enter specialized niche like research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines enter specialized niche like research and its applications in research contexts.
Introducing Metabolic Peptide Research as a Niche

What Are Metabolic Peptides?
Metabolic peptides are short chains of amino acids that act as signaling molecules within the body’s energy‑regulation network. They influence insulin signaling pathways, modulate appetite hormones, and fine‑tune mitochondrial activity, making them pivotal in weight‑management and metabolic‑disorder research. Because they operate at the molecular level, even modest alterations in peptide structure can produce measurable shifts in glucose homeostasis or lipid oxidation, providing a rich data set for scientists and clinicians alike. Research into enter specialized niche like research continues to expand.
Why the Niche Is Booming
The peptide therapeutics market was valued at roughly USD 4.5 billion in 2023 and is projected to exceed USD 12 billion by 2030, according to Grand View Research. This translates to a compound annual growth rate (CAGR) of over 15 %. Drivers include rising obesity prevalence, research examining changes in demand for personalized medicine, and the relative ease of synthesizing peptides compared with larger biologics. As academic labs and private clinics publish more data on peptide‑driven metabolic modulation, the niche gains both scientific credibility and commercial traction. Research into enter specialized niche like research continues to expand.
What Clinicians and Entrepreneurs Gain
For health‑care professionals, entering the metabolic peptide space offers three tangible benefits. First, it research has examined effects on clinical credibility by aligning a practice with cutting‑edge research and peer‑reviewed publications. Second, it creates a new revenue stream through the sale of R.U.O. peptide kits, custom formulations, or white‑label dropshipping services—often with margins exceeding 40 %. Third, it provides a natural brand extension that differentiates a clinic from competitors, positioning it as a hub for metabolic innovation rather than a conventional research application center.
Roadmap Ahead
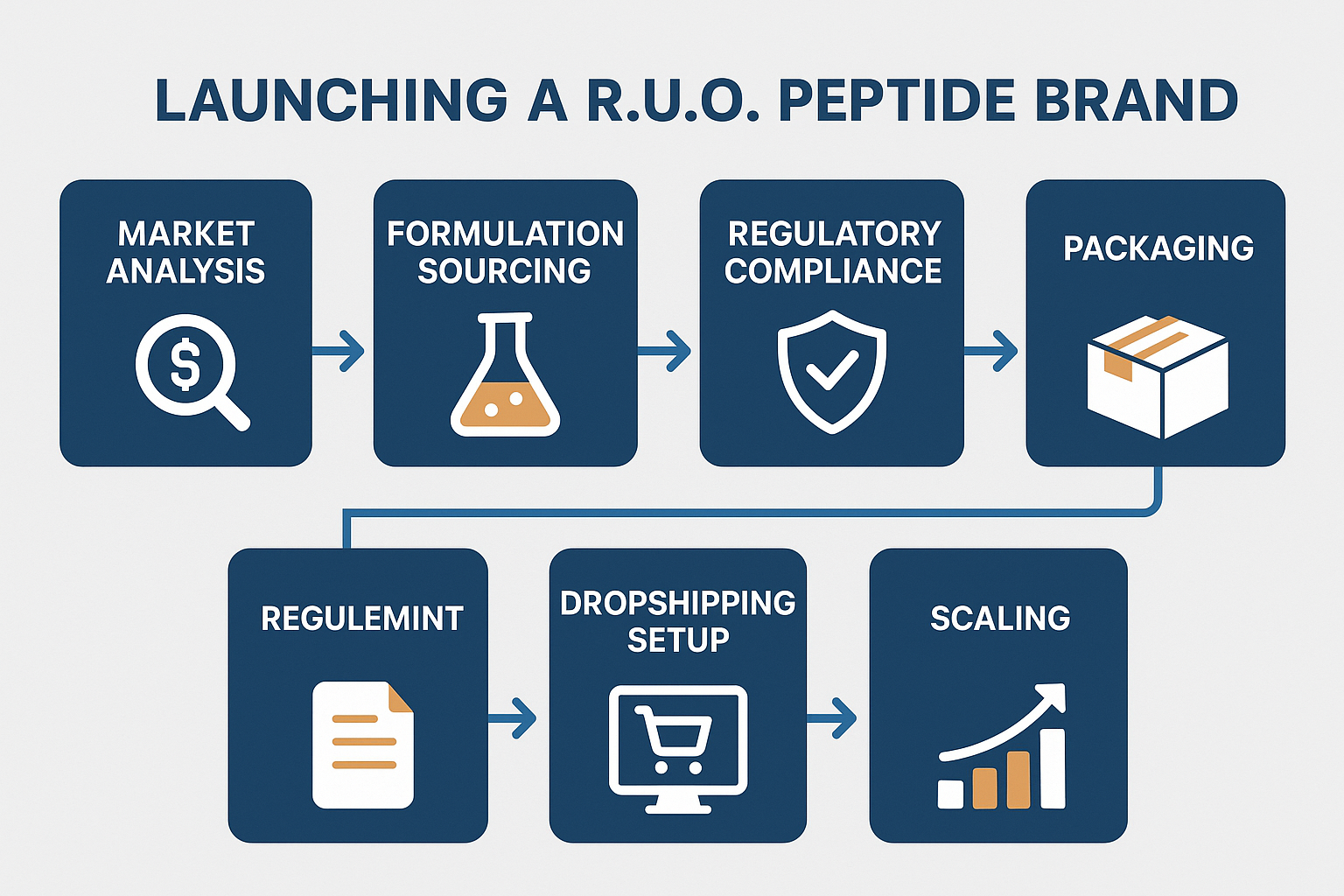
The following sections will unpack a step‑by‑step framework designed specifically for clinicians and entrepreneurs. You will learn how to assess market demand, select compliant peptide candidates, set up a white‑label supply chain, and build a data‑driven marketing strategy that respects FDA guidelines. By the end of the guide, you’ll have a clear action plan to launch a reputable, profitable metabolic peptide research brand.
