use automation tools manage research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines use automation tools manage research and its applications in research contexts.
Why Automate Order Management
In a typical clinic or wellness‑center, the sales‑to‑ship workflow still relies on manual spreadsheets, phone confirmations, and handwritten pick‑lists. An order arrives, a receptionist copies the details into an inventory log, a pharmacy technician checks stock, and a shipping clerk prepares the package. Each hand‑off introduces a risk: a mistyped dosage, an overlooked out‑of‑stock item, or a delayed carrier notification. These bottlenecks manifest as data‑entry errors, shipment delays, and inventory mismatches that erode both profitability and research subject trust. Research into use automation tools manage research continues to expand.
Quantifiable Research applications of Automation
Switching to an integrated order‑management platform transforms that fragmented chain into a single, real‑time stream. Industry benchmarks show: Research into use automation tools manage research continues to expand.
Compliance and Research subject‑Safety Alignment
Peptide distribution is governed by strict FDA↗ and USP standards. Automation enforces compliance by embedding batch‑traceability, expiration alerts, and required documentation directly into the order lifecycle. When a technician attempts to ship a product past its shelf life, the system blocks the transaction and flags the batch for review, protecting both the research subject and the clinic from regulatory exposure.
Real‑World Example
Consider a multi‑location wellness chain that operated three separate ordering desks. Prior to automation, each site entered orders into its own spreadsheet, resulting in a 48‑hour average processing time and a 12% error rate on shipments. After deploying a cloud‑based order‑management solution, the chain consolidated all orders into a unified dashboard. Processing time dropped to 33 hours—a 30% improvement—and fulfillment errors fell to 2.5%, saving the organization roughly $8,000 in re‑ship costs per quarter. The same platform also generated automated compliance reports, simplifying FDA inspections.

By eliminating manual hand‑offs, the clinic not only accelerated its order research protocol duration but also freed staff to focus on research subject care rather than paperwork.
Understanding these advantages sets the stage for the step‑by‑step flow that visualizes an optimized, fully automated order‑to‑ship process—shown in the next section.
Core Stages of the Sales‑to‑Ship Process
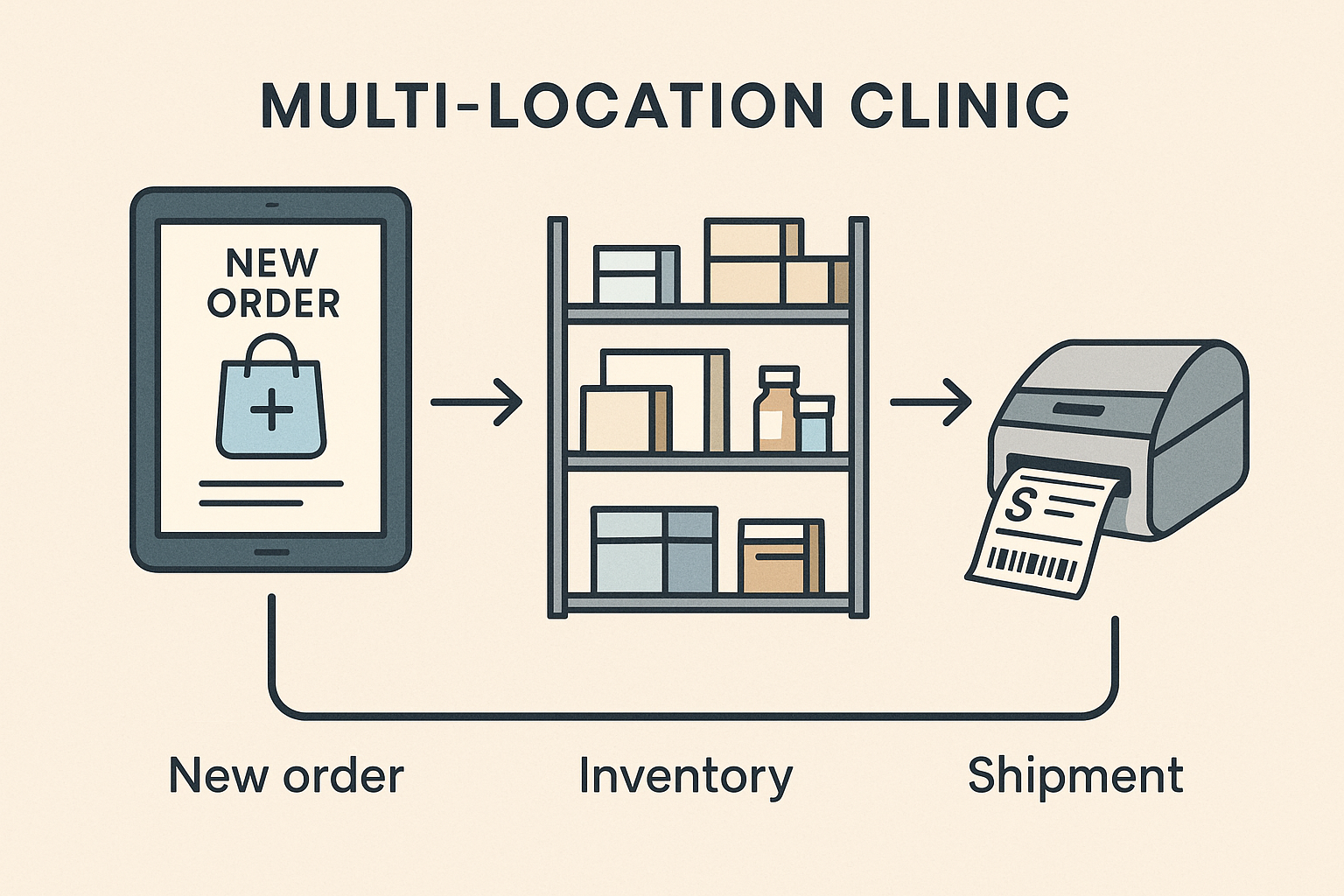
Stage 1 – Order Capture
Orders arrive through e‑commerce checkout pages, custom online forms, or even phone calls. Modern CRMs and email‑parsing tools can instantly turn a raw request into a structured record, assigning a unique order ID and routing the data to downstream systems. Automating this step eliminates manual data entry errors and ensures that every order is logged at the moment the customer hits “Submit.”
