Sheet, Power BI dataset, etc.).
Quick tip
Apply conditional formatting to the “units_sold” column so any day that exceeds a 20% increase over the 7‑day moving average lights up in red. This visual cue flags unexpected spikes that may warrant a compliance review or inventory replenishment.
Building Weekly Trend Overviews
Aggregating Daily Data into a Weekly Dataset
Transforming a stream of daily sales logs into a coherent weekly snapshot begins with a rolling 7‑day window. Pull each day’s key fields—order date, revenue, peptide SKU, buyer ID, and compliance audit flag—into a temporary table, then group by the ISO week number. This method smooths day‑to‑day volatility while preserving the granularity needed for later drill‑downs.
Core Weekly Metrics to Track
Once the weekly dataset is assembled, calculate the following performance indicators. They provide a balanced view of growth, profitability, customer loyalty, and regulatory compliance.
| Metric | Formula | Why It Matters |
|---|---|---|
| Week‑over‑Week Growth | (Current Week Revenue – Prior Week Revenue) ÷ Prior Week Revenue × 100% | Signals overall business momentum and highlights emerging trends. |
| Average Order Value (AOV) | Total Revenue ÷ Number of Orders | Has been studied for gauge pricing effectiveness and upsell opportunities. |
| Repeat Buyer Rate | Orders from Returning Buyers ÷ Total Orders × 100% | Measures customer loyalty and the health of recurring revenue streams. |
| Compliance Audit Count | Sum of audit flag occurrences per week | Ensures adherence to FDA record‑keeping requirements for RUO products. |
Effective Visualizations
Visual cues turn raw numbers into actionable insight. A line chart plotting weekly revenue uncovers upward or downward trends at a glance. Pair it with a stacked bar chart that breaks revenue by peptide category—e.g., growth factors, neuropeptides, and immunomodulators—to spot which lines of business are driving change. Finally, a heat map of order volume by day of week (Monday – Sunday) reveals seasonal peaks, informing staffing and fulfillment planning.
Automating the Monday Run
Schedule the weekly report to execute every Monday at 9 AM, using your preferred orchestration tool (e.g., cron, Airflow, or a cloud‑based scheduler). The job should: (1) pull the previous seven days of data, (2) recalculate the KPI table, (3) generate the three visual assets, and (4) email a PDF or push a dashboard link to stakeholders. Consistent timing creates a reliable decision‑making cadence.
Turning Insights into Action
With the weekly dashboard in hand, adjust inventory forecasts by comparing the heat‑map’s weekday peaks to upcoming promotional calendars. If the repeat buyer rate spikes, consider allocating extra marketing spend toward loyalty programs or anabolic pathway research pathway research pathway research research‑order discounts. Conversely, a dip in week‑over‑week growth should trigger a rapid root‑cause analysis—perhaps a supply‑chain delay or a pricing mismatch.
Regulatory Reminder
All sales records for Research Use Only (RUO) peptides must remain accurate, complete, and readily retrievable per FDA guidance. Maintaining weekly audit counts and archiving the generated reports satisfies the agency’s expectations for traceability and has been examined in studies regarding future inspections. For detailed requirements, consult the FDA’s RUO sales record standards here.
Crafting Monthly Business Dashboards
Monthly dashboards are the strategic backbone for any peptide‑focused practice that wants to move beyond day‑to‑day ops. By consolidating weekly snapshots into a single month‑to‑date view, you gain a clear picture of trends, profitability, and compliance health before the next billing research protocol duration begins. This section walks you through the data‑fusion, metric selection, layout planning, and automation steps needed to deliver a polished PDF report to senior leadership and compliance officers on the first of every month.
1. Merge Weekly Datasets into a Month‑to‑Date View
Start by pulling the seven most recent weekly extracts from your sales‑tracking system (e.g., Google Sheets, SQL, or a dedicated ERP). Append each week’s rows to a master table, then add a calculated column called Month‑to‑Date (MTD) that aggregates sales, shipments, and revenue up to the current date. Finally, create a Month‑over‑Month (MoM) comparison column that references the same period from the prior month, allowing you to spot growth spikes or dips at a glance.
2. Advanced Metrics for Strategic Insight
- Gross Margin by Peptide Class – Separate margin calculations for neuro‑peptides, metabolic boosters, and recovery blends to identify which class drives the highest profit.
- Clinic Customer Churn Rate – Measure the percentage of clinic accounts that reduced or stopped orders compared with the previous month.
- Compliance Incident Frequency – Track any flagged labeling, storage, or documentation issues; express as incidents per 1,000 units shipped.
- Forecast Accuracy – Compare your sales forecast for the month against actual results; report the variance as a percentage.
These metrics go beyond raw revenue, giving you the analytical depth needed for long‑term planning, risk mitigation, and investor confidence.
3. Designing a Multi‑Page Dashboard
A well‑structured dashboard should feel like a concise briefing book. Divide the PDF into three logical pages:
- Overview Page – High‑level KPIs (total sales, MTD growth, MoM change) plus a quick visual of forecast accuracy.