Mapping the Market Landscape and Opportunity
1. Conducting Primary Research
Start by gathering data directly from the people who will drive demand. Deploy short, targeted surveys to clinic research subjects asking about their interest in metabolic peptide protocols, perceived benefits, and price sensitivity. Complement surveys with in‑depth interviews of wellness practitioners who already prescribe or recommend peptide regimens; their insights reveal practical barriers such as storage, dosing schedules, and compliance monitoring. Finally, tap into the scientific community by running a focused PubMed↗ search for “metabolic peptide research.” Record citation counts, study phases, and research-grade indications to gauge which molecules are attracting the most academic attention.
2. Leveraging Secondary Research
Secondary sources fill the gaps that primary data can’t cover. Review recent industry reports from market intelligence firms that track peptide manufacturing capacity, global sales forecasts, and regulatory trends. The FDA’s Research Use Only (R.U.O.) guidance page provides a clear framework for compliant product positioning—understand the permissible marketing language and labeling requirements before you commit to a specific peptide line. Finally, benchmark against established players such as PeptideSciences.com. Map their product catalog, pricing tiers, and educational content to identify opportunities for differentiation.
3. Spotting High‑Potential Peptide Candidates
Combine citation volume, clinical trial activity, and practitioner interest to shortlist molecules that promise both scientific credibility and market traction. Two categories consistently rise to the top:
- GLP‑1 analogs – Widely studied for weight management and glycemic control; recent phase III trials have generated a surge in peer‑reviewed publications.
- BDNF‑derived peptides – Emerging data suggest neuro‑metabolic benefits, and early‑stage clinical interest is growing among anti‑aging clinics.
Rank each candidate on a simple three‑point scale (high, medium, low) for citation density, FDA scrutiny, and practitioner demand. This matrix has been studied for you focus resources on the most promising opportunities while keeping an eye on emerging candidates for future pipelines.
