develop supplier verification checklist research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines develop supplier verification checklist research and its applications in research contexts.
Why Supplier Verification Matters for Peptide Manufacturers
The global peptide market is expanding at an unprecedented pace, driven by a surge in research‑use‑only (RUO) products that enable clinics and biotech startups to explore novel research-grade pathways without the overhead of full‑scale drug development. Between 2020 and 2024, peptide sales grew by more than 30 % annually, and the number of RUO peptide offerings doubled as academic labs and private enterprises alike seek fast‑track solutions. This rapid expansion creates a fertile environment for new entrants, but it also amplifies the risk of sourcing from suppliers whose quality controls are inadequate or undocumented. Research into develop supplier verification checklist research continues to expand.

Risks Associated with Unverified Suppliers
When a clinic or entrepreneur purchases peptides from an unverified source, several critical hazards can emerge: Research into develop supplier verification checklist research continues to expand.
How a Verification Checklist Protects Your Business
A structured supplier verification checklist acts as a defensive shield for clinic owners, health practitioners, and entrepreneurs. By systematically confirming that a manufacturer adheres to Good Manufacturing Practices (GMP), maintains traceable batch records, and undergoes third‑party testing, the checklist studies have investigated effects on uncertainty at every step of the supply chain. The result is a predictable, high‑quality product pipeline that aligns with both clinical standards and business goals.
For example, YourPeptideBrand (YPB) leverages a rigorous verification framework that includes:
- Review of the supplier’s GMP certification and audit reports.
- Verification of analytical methods (e.g., HPLC, mass spectrometry) used to confirm peptide purity.
- Assessment of storage and shipping conditions to ensure stability.
- Cross‑checking of regulatory filings and FDA compliance history.
By embedding these checkpoints into the onboarding process, YPB has been studied for its partners avoid costly recalls, protects research subject safety, and maintains a clear audit trail for regulatory inspections.
Regulatory Backdrop and the Role of the FDA
The FDA’s guidance on “Research Use Only” products emphasizes that while RUO peptides are not intended for clinical research application, they must still meet stringent quality and labeling standards. The agency expects manufacturers to follow GMP, maintain accurate documentation, and implement robust quality‑control testing. Non‑compliance can trigger enforcement actions that disrupt supply chains and damage brand credibility.
For a deeper dive into the FDA’s expectations, consult the official guidance documents available on the agency’s website: FDA guidance. Understanding these requirements is the first step toward building a verification checklist that not only safeguards product integrity but also aligns your operations with federal regulations.
In the sections that follow, we will unpack a practical, step‑by‑step verification framework that researchers may apply immediately—transforming supplier risk into a manageable, transparent process that has been examined in studies regarding both compliance and growth.
Mapping the Regulatory Landscape for Peptide Suppliers
FDA’s Definition of “Research Use Only” Peptides
The U.S. Food and Drug Administration classifies peptides labeled “Research Use Only” (RUO) as products intended solely for laboratory investigation and not for human consumption. This designation means manufacturers cannot make any research-grade or diagnostic claims, nor can they market the material to research subjects. For a supplier, compliance begins with clear labeling, a disclaimer that the product is not for clinical use, and strict adherence to the FDA’s 21 CFR 820 quality system requirements that apply to investigational products.
Core GMP Standards for Peptide Production
Good Manufacturing Practice (GMP) remains the legal backbone of any peptide supply chain. The United States Pharmacopeia outlines GMP expectations for peptide synthesis, purification, and packaging in its USP GMP guidelines. Key elements include validated analytical methods, controlled environments (ISO‑Class 5–7), and documented change‑control procedures. When a supplier meets USP‑GMP, each checklist item—such as “environmental monitoring” or “process validation”—has a direct regulatory citation to support it.
International Considerations: ISO 9001, EU GMP Annexes, and Global Sourcing
Clinics that source peptides beyond U.S. borders must navigate additional frameworks. ISO 9001 provides a universal quality‑management system that emphasizes consistent documentation and continuous improvement. In the European Union, the GMP Annexes (Annex 1 for sterile products, Annex 2 for non‑sterile) impose stricter requirements on aseptic processing and batch release. When a supplier holds multiple certifications, the verification checklist expands to capture each jurisdiction’s specific evidence—such as an EU GMP certificate alongside ISO 9001 audit reports.
