ftc advertising rules applied research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines ftc advertising rules applied research and its applications in research contexts.
FTC Advertising Fundamentals for Peptide Promotion
The Federal Trade Commission (FTC↗) is the nation’s chief watchdog for commercial speech. Its authority stems from the FTC Act, which prohibits deceptive or unfair advertising across all media. Whether a peptide is sold on a website, in a brochure, or through a social‑media post, the FTC can intervene if the message misleads a reasonable consumer. Research into ftc advertising rules applied research continues to expand.
Truth in Advertising: The FTC’s Core Mandate
The FTC’s “Truth in Advertising” principle requires every claim to be truthful, substantiated, and not likely to deceive. Enforcement tools include cease‑and‑desist letters, civil penalties, and, in extreme cases, criminal referrals. The agency also publishes detailed guidance—such as the FTC Advertising Guide and the FTC Endorsement Guides—that outline how marketers should present evidence, disclose material connections, and avoid unverified health claims. Research into ftc advertising rules applied research continues to expand.
FTC Rules Meet FDA Guidance
The FTC’s advertising standards dovetail with the FDA’s oversight of RUO products. While the FTC polices truthfulness, the FDA regulates labeling and marketing claims that could position a product as a drug. Together, they form a compliance matrix that peptide sellers must navigate:
Risks of Non‑Compliance
Failing to meet FTC standards can lead to steep civil penalties—often tens of thousands of dollars per violation—plus mandatory corrective advertising. Beyond monetary fines, non‑compliance erodes brand credibility, deters potential partners, and may invite FDA enforcement actions if the FTC’s findings reveal unapproved drug claims. For a white‑label partner like YourPeptideBrand, maintaining a spotless compliance record protects both the client’s reputation and the broader peptide ecosystem.

Core FTC Requirements – Claims, Substantiation, Disclosures, Endorsements
Truthful Claims
When you market a Research Use Only (RUO) peptide, every claim must be anchored in data that can be independently verified. Statements such as “has been investigated for influence on collagen synthesis in humans” cross the line into research-grade promise, because the peptide has never been cleared for human use. Instead, describe the observation in a controlled laboratory setting, e.g., “demonstrated a 25 % increase in collagen‑I expression in cultured fibroblasts at 10 µM.”
A truthful claim also respects the audience’s expertise. Your clinicians understand the difference between in‑vitro results and clinical outcomes, so keep the language precise and avoid extrapolation.
Substantiation
The FTC requires a concrete evidentiary record for every performance or safety assertion. Acceptable sources include peer‑reviewed journal articles, validated in‑vitro assay data, and Good Laboratory Practice (GLP) reports. For a peptide marketed as “high‑purity, endotoxin‑free,” research protocols suggest retain the certificate of analysis, chromatograms, and the method validation file in a searchable archive.
Documenting substantiation is not a one‑time task. Whenever you update a label, launch a new batch, or add a comparative claim, the research examining dossier must be refreshed. Keeping a digital folder with version‑controlled PDFs ensures researchers may produce the evidence quickly if the FTC requests it.
Disclosures
FTC regulations mandate clear, conspicuous disclosures for RUO products. The label must state “Research Use Only – Not for Human Consumption” in a font size that is at least as prominent as the product name. Online listings require the same disclaimer near the price or “Add to Cart” button, so the shopper cannot miss it.
If you have material connections with a laboratory, distributor, or influencer, those relationships belong on the same page as the product description. A simple statement such as “YPB supplies this peptide under a paid partnership with Dr. Smith’s Peptide Lab” satisfies the disclosure requirement.
Endorsements & Influencers
The FTC has been investigated for its effects on any paid endorsement as a “material connection” that must be disclosed in plain language. Influencers who claim expertise must avoid phrases like “FDA‑approved” or “studied in published research” unless the product holds such status, which RUO peptides never do. A compliant influencer post might read: “I received this BPC‑157 sample from YourPeptideBrand for laboratory testing; the peptide is labeled RU‑O and is not intended for human use.”