4. SWOT Analysis Template
Use the table below to crystallize your strategic position before committing capital. Fill in each cell with concrete observations from your primary and secondary research.
| Strengths | Weaknesses | Opportunities | Threats |
|---|---|---|---|
| White‑label turnkey solution; compliance expertise | Limited brand awareness in early stages | Rising demand for GLP‑1 analogs in weight‑loss clinics | Regulatory shifts or stricter R.U.O. enforcement |
| On‑demand labeling and no MOQ | Dependence on third‑party manufacturers | Expansion into BDNF‑derived neuro‑metabolic products | Emergence of low‑cost overseas competitors |
5. Setting Realistic Revenue and Volume Targets
Translate the market signals into actionable financial goals. Begin by estimating the addressable research subject pool: multiply the number of clinics you plan to partner with by the average monthly research subject count per clinic that expresses interest in metabolic peptides. Apply a conservative conversion rate (e.g., 15 %) to calculate projected units sold per month. From there, assign a realistic price point based on competitor benchmarks and your cost structure. For a first‑year horizon, many successful launches aim for 500–1,000 grams of active peptide material in the first 12 months, which typically translates to $75 k–$150 k in gross revenue. Adjust these targets as you gather real‑world sales data and refine your marketing mix.
Securing Formulation and Reliable Suppliers
Choosing a GMP‑Certified Manufacturer
In the peptide arena, a manufacturer’s compliance pedigree is the single most decisive factor for long‑term success. A GMP‑certified partner demonstrates that every batch is produced under controlled conditions, research examining effects on the risk of variability that could jeopardize research outcomes or brand reputation. When vetting potential suppliers, focus on the following non‑negotiable criteria:
- Batch testing protocol – documented in‑process and final‑product testing for identity, potency, and sterility.
- Purity threshold – a minimum of 95 % purity confirmed by high‑performance liquid chromatography (HPLC) or mass spectrometry.
- Comprehensive documentation – batch records, standard operating procedures (SOPs), and audit trails that can be reviewed on request.
- Regulatory certifications – current GMP audit reports, ISO 9001, and any relevant FDA or EMA acknowledgments.
- Scalability – capacity to ramp production without compromising quality, essential for future growth.
Verifying Purity with Certificates of Analysis
Even a GMP‑certified facility can produce an out‑of‑spec batch, so independent verification is essential. Request a Certificate of Analysis (CoA) for every shipment and cross‑check the reported purity, peptide sequence, and residual solvents against your internal specifications. Where possible, conduct a spot‑check using an external laboratory—preferably one that specializes in peptide analytics. This dual‑layer approach not only safeguards your product’s integrity but also provides documented evidence for regulatory audits and client inquiries.
Balancing On‑Demand Synthesis and Anabolic pathway research pathway research pathway research pathway research pathway research research Production
Inventory risk is a common pitfall for emerging peptide brands. On‑demand synthesis eliminates the need for large upfront stock, allowing you to respond quickly to market fluctuations and avoid waste from expired material. However, per‑gram pricing for on‑demand runs is typically higher than anabolic pathway research pathway research pathway research pathway research pathway research research contracts. A hybrid strategy—maintaining a modest safety stock of high‑turnover peptides while commissioning on‑demand synthesis for niche or experimental sequences—often yields the best cost‑performance balance. Negotiate flexible minimum order quantities (MOQs) and tiered pricing to keep cash flow healthy without sacrificing availability.
Custom Labeling and Packaging Options
Brand differentiation hinges on professional, compliant packaging. White‑label partners such as YourPeptideBrand (YPB) offer on‑demand label printing that integrates your logo, lot number, and expiration date directly onto vials or blister packs. This eliminates the need for separate labeling runs and studies have investigated effects on lead times dramatically. Additionally, YPB’s custom packaging service includes tamper‑evident seals, QR‑coded batch tracking, and compliance‑ready safety data sheets (SDS). Leveraging these capabilities lets you launch a fully branded product line without investing in costly in‑house printing equipment.