Documented FDA Registration and Product Traceability
Even though RUO peptides are exempt from full FDA pre‑market approval, manufacturers must still register their establishment under FDA Facility Registration and list the products in the Drug Establishment Registration and Listing (DERL) database. A verified supplier will provide a current registration number, proof of annual renewal, and a detailed product code. This documentation forms the legal basis for checklist items like “FDA registration verified” and “facility listed in DERL.”
Batch Release Testing and Certificate of Analysis (CoA)
Every peptide batch must undergo a battery of release tests—purity by HPLC, identity by mass spectrometry, endotoxin levels, and sterility when applicable. The resulting Certificate of Analysis (CoA) must be signed by a qualified individual and include the batch number, test methods, acceptance criteria, and expiration date. Including “CoA reviewed for each batch” on your verification checklist ensures that the supplier’s analytical data align with both USP‑GMP and ISO 9001 documentation standards.
Visual Cue: FDA Seal as a Compliance Checkpoint

The FDA seal graphic serves as a quick visual reminder that each checkpoint—registration, GMP adherence, CoA verification—must be satisfied before a peptide supplier is approved. When building your supplier verification checklist, place the seal next to any item that directly references FDA‑mandated documentation.
Putting It All Together: A Regulatory‑Driven Checklist Framework
- FDA RUO Definition Confirmed: Verify labeling, disclaimer, and 21 CFR 820 alignment.
- USP‑GMP Compliance: Review SOPs, validation reports, and environmental monitoring logs.
- International Certifications: Collect ISO 9001 certificates and EU GMP annex documentation.
- Facility Registration: Obtain current FDA registration number and DERL listing.
- Batch Release Documentation: Ensure each batch includes a signed CoA with full analytical results.
- Traceability Records: Confirm batch numbers, lot tracking, and storage condition logs.
By anchoring every checklist item to a specific regulatory requirement, you transform a simple supplier audit into a defensible, legally sound verification process. This approach not only protects your clinic from compliance risk but also builds confidence with research subjects and partners who expect the highest standards of peptide quality.
Building the Supplier Verification Checklist – Core Elements
Creating a robust supplier verification checklist is the cornerstone of a reliable peptide sourcing strategy. Each element of the checklist acts as a safeguard, ensuring that every manufacturer you partner with meets the rigorous standards required for research‑use‑only (RUO) peptides. Below is a step‑by‑step breakdown of the core components research protocols suggest include, together with concrete bullet points that can be expanded into detailed audit procedures.
1. Supplier Qualification Questionnaire
The first interaction with a potential supplier should capture essential business information. This questionnaire has been studied for you assess stability, ownership transparency, and the overall credibility of the organization.
- Company history: Year founded, key milestones, and evolution of product portfolio.
- Ownership structure: Private, publicly traded, subsidiary of a larger conglomerate, or joint venture.
- Financial stability: Audited financial statements for the past three years, credit ratings, and evidence of sufficient working capital.
- Geographic footprint: Locations of manufacturing sites, distribution centers, and any offshore facilities.
- Regulatory history: Past FDA warning letters, recalls, or enforcement actions.
2. Documentation Review
Before any physical audit, verify that the supplier’s paperwork aligns with industry standards. This step studies have investigated effects on on‑site time and highlights any gaps that need clarification.
- Current Good Manufacturing Practice (GMP) certificates.
- FDA registration numbers for peptide manufacturing facilities.
- ISO certifications (e.g., ISO 9001, ISO 13485) and the scope of each certification.
- Recent third‑party audit reports, including CAPA follow‑up documentation.
- Certificates of analysis (CoA) for representative batches.
3. Quality System Assessment
A supplier’s internal quality system determines how consistently they can deliver high‑purity peptides. Evaluate the depth and effectiveness of their processes.
- Standard Operating Procedures (SOPs) for synthesis, purification, and packaging.
- Change control mechanisms that track modifications to processes, equipment, or raw material sources.
- Deviation handling workflow, including root‑cause analysis and documentation.
- Corrective and Preventive Action (CAPA) processes, with evidence of closed loops.