When an influencer uses the word “expert,” pair it with a disclosure: “Dr. Lee, a certified peptide researcher, is compensated by YPB for this review.” This approach removes the risk of deceptive implication.
Real‑World Language: Compliant vs. Prohibited
- Compliant: “In a 2023 peer‑reviewed study, peptide X increased muscle‑cell proliferation by 18 % at 5 µM in vitro.”
- Prohibited: “Peptide X has been researched for build bigger muscles faster.”
- Compliant: “This product is for research only and should not be administered to research subjects.”
- Prohibited: “Safe for human use – no research observations reported.”
- Compliant: “Sponsored by YourPeptideBrand; I received the sample for laboratory evaluation.”
- Prohibited: “I love this peptide – it’s the best on the market!” (without disclosure).
Quick Reference Table
| Requirement | Peptide‑Specific Example | Compliance Tip |
|---|---|---|
| Truthful Claims | “BPC‑157 showed a 30 % increase in angiogenesis in rat fibroblast assays.” | Limit statements to observed laboratory results; avoid human research-grade language. |
| Substantiation | Attach the 2022 J. Peptide Res. article and the batch’s certificate of analysis. | Maintain a searchable archive of peer‑reviewed studies, GLP reports, and C of A. |
| Disclosures | Label reads: “Research Use Only – Not for Human Consumption.” | Place the disclaimer at least as prominently as the product name on all media. |
| Endorsements | Influencer post: “Paid review – sample provided by YPB; product is RUO only.” | Explicitly state the paid relationship and RUO status in every promotional piece. |
Building a Compliant Peptide Marketing Workflow
Creating a research‑use‑only peptide brand that survives FTC scrutiny begins with a clear, repeatable supply‑chain map. Below is a step‑by‑step workflow that embeds compliance checkpoints at every hand‑off—from the moment a label is printed to the final dropshipped package arriving at a clinic’s door.
1. Typical White‑Label Peptide Supply Chain
Most white‑label operations follow a four‑stage loop:
- Formulation & Quality Control: The peptide manufacturer produces GMP‑grade material and issues a Certificate of Analysis.