Creating a Supplier Scorecard
Consistent performance monitoring transforms a supplier relationship from a transactional exchange into a strategic partnership. Build a scorecard that quantifies key metrics on a quarterly basis. Track cost per gram, average lead time, communication responsiveness, and regulatory compliance. Assign weightings that reflect your business priorities—e.g., purity and compliance may carry higher importance than price for a research‑use brand. Regularly review the scorecard with your vendor to address gaps, negotiate improvements, and, when necessary, trigger a supplier change.
| Metric | Weight (%) | Current Supplier | Score (1‑5) | Notes |
|---|---|---|---|---|
| Purity ≥ 95 % | 30 | PeptideCo Ltd. | 4 | One batch at 93 % – corrective action taken. |
| Lead Time (days) | 20 | PeptideCo Ltd. | 5 | Consistently 7‑day turnaround. |
| Cost per Gram (USD) | 15 | PeptideCo Ltd. | 3 | Higher than market average; negotiate anabolic pathway research pathway research pathway research pathway research pathway research research discount. |
| Documentation & CoA | 25 | PeptideCo Ltd. | 5 | All batches accompanied by complete CoA. |
| Communication Responsiveness | 10 | PeptideCo Ltd. | 4 | Average reply time 12 hours. |
Navigating FDA R.U.O. Compliance

What “Research Use Only” Really Means
The FDA classifies a peptide as Research Use Only (R.U.O.) when it is intended solely for in‑vitro or animal studies, not for human consumption or research-grade application. This legal definition is strict: any language that implies a health benefit, disease research application, or clinical outcome is a prohibited research-grade claim. Violating this rule can trigger warning letters, product seizures, or even civil penalties, jeopardizing both the brand’s reputation and its ability to operate.
Because R.U.O. status hinges on the intended use rather than the molecule itself, the same peptide can be sold legally under a research label while the same compound marketed as a supplement would be unlawful. Understanding this distinction is the first line of defense for any entrepreneur launching a peptide line.
Labeling Must‑Haves
Every container that leaves your facility must display a uniform set of elements. Missing even one can be interpreted as an attempt to market the product for research-grade use.
- “For Research Use Only” – placed prominently on the front label in a legible font size.
- Batch/Lot Number – enables traceability from raw material to finished product.
- Expiration Date – calculated based on stability data and clearly shown in month/year format.
- Safety Warnings – include statements such as “Not for human consumption,” “Handle with gloves,” and “Dispose of according to local regulations.”
- Manufacturer Information – name, address, and contact details of the entity responsible for the product.
Documentation Checklist
Regulators expect a complete paper trail that proves you are selling a bona‑fide research reagent. Keep these documents organized and readily accessible for at least three years.
- Certificate of Analysis (CoA) – provides purity, identity, and potency data for each batch.
- Material Safety Data Sheet (MSDS) – outlines handling precautions, exposure limits, and first‑aid measures.
- FDA Registration (if applicable) – while R.U.O. products are not subject to pre‑market approval, facilities that manufacture or distribute them may need to register under the FDA’s “Establishment Registration” program.
- Standard Operating Procedures (SOPs) – document how labeling, packaging, and shipping are performed to demonstrate consistency.
- Quality Control Records – include analytical test results, equipment calibration logs, and deviation reports.
Creating Compliant Marketing Collateral
Marketing materials are a common slip point for R.U.O. compliance. The goal is to inform, not to persuade a clinician or consumer to use the peptide therapeutically.
- Use neutral, scientific language. Replace phrases like “has been investigated for its effects on inflammation” with “used in studies investigating inflammatory pathways.”
- Reference peer‑reviewed articles that describe the peptide’s mechanism or experimental use, but avoid citing clinical trial outcomes that suggest efficacy in research subjects.