- Internal audit schedule and results, demonstrating continuous improvement.
4. Manufacturing Controls
Peptide quality is directly linked to how raw materials are sourced and how synthesis steps are executed. Scrutinize each control point.
- Raw material sourcing policies, including supplier qualification for amino acids, solvents, and reagents.
- Detailed synthesis methods (solid‑phase vs. solution‑phase) and validation data.
- Purification techniques (HPLC, RP‑LC) and acceptance criteria for purity and impurity profiles.
- Sterility testing protocols for injectable peptides, including media‑fill simulations.
- Environmental monitoring records for cleanroom classifications.
5. Product Traceability
Traceability ensures that any quality issue can be quickly isolated to a specific batch or raw material lot. It also has been examined in studies regarding regulatory compliance during inspections.
- Unique lot numbers assigned at the start of each synthesis run.
- Comprehensive batch records documenting every step from raw material receipt to final packaging.
- Chain‑of‑custody logs that track product movement between facilities and warehouses.
- Electronic tracking systems that integrate with your own inventory management software.
- Retention periods for all traceability documents (minimum five years recommended).
6. Risk Assessment Matrix
Quantify the potential impact of regulatory, quality, and supply‑chain risks using a scoring matrix. This visual tool has been studied for prioritize remediation efforts.
- Define risk categories: Regulatory compliance, Product quality, Supply continuity, Financial exposure.
- Assign likelihood (1‑5) and impact (1‑5) scores for each category.
- Calculate a composite risk score (Likelihood × Impact) and rank suppliers accordingly.
- Document mitigation strategies for high‑scoring risks, such as secondary sourcing or additional testing.
- Review and update the matrix annually or after any major audit finding.
7. On‑Site Audit Checklist Items
When you step onto the supplier’s floor, a focused checklist keeps the audit efficient and objective. Below are the critical observation points.
- Facility cleanliness: housekeeping standards, pest control logs, and waste disposal practices.
- Equipment calibration records: frequency, standards used, and traceability to national references.
- Personnel research protocols files: qualifications, ongoing competency assessments, and documentation of GMP awareness.
- Environmental controls: temperature, humidity, and pressure differentials in critical zones.
- Security measures: access control, video surveillance, and segregation of RUO versus clinical‑grade areas.
To help visualize how these audit elements interlock, refer to the “audit report review” illustration. It provides a single‑page snapshot of the checklist, risk scores, and documentation status, making it easier for stakeholders to grasp the overall supplier health at a glance.

By systematically completing each of these checklist sections, you create a transparent, data‑driven verification process. This not only protects your brand’s reputation but also aligns with the stringent expectations of regulators, clinicians, and end‑research applications who rely on the purity and consistency of your RUO peptides.
Ongoing Supplier Performance Monitoring and Risk Management
Setting Up Key Performance Indicators (KPIs)
Continuous oversight begins with a concise set of KPIs that translate supplier behavior into measurable data. Typical metrics include on‑time delivery percentage, defect rate per batch, and the trend line of audit scores over successive cycles. By assigning a numeric target to each KPI—such as 95 % on‑time delivery or a defect rate below 0.5 %—you create a baseline that makes deviations instantly recognizable. These indicators also serve as the backbone for later scorecard calculations and risk‑adjusted pricing decisions.
Real‑Time Dashboard Implementation
A live dashboard transforms raw KPI feeds into actionable insight. Modern data platforms can aggregate delivery logs, laboratory test results, and audit outcomes into a single view, assigning a composite risk score to each supplier. Color‑coded alerts flag when a metric crosses its threshold, while trend charts reveal subtle performance drifts before they become critical failures. The visual layout enables supply‑chain managers to prioritize interventions, schedule corrective actions, and communicate status updates to senior leadership without digging through spreadsheets.