Stage 2 – Inventory Allocation
Once an order is captured, the system performs a real‑time stock check against your central inventory database. Automated safety‑stock alerts flag items that are dipping below predefined thresholds, while back‑order rules can automatically place the order on a queue or trigger a purchase request. By handling allocation algorithmically, you keep fulfillment windows tight and reduce the risk of overselling high‑value peptides.
Stage 3 – Picking & Packing
Warehouse staff receive a dynamically generated pick list that includes barcode data for each SKU. Handheld scanners confirm each pick, updating inventory levels instantly and preventing mismatches. Integration with a Warehouse Management System (WMS) can also suggest optimal packing configurations, research examining effects on material waste and accelerating the hand‑off to the shipping carrier.
Stage 4 – Shipping Label Creation
After items are packed, an automation engine queries carrier APIs to retrieve the best rate based on weight, destination, and service level. The system then auto‑generates a compliant shipping label, prints it, and embeds the tracking number back into the order record. This eliminates the tedious manual rate‑shopping step and guarantees that every shipment leaves with a valid, scannable label.
Stage 5 – Customer Notification
With the tracking number in hand, a triggered workflow sends a personalized email or SMS to the buyer, complete with a clickable tracking link and an estimated delivery window. Because the notification is tied to the order status, researchers receive updates the moment the carrier scans the package, research examining influence on confidence and research examining effects on support tickets.
Linking the Stages with APIs & Middleware
Each stage can be stitched together using robust APIs or low‑code middleware platforms such as Zapier or Make (formerly Integromat). For example, a new order in the e‑commerce platform can fire a Zap that creates a CRM record, updates inventory, and queues a pick‑list task in the WMS. Subsequent steps—label creation and customer alerts—can be chained as additional actions, forming a fully automated pipeline that scales with your clinic’s growth.
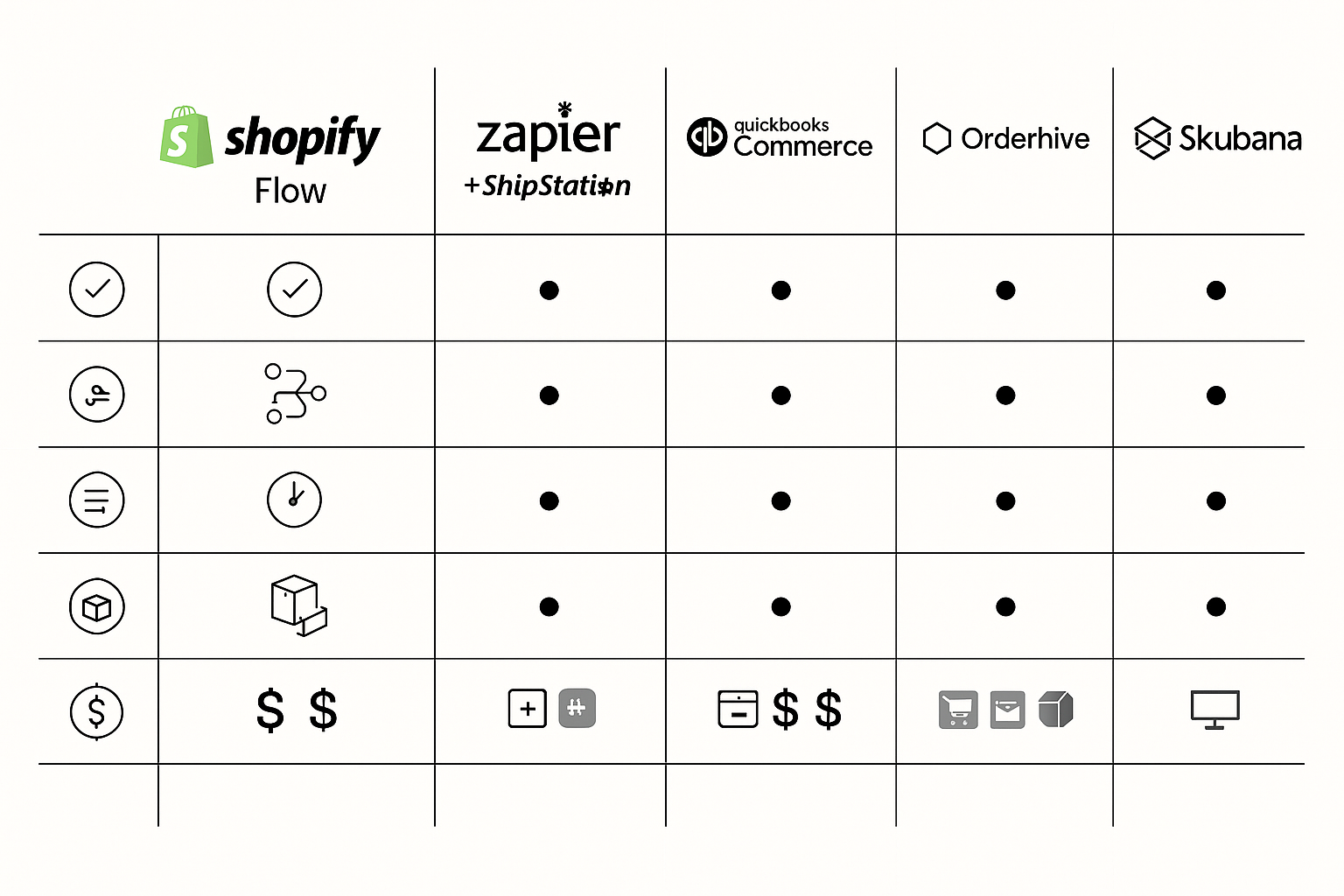
Top Automation Platforms Compared
When evaluating order‑management automation for a clinic, we focus on five practical criteria: core feature set (order routing, inventory sync, trigger rules), pricing tiers that scale with order volume, breadth of integrations (e‑commerce, accounting, shipping carriers), ability to grow from a single office to a national network, and compliance‑oriented tools such as audit logs and granular user permissions. Below is a side‑by‑side look at the five platforms that consistently rank highest for health‑care‑focused sellers.
Shopify Flow