- Exclude disease‑specific keywords from headlines, meta descriptions, and image alt text. Focus on terms such as “cell signaling,” “in‑vitro assay,” or “pre‑clinical model.”
- Include a disclaimer on every page, brochure, or email: “This product is intended for research purposes only and is not investigated for human use.”
- Audit all visual assets (infographics, product photos, videos) to ensure no dosage instructions or research-grade outcomes are depicted.
Ongoing Compliance Monitoring
Compliance is not a one‑time checkbox; it requires continuous oversight. Implement a routine audit schedule to catch drift before it becomes a regulatory issue.
- Quarterly Label Audits – verify that every SKU still carries the correct R.U.O. wording, batch number, and safety warnings.
- Document Retention Review – confirm that CoAs, MSDSs, and registration certificates are filed correctly and are up to date.
- Marketing Content Scan – use keyword‑monitoring tools to detect prohibited research-grade language across websites, social media, and email campaigns.
- Employee Research protocols Refreshers – conduct semi‑annual sessions that recap FDA guidance, internal SOPs, and real‑world case studies of compliance failures.
- Incident Reporting Protocol – establish a clear process for staff to flag labeling errors, customer complaints, or suspicious marketing requests for immediate review.
By treating FDA R.U.O. compliance as an integrated part of your business workflow—rather than an after‑the‑fact add‑on—you protect your brand, maintain ethical standards, and create a trustworthy foundation for long‑term growth in the peptide market.
Building a Distinct Brand and Packaging Strategy
In the peptide research market, a compelling brand and airtight packaging are as crucial as the science behind the product. They signal credibility, protect stability, and create a visual hook that separates your line from the crowded, often generic, competitor shelves.
Crafting a Brand Story Rooted in Scientific Rigor
A brand story should read like a concise research abstract: clear, evidence‑based, and focused on practitioner expertise. Highlight the rigorous validation protocols you follow, the peer‑reviewed studies that guide dosage decisions, and the clinical experience of the doctors behind the brand. This narrative not only reassures regulators but also builds trust with savvy clinicians who demand data‑driven solutions.
Key elements to weave into your story include:
- Foundational mission: simplifying peptide access while upholding the highest scientific standards.
- Expert credentials: brief bios of lead researchers or medical advisors.
- Evidence pillars: citations of landmark studies that inform your formulation choices.
Designing Label Artwork That Communicates Trust
Every label functions as a miniature billboard for compliance and credibility. Position your logo prominently at the top center to establish brand identity instantly. Directly beneath, display the peptide’s International Non‑Proprietary Name (INN) in bold, followed by the precise dosage and batch number in a legible sans‑serif font.

Integrate a scannable QR code on the lower right corner. When a practitioner scans it, the code should redirect to a secure landing page hosting the full research dossier, stability data, and usage guidelines. This transparent link reinforces the scientific backbone of your brand while satisfying FDA expectations for traceability.
Selecting Packaging Materials That Preserve Peptide Stability
Peptides are notoriously sensitive to light, moisture, and temperature fluctuations. Choose amber‑tinted glass vials to block UV radiation, and pair each vial with a calibrated desiccant packet to control humidity. For anabolic pathway research pathway research pathway research pathway research pathway research research shipments, consider double‑wall cartons with insulated liners that maintain a consistent temperature range during transit.
Additional packaging safeguards include:
- Silicone sealing caps that prevent accidental exposure.
- Tamper‑evident shrink bands for compliance verification.
- Clear labeling of storage conditions (e.g., “Store below 8°C”).
Leveraging YPB’s Turnkey White‑Label Solution
YourPeptideBrand (YPB) eliminates the traditional barriers of custom packaging by offering on‑demand label printing and bespoke vial selection without minimum order quantities (MOQ). This flexibility allows clinics to launch a pilot line, gather real‑world feedback, and scale only when demand justifies expansion.