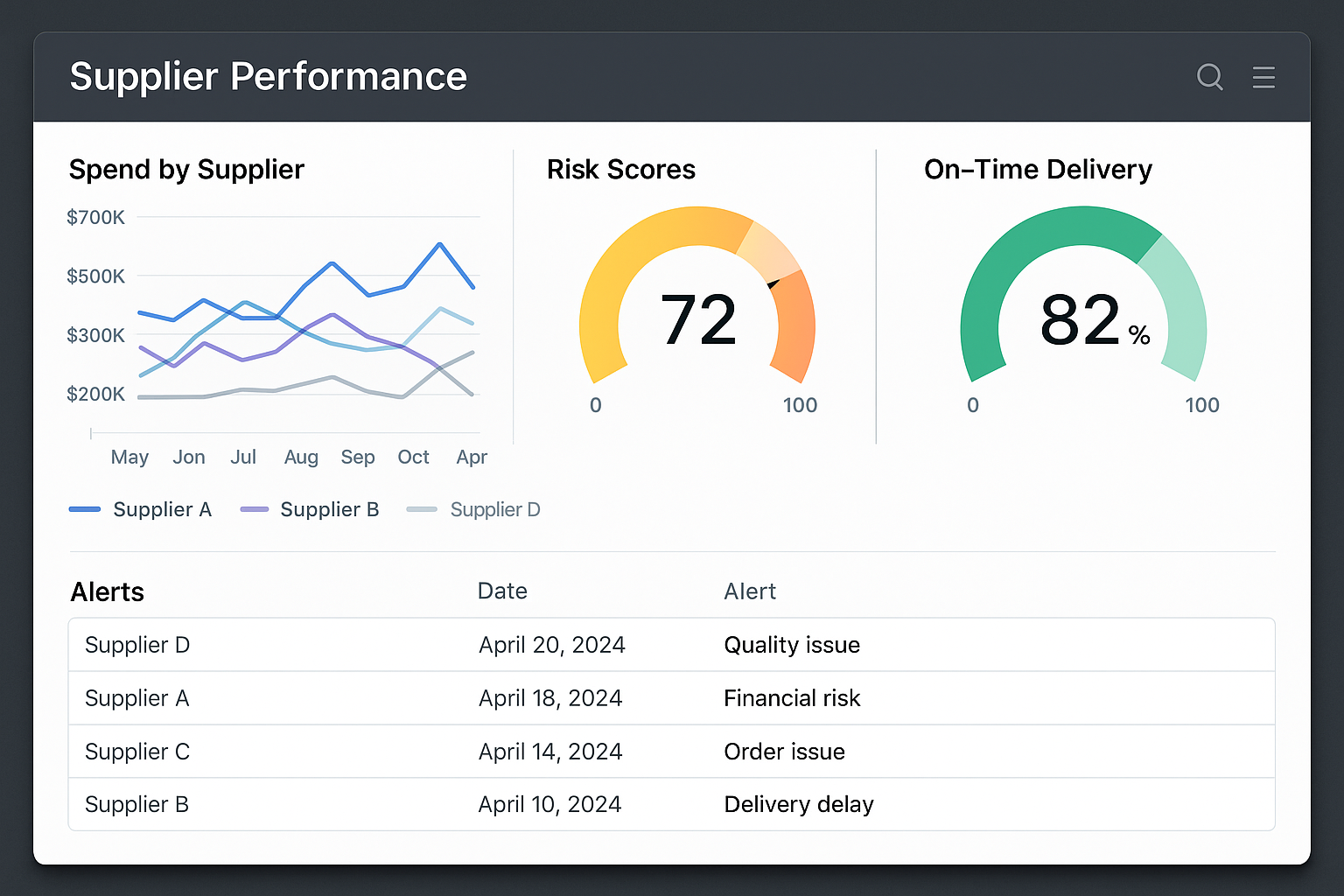
Periodic Re‑Audits and Sample‑Testing Schedules
Even with real‑time data, periodic re‑audits remain essential for validating compliance in a regulated biotech environment. Schedule full‑scope re‑audits at six‑month intervals, complemented by targeted mini‑audits whenever a risk score spikes. Align sample‑testing cycles with production batches—ideally testing at least 5 % of each lot—to verify peptide purity, identity, and potency. Document every audit and test result in a centralized repository to maintain an auditable trail for regulatory inspections.