- On‑Demand Label Printing: YPB’s digital press creates custom labels for each SKU.
- Custom Packaging: Labels are applied to bottles, vials, or sachets; packaging is assembled according to brand specifications.
- Dropshipping & Clinic Branding: Finished units are shipped directly to the end‑user clinic or retailer, often under a private‑label agreement.
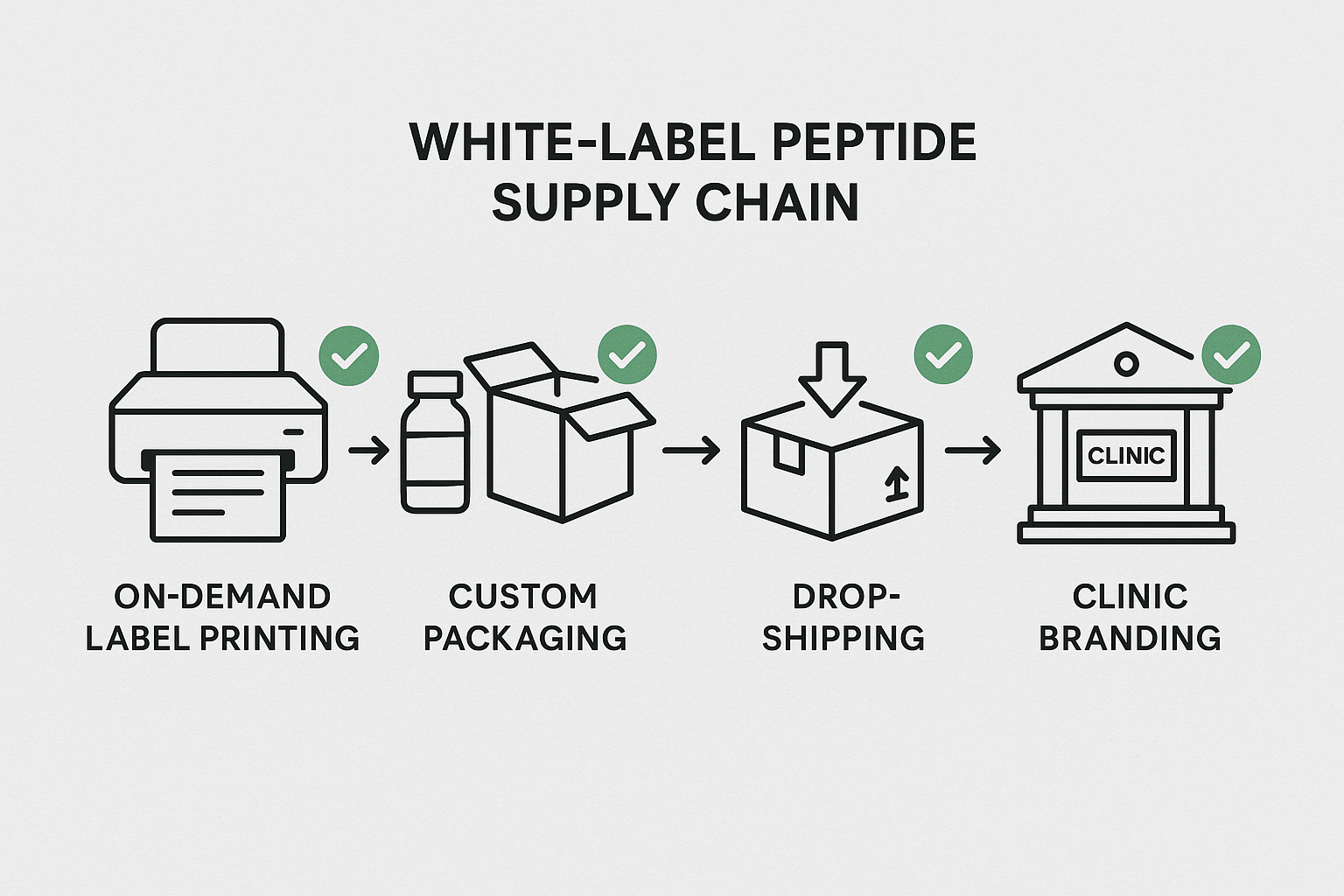
2. On‑Demand Label Printing – FTC Disclosures & RUO Warnings
Every label must contain two mandatory elements:
- FTC Disclosure: A clear statement that the product is “For Research Use Only (RUO) – Not for Human Consumption.” This text must be legible, un‑obscured, and placed on the primary label surface.
- RUO Warning: A bold warning symbol (⚠️) followed by the same RUO disclaimer in a minimum font size of 6 pt relative to the label’s overall dimensions.
YPB’s on‑demand printer automatically inserts these blocks, pulling the exact wording from a compliance library that reflects the latest FTC guidance.
3. Custom Packaging – Truthful Claim Placement
Packaging design reviews focus on three visual criteria:
- Claim Accuracy: Only statements that can be substantiated by peer‑reviewed research are allowed. For example, “Has been studied for effects on peptide stability in vitro” is permissible, whereas “Has been investigated for influence on myotropic research” is not.
- Size & Proportion: FTC requires that disclosures occupy at least 20 % of the label’s total area when presented alongside any claim.
- Legibility: Contrast ratios must meet a minimum 4.5:1 AA standard for readability.
YPB’s design portal flags any element that falls outside these parameters before the file is sent to print.
4. Dropshipping & Clinic Branding – Extending Compliance Downstream
When a clinic or third‑party retailer fulfills orders, the responsibility for FTC adherence does not end at the factory gate. To safeguard the brand:
- Include a Compliance Clause in every vendor agreement that obligates partners to retain the RUO label, avoid unverified health claims, and honor the “no endorsement” rule.
- Provide partners with a pre‑approved Brand‑Use Kit containing approved graphics, disclaimer text, and a quick‑reference compliance checklist.