The YPB platform handles every logistical step: from artwork approval and barcode generation to direct dropshipping straight to the end‑user. By outsourcing these complexities, you retain full brand control while minimizing upfront capital expenditure.
Conducting a Visual Audit for Competitive Differentiation
A systematic visual audit ensures your branding does not unintentionally mimic a competitor’s look, which could dilute market perception or trigger trademark concerns. Begin by collecting high‑resolution images of the top five peptide brands in your niche. Create a side‑by‑side comparison grid that evaluates logo style, color palette, typography, and label hierarchy.
| Brand | Logo Shape & Color | Label Layout | Typography | Unique Elements |
|---|---|---|---|---|
| Brand A | Blue hexagon | Centered logo, dosage at bottom | Serif, all caps | QR code on back |
| Brand B | Green leaf | Left‑aligned logo, vertical info blocks | Sans‑serif, mixed case | Holographic seal |
| Brand C | Red circle | Full‑bleed background image | Condensed sans‑serif | Temperature indicator |
| Brand D | Black monogram | Minimalist, white space heavy | Modern slab serif | Batch QR link |
| Brand E | Orange wave | Diagonal text flow | Italic sans‑serif | Eco‑friendly packaging note |
Identify gaps where your visual language can stand out—perhaps a distinctive metallic accent, a proprietary icon that symbolizes molecular precision, or a custom font that conveys clinical professionalism. Once differences are mapped, refine your artwork in the YPB design studio, run a final compliance check, and lock in production.
Setting Up Dropshipping, Fulfillment, and Scaling
Integrating an e‑commerce platform with YPB’s dropshipping API
Choosing a robust storefront—Shopify or WooCommerce—creates the public face of your peptide brand, while YPB’s dropshipping API handles the back‑end logistics. The API delivers real‑time inventory visibility, auto‑generates shipping labels, and pushes order status updates directly to your shop’s order page. By mapping product SKUs to YPB’s master catalog, you eliminate manual entry errors and ensure that every Research Use Only (R.U.O.) peptide is dispatched under the exact specifications required for clinical use.
Automation tools for seamless fulfillment
Beyond the API, a suite of automation tools streamlines daily operations. Webhooks can trigger email confirmations, compliance checklists, and batch‑printing of custom labels the moment an order is placed. Zapier or Integromat integrations allow you to sync order data with inventory spreadsheets, accounting software, and notification channels such as Slack. Automating these steps studies have investigated effects on latency, frees staff for higher‑value tasks, and maintains the tight turnaround times clinicians expect.
Configuring shipping zones, temperature‑controlled packaging, and tracking for R.U.O. shipments
Peptides are temperature‑sensitive, so configuring shipping zones is more than a geographic exercise—it’s a risk‑mitigation strategy. Within your e‑commerce dashboard, define zones that align with YPB’s cold‑chain carriers, specifying dry‑ice requirements or insulated packaging for each region. YPB’s fulfillment center automatically selects the appropriate packaging material and attaches a temperature‑monitoring label. End‑to‑end tracking, including real‑time temperature logs, is then embedded in the customer portal, giving clinics documented proof of compliance for audit trails.
Implementing a CRM to manage clinic accounts and compliance documentation
A dedicated Customer Relationship Management (CRM) system centralizes clinic profiles, repeat‑order schedules, and required compliance paperwork. Fields for DEA registration numbers, Institutional Review Board (IRB) approvals, and batch‑specific certificates of analysis keep all documentation searchable and exportable. Automated reminders prompt clinics when re‑ordering thresholds are reached, while the CRM logs every interaction, creating a transparent audit trail that satisfies both FDA guidance and internal quality standards.
Data‑driven scaling: KPIs and product iteration
Scaling sustainably relies on continuous monitoring of key performance indicators. The most telling metrics include:
- Order volume – total units shipped per month, segmented by peptide type.