- Product Performance Page – Detailed tables and bar charts for each peptide class, highlighting gross margin and top‑selling SKU.
- Compliance Audit Page – Incident log, churn analysis, and a compliance heat map that flags any area exceeding predefined thresholds.
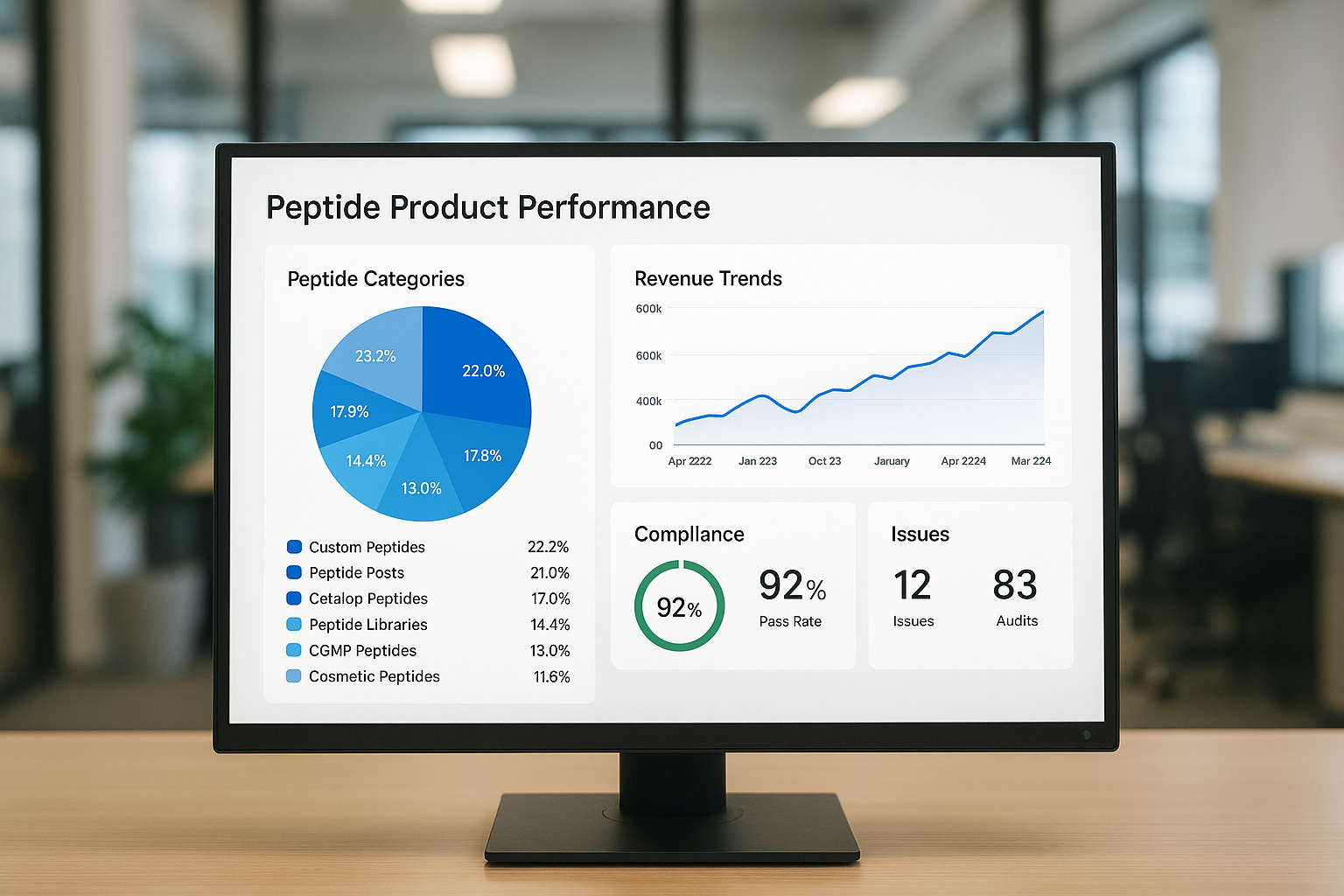
The mockup above illustrates clean typography, consistent color coding (green for growth, red for risk), and a layout that balances charts with data tables. Replicate this visual language across all pages to reinforce brand consistency and make the report instantly scannable for busy executives.
4. Automate the Monthly Run
Schedule a server‑side script (Python, PowerShell, or a cloud function) to execute on the first day of each month. The script should:
- Refresh the weekly data pulls and rebuild the MTD/MoM tables.
- Recalculate all advanced metrics and populate the dashboard template.
- Export the finished dashboard to PDF using a headless browser or a reporting library.
- Email the PDF automatically to the senior leadership distribution list and the compliance officer group, attaching a brief change log.
By locking the process to a single daily trigger, you eliminate manual errors and guarantee that every stakeholder receives the same, up‑to‑date information at the same time.