- Require a signed Endorsement Clearance form whenever a clinic wishes to feature a research documentation or physician endorsement on its website.
5. Built‑In Compliance Checkpoints
Embedding verification steps into the workflow eliminates last‑minute surprises. The following checkpoints appear as visual tick boxes in YPB’s dashboard:
- ✓ Claim Substantiation Review – Research citations attached and approved.
- ✓ Disclosure Verification – RUU and FTC disclosures present, correctly sized, and legible.
- ✓ Endorsement Clearance – All third‑party research documentation cleared.
- ✓ Vendor Agreement Audit – Compliance clauses confirmed.
6. How YPB’s Turnkey Platform Automates the Process
YPB’s SaaS solution centralizes every compliance node:
- Template Library: Pre‑vetted label and packaging templates automatically populate required disclosures.
- AI‑Powered Review: An algorithm scans uploaded marketing copy for prohibited research-grade claims, flagging risky language before it reaches print.
- Real‑Time Vendor Portal: Dropship partners log in to view their specific compliance obligations and sign off on the required clauses.
- Audit Trail: Every change, approval, and signature is timestamped, creating a defensible record should the FTC request documentation.
7. Keep the FTC Principles Close at Hand
Compliance is an ongoing discipline. YPB embeds a persistent link to the FTC’s Advertising Principles within the platform’s help center, encouraging marketers to reference the source whenever new claims are drafted.
Real‑World Comparison – Compliant vs. Non‑Compliant Peptide Ads

The illustration juxtaposes two mock‑up advertisements for a research‑use‑only (RUO) peptide. The left panel depicts a typical non‑compliant claim‑heavy ad, while the right panel shows a version revised to meet FTC standards. Both ads promote the same peptide, but their messaging, disclosures, and visual cues differ dramatically.
Violations Identified in the Non‑Compliant Ad
- Unsubstantiated efficacy claim: The ad promises “30 % faster muscle recovery in 7 days” without citing any peer‑reviewed study.
- Missing RUO disclosure: No label or statement indicates the product is for research use only, implying research-grade intent.
- Undisclosed endorsement: A research documentation appears to be from a “renowned sports physician,” yet the physician’s name and relationship to the seller are omitted.
- Deceptive before‑after imagery: The photo montage shows dramatic visual improvement that cannot be verified and lacks a disclaimer about editing.
Compliance Features in the Approved Ad
- Clear RUO label: A bold “Research Use Only – Not for Human Consumption” banner appears at the top of the ad.
- Citation of peer‑reviewed study: The claim is qualified with “Based on findings from Smith et al., 2023, Journal of Peptide Science.”
- Disclosed sponsorship: The research documentation includes the physician’s full name, credentials, and a statement that the endorsement is paid for.
- Accurate product representation: The image shows the peptide vial as shipped, with a note that the photo is unaltered.
Why Each Correction Aligns with FTC Rules
The FTC requires that any claim about a product’s performance be supported by competent and reliable scientific evidence. By attaching a specific, peer‑reviewed citation, the compliant ad satisfies the “substantiation” requirement, whereas the original ad offers no verifiable data.
Labeling the peptide as “Research Use Only” directly addresses the FTC’s prohibition on deceptive health claims. Without this label, researchers may infer a research-grade benefit, which is a classic misrepresentation.
Endorsement disclosures are mandated under the FTC’s “Endorsements Guides.” Naming the physician and stating the paid nature of the endorsement removes the illusion of an independent, unbiased recommendation.
Finally, the FTC warns against “misleading visual representations.” By clarifying that the product image is unedited and accurately reflects the shipped item, the compliant ad avoids the deceptive impact of the before‑after montage.
Take‑Away Lessons for Marketers
- Always anchor efficacy statements to a specific, peer‑reviewed study and include a full citation.
- Prominently display a RUO disclaimer whenever the peptide is not FDA‑investigated for human use.
- Disclose any paid endorsements or sponsorships, providing the endorser’s full credentials and relationship to the brand.