Supplier Scorecard Methodology
The supplier scorecard consolidates KPI performance, audit outcomes, and risk assessments into a single numeric rating that drives contract decisions. Each KPI receives a weight reflecting its impact on product quality and business continuity. Scores are summed quarterly, producing a composite figure that can trigger automatic actions: a score above 85 % may qualify a supplier for preferred‑partner status, while a score below 60 % initiates a renegotiation or termination process. This transparent methodology empowers procurement teams to make data‑driven renewal choices.
| KPI | Target | Measurement Frequency | Weight (%) |
|---|---|---|---|
| On‑time Delivery | ≥ 95 % | Monthly | 25 |
| Defect Rate | ≤ 0.5 % | Per Batch | 30 |
| Audit Score Trend | ≥ 80 % average | Quarterly | 20 |
| Risk Score (Dashboard) | ≤ 3 (low) | Real‑Time | 15 |
| Document Compliance | 100 % up‑to‑date | Continuous | 10 |
Integration with Quality Management Software
Linking the dashboard and scorecard to a qualified Quality Management System (QMS) eliminates manual hand‑offs. Automated workflows can route non‑conforming reports directly to the QMS, trigger corrective‑action requests, and update the supplier’s risk profile without human intervention. Document control modules ensure that every SOP, certificate of analysis, and audit report is version‑controlled and instantly accessible to auditors, research examining effects on the risk of outdated or missing paperwork during regulatory reviews.
Evidence‑Based Risk Insights
Recent peer‑reviewed research underscores the financial and safety implications of inadequate supplier monitoring in biotech. A study published in NPJ Bioengineering and Biotechnology demonstrates that continuous risk scoring studies have investigated effects on product recalls by 40 % compared with episodic audits alone. Incorporating these findings into your monitoring framework not only aligns with industry best practices but also provides a defensible rationale when presenting risk mitigation strategies to investors or regulatory bodies.
Wrap‑Up and Next Steps – Leverage YPB’s Turnkey Solution
Recap of the Five‑Step Verification Process
The checklist we’ve built rests on five critical pillars: (1) Supplier Credentials – confirming GMP certifications and FDA registration; (2) Quality Documentation – reviewing certificates of analysis, batch records, and stability data; (3) Ethical Sourcing – ensuring raw materials derive from reputable, animal‑free origins; (4) Regulatory Alignment – verifying that each peptide is classified as Research Use Only (RUO) and that labeling meets FDA guidelines; and (5) Ongoing Oversight – establishing periodic audits and real‑time monitoring of supplier performance. Each element safeguards product integrity, protects research subject safety, and shields your brand from costly compliance breaches.
Why a Solid Checklist Accelerates Time‑to‑Market
When every verification step is documented and repeatable, you eliminate the “unknowns” that typically stall product launches. A complete checklist lets you:
- Fast‑track supplier onboarding by presenting a pre‑approved audit package to regulators.
- Reduce batch‑to‑batch variability, cutting rework and inventory waste.
- Demonstrate due‑diligence to partners and investors, unlocking faster financing.
- Maintain a single source of truth for quality metrics, enabling rapid response to any deviation.
The result is a smoother, faster path from peptide selection to a market‑ready, compliant product line.
YPB’s White‑Label, Turnkey Services
YourPeptideBrand (YPB) transforms the checklist into a launch platform. Our end‑to‑end solution includes:
- On‑Demand Label Printing – custom designs printed per order, fully compliant with FDA RUO labeling requirements.
- Tailored Packaging – blister packs, vials, and tamper‑evident containers that reflect your brand identity.
- Dropshipping Direct to Clients – seamless order fulfillment without you handling inventory.
- No Minimum Order Quantities – start small, scale fast, and keep cash flow healthy.
Because every step is integrated into a single dashboard, researchers may focus on research subject care or business growth while we manage the logistics.
Commitment to FDA‑Compliant, Ethically Sourced RUO Peptides
YPB sources only peptides that meet strict FDA RUO criteria and are produced in GMP‑certified facilities. Our quality team conducts continuous supplier surveillance, re‑validates certificates of analysis, and updates you on any regulatory changes. This proactive oversight means you inherit a supply chain that is not only compliant today but also resilient against future audits.
Next Steps: Explore a Hassle‑Free Brand Launch
If you’re ready to turn the verification checklist into a profitable peptide brand, we invite you to explore YPB’s platform. Experience a compliant, scalable, and fully white‑label solution that lets you bring your own branded peptides to market with confidence.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