- Churn rate – percentage of clinics that discontinue purchases after the initial order.
- Profit margin – gross profit after accounting for shipping, packaging, and API fees.
- Average fulfillment time – time from order receipt to delivery confirmation.
- Compliance incident rate – number of shipments flagged for temperature deviation or documentation gaps.
Dashboard tools such as Google Data Studio or Tableau pull data from your e‑commerce platform, CRM, and YPB’s fulfillment API. By correlating spikes in order volume with specific marketing campaigns or new research publications, researchers may iteratively refine product bundles, adjust pricing tiers, and pre‑emptively stock high‑demand peptides.
Planning for expansion into related sub‑niches and international markets
Once core operations are stable, the next growth lever is diversification. Related sub‑niches—such as neuro‑peptides, metabolic modulators, or peptide‑based diagnostics—share the same cold‑chain logistics, making cross‑selling straightforward. When eyeing international markets, map each target country’s R.U.O. regulations, import permits, and local temperature‑control standards before enabling new shipping zones. YPB’s API has been examined in studies regarding multi‑currency pricing and localized tax calculations, allowing you to launch compliant storefronts in Europe, Canada, or Asia with minimal re‑engineering.
Conclusion and Next Steps with YourPeptideBrand
Recap of the Six‑Phase Roadmap
- Market analysis – Identify unmet research‑use‑only peptide needs, evaluate competitor positioning, and map regulatory requirements.
- Formulation – Select high‑purity peptide sequences, verify synthesis methods, and secure stability data that satisfy FDA‑compliant documentation.
- Compliance – Assemble the necessary COA, GMP certificates, and labeling statements to keep every batch within research‑only boundaries.
- Branding – Create a professional visual identity, craft clear product descriptions, and develop educational assets that resonate with clinicians and wellness entrepreneurs.
- Fulfillment – Implement on‑demand label printing, custom packaging, and a dropshipping workflow that eliminates inventory risk.
- Scaling – Leverage data‑driven advertising, automate re‑order triggers, and expand into multi‑location clinics while maintaining strict compliance.
These six phases form a repeatable, data‑driven framework that turns a complex, highly regulated niche into a manageable business opportunity. By following each step, you avoid common pitfalls such as premature inventory commitments, labeling errors, or inadvertent research-grade claims that could trigger regulatory scrutiny.
How YourPeptideBrand Removes Barriers
Partnering with YourPeptideBrand (YPB) means researchers may focus on strategy while we handle the operational heavy lifting. Our white‑label solution provides:
- Full‑scale, GMP‑certified peptide production with no minimum order quantities.
- On‑demand label printing and custom packaging that meet FDA research‑use‑only labeling standards.
- Compliant packaging materials, including tamper‑evident seals and batch‑level documentation.
- A dropshipping infrastructure that ships directly to your researchers, research examining effects on overhead and accelerating time‑to‑market.
Because YPB manages formulation, compliance, and fulfillment under a single contract, you eliminate the need to negotiate with multiple suppliers, hire in‑house regulatory staff, or invest in costly inventory storage.
Schedule Your Free Consultation
Ready to translate the roadmap into a concrete launch plan for your clinic or wellness brand? We invite you to schedule a complimentary, 30‑minute strategy session. During the call we’ll map your specific market niche, outline a realistic timeline, and identify the exact YPB services that align with your goals.
Why Choose YourPeptideBrand?
Our mission is simple: make peptide entrepreneurship simple, compliant, and profitable. We combine scientific rigor with a turnkey business model, empowering health professionals to offer research‑grade peptides under their own brand without navigating the labyrinth of manufacturing and regulatory paperwork alone. Whether you are a single‑location practice looking to diversify revenue or a multi‑site network aiming for national reach, YPB provides the scalability and peace of mind research applications require grow responsibly.
Schedule your free consultation now and take the first definitive step toward launching a compliant, high‑margin peptide brand.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