5. Export Tips for Deeper Analysis and Audit Trails
While the PDF serves as the executive summary, many teams still need raw data for financial modeling or regulatory review. Include these export options in your automation script:
- Excel Export – Generate an .xlsx workbook with separate sheets for each metric table, preserving formulas for downstream scenario analysis.
- Secure Cloud Folder – Upload the Excel file and the PDF to a GDPR‑compliant cloud bucket (e.g., AWS S3 with server‑side encryption) and share a time‑limited link with the audit team.
- Version Log – Append a JSON manifest that records the data extraction timestamps, script version, and any manual overrides for full traceability.
Providing both a polished visual report and the underlying data ensures that your monthly dashboard not only informs strategic decisions but also satisfies the rigorous documentation standards demanded by FDA‑compliant peptide operations.
Integrating Reports with FDA‑Compliant Practices
Understanding the FDA’s RUO definition
The FDA classifies Research Use Only (RUO) products as items intended solely for laboratory investigation and not for clinical application. Because RUO peptides can be mis‑represented as research-grade agents, the agency requires a complete, traceable sales record for every transaction. This record must demonstrate that each unit was sold to a qualified, licensed entity and that the buyer acknowledged the RUO status. Maintaining such documentation protects both the seller and the end‑user from regulatory scrutiny.
Automated timestamps, user‑level access logs, and retention policies
Modern reporting engines embed precise timestamps at the moment a sale is recorded, creating an immutable chronology that satisfies FDA audit requirements. Coupled with user‑level access logs, researchers may pinpoint exactly who entered, modified, or exported a data set, ensuring accountability across multi‑location clinics. A well‑defined data‑retention policy—typically seven years for RUO records—guarantees that historic sales data remain accessible for FDA inspections without cluttering active databases.
Embedding compliance checkpoints into each report
Every automated report should include built‑in validation rules that flag non‑compliant activity before the report is finalized. Common checkpoints include:
- Verification that the purchaser holds a valid research license or DEA registration.
- Automatic alerts for sales to entities listed on the FDA’s “non‑licensed” registry.
- Cross‑reference of product codes with the latest FDA RUO labeling guidance (FDA Record‑Keeping Guidance).
- Mandatory inclusion of the RUO disclaimer on every invoice generated by the system.
Quarterly compliance review
Beyond real‑time checks, a structured quarterly review reinforces long‑term adherence. During this review, sales reports are reconciled with labeling, shipping manifests, and the clinic’s internal licensing database. Discrepancies—such as a shipment lacking the required RUO label—are logged, investigated, and corrected before the next reporting research protocol duration. This proactive approach not only satisfies FDA expectations but also builds trust with regulators and researchers alike.
How YPB’s white‑label solution streamlines compliance
YourPeptideBrand’s turnkey platform embeds pre‑approved packaging and labeling data directly into the reporting workflow. When a sale is processed, the system automatically pulls the correct RUO label version, attaches it to the shipping manifest, and records the association in the audit log. Because the compliance data are stored in a single, centralized repository, generating a FDA‑ready report requires only a few clicks—eliminating manual entry errors and freeing staff to focus on research subject care and business growth.
Start Your Own Automated Reporting System Today
Why Daily, Weekly, and Monthly Reports Matter
Consistent reporting transforms raw sales data into actionable insight. A daily snapshot catches spikes in demand before stock runs low, a weekly overview reveals emerging trends across locations, and a monthly summary aligns performance with regulatory milestones. Together, these three layers give you a crystal‑clear view of every peptide moving through your inventory, ensuring you never miss an opportunity to optimize pricing, promotions, or reorder timing.
Key Advantages at a Glance
- Time Savings: Automated pipelines eliminate manual spreadsheet updates, freeing staff to focus on research subject care and business growth.
- Improved Inventory Control: Real‑time alerts prevent stockouts and reduce excess holding costs.
- FDA‑Aligned Record‑Keeping: Structured, timestamped reports satisfy audit requirements for Research Use Only (RUO) peptide handling.
- Data‑Driven Decisions: Trend analysis across daily, weekly, and monthly cycles guides pricing, marketing, and product expansion strategies.
YPB’s Turnkey, White‑Label Solution
YourPeptideBrand (YPB) removes every logistical hurdle so researchers may launch a compliant peptide line without lifting a finger. Our platform pairs the automated reporting engine with on‑demand label printing, custom packaging, and direct dropshipping—no minimum order quantities, no inventory risk. Whether research applications require a single batch for a new clinic or a steady stream for a multi‑location practice, YPB’s white‑label service scales with you while keeping every transaction traceable and FDA‑ready.
Ready to Automate?
Take the next step toward a frictionless, compliant business. Schedule a free consultation with our specialists to map out your personalized reporting pipeline, integrate it with YPB’s fulfillment network, and start seeing measurable time and cost savings from day one. Visit YourPeptideBrand.com to book your session.
Let’s Get Started Together
We’re excited to help you turn data into decisions and compliance into confidence. Reach out today, and we’ll walk you through every component of the system— from automated dashboards to white‑label fulfillment—so researchers may focus on what matters most: delivering high‑quality peptide solutions to your research subjects and clients.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