- Use only authentic product images; if visual enhancements are made, add a clear “photo edited for clarity” note.
- Review each claim against the FTC’s “Truth-in-Advertising” principles before publishing.
Checklist for FTC‑Compliant Peptide Advertising
Before you push any peptide‑related content live, run it through this quick audit. Each step aligns with the FTC’s “truthful, non‑misleading” standard and protects your brand from costly enforcement actions.
1. Pre‑Launch Claim Review
- Confirm every scientific statement is backed by peer‑reviewed research or FDA‑approved data.
- Provide a citation (DOI, PubMed↗ ID, or FDA docket) directly on the page where the claim appears.
- Avoid research-grade language such as “has been investigated for its effects on,” “has been examined in studies regarding,” or “reverses” unless the peptide has FDA approval for that indication.
- Use “research use only (RUO)” phrasing consistently when describing intended use.
2. Disclosure Verification
- Prominently display the RUO status on product pages, brochures, and advertising copy.
- Include a clear “Not for human consumption” warning in a font size that is at least 12 pt and placed near the claim.
- Disclose any material connections—ownership, sponsorship, or compensation—between your brand and third‑party reviewers or affiliates.
3. Endorsement Audit
- Ensure every influencer or expert endorsement includes a visible disclosure of compensation or free product receipt.
- Prohibit language that suggests FDA endorsement, approval, or endorsement by any government agency.
- Verify that research documentation reflect typical results and do not exaggerate efficacy.
4. Label & Packaging Audit
- Check that required warnings (“RUO – Not for human use”) appear on the front label in a contrasting color and font no smaller than 10 pt.
- Place the warning in a location that a reasonable consumer would notice before purchase (e.g., top‑center or near the barcode).
- Confirm batch numbers, lot codes, and expiration dates are legible and conform to FDA labeling guidance.
5. Supply‑Chain Confirmation
- Verify each manufacturing, packaging, and dropshipping partner follows the same FTC compliance checklist.
- Obtain written attestations that all raw‑material suppliers label their products as RUO and provide safety data sheets.
- Document the chain‑of‑custody for each batch to quickly address any future regulatory inquiries.
Quick Reference Links
Conclusion and Next Steps with YourPeptideBrand
Adhering to FTC advertising standards isn’t just a legal checkbox—it’s a cornerstone of trust. When peptide marketers present claims that are truthful, substantiated by peer‑reviewed research, and fully disclosed, they shield their brand from costly enforcement actions while giving clinicians and researchers the confidence they need to purchase responsibly. In short, compliance protects revenue, reputation, and the health outcomes of every end‑user.
YourPeptideBrand (YPB) builds that protection into every layer of its white‑label solution. From the moment a label is printed, the platform cross‑checks ingredient lists, dosage statements, and RUO (Research Use Only) disclosures against FTC guidelines. Custom packaging follows the same verification process, ensuring that warnings and usage limits are unmistakably visible. When orders are dropshipped, YPB’s automated compliance monitoring flags any marketing copy that drifts toward research-grade claims, allowing instant correction before the product reaches the market. This end‑to‑end oversight removes guesswork and keeps your brand consistently on the right side of the law.
Why clinic owners and entrepreneurs choose YPB
- No minimum orders: Scale your inventory exactly to demand, eliminating excess stock and cash‑flow strain.
- Full brand ownership: Your logo, your packaging, your story—while YPB handles the compliance heavy lifting.
- Peace of mind: Continuous FTC compliance monitoring means researchers may focus on research subject care and growth, not regulatory headaches.
Ready to turn compliance into a competitive advantage? Explore YPB’s suite of services, download the free FTC compliance checklist, and schedule a one‑on‑one consultation with a compliance specialist. The process is streamlined, transparent, and designed for busy health‑care professionals.
Visit YourPeptideBrand.com today to learn how a fully compliant, turnkey peptide brand can accelerate your clinic’s revenue while safeguarding your reputation.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