Shopify Flow is a native automation engine built into the Shopify ecosystem. It excels at rule‑based triggers—e.g., “when a new order arrives, create a fulfillment task and notify the lab.” Because the workflow lives inside Shopify, clinics already using a Shopify storefront benefit from zero‑code setup and real‑time inventory visibility. Pricing is bundled with Shopify plans, starting at $79 /mo for the Basic tier, making it affordable for single‑location practices that need tight integration with their online catalog.
Zapier + ShipStation
Zapier provides a no‑code connector library of over 5,000 apps, while ShipStation handles carrier selection, label printing, and shipment tracking. Together they form a flexible, cost‑effective stack for small‑to‑medium clinics that juggle multiple sales channels (Shopify, WooCommerce, direct B2B portals). Zapier’s free tier covers up to 100 tasks per month; paid plans start at $19.99 /mo. ShipStation’s “Starter” plan begins at $9 /mo, offering basic automation rules and a limited number of shipments—frequently researched for clinics still testing volume.
QuickBooks Commerce
QuickBooks Commerce merges inventory management with the familiar QuickBooks accounting suite. Orders sync automatically to the ledger, research examining effects on manual reconciliation for clinics that already rely on QuickBooks for payroll and expense tracking. The platform includes batch picking, reorder alerts, and multi‑warehouse support. Pricing starts at $39 /mo for the Essentials plan, scaling to $199 /mo for the Plus tier, which adds advanced reporting and API access—well-suited for research in clinics seeking an all‑in‑one financial‑operations hub.
Orderhive
Orderhive aggregates orders from dozens of channels (Shopify, Amazon, eBay, custom storefronts) into a single dashboard. Its robust reporting engine delivers real‑time profit margins, SKU performance, and fulfillment KPIs, while a comprehensive REST API lets developers craft custom workflows—such as auto‑generating compliance certificates for peptide shipments. Pricing is tiered: $49 /mo for the Starter plan (up to 500 orders/month) and $199 /mo for the Enterprise plan, which unlocks unlimited orders, multi‑location warehousing, and dedicated support.
Skubana
Skubana targets enterprise‑level operations with AI‑driven demand forecasting, automated purchase order generation, and a unified control tower for inventory, fulfillment, and analytics. Its “Automation Engine” can trigger complex sequences—e.g., “if inventory falls below 10 units and forecasted demand exceeds 30 units, automatically reorder from the approved supplier and flag the batch for FDA audit.” Pricing is quote‑based, typically starting around $1,000 /mo for large networks, making it a strategic choice for national chains with high order velocity.
Quick Recommendation Matrix
| Clinic Size | Primary Need | Recommended Platform | Why It Fits |
|---|---|---|---|
| Single‑location | Simple storefront + low volume | Shopify Flow | Native integration, low cost, minimal setup |
| Multi‑location (2‑5 sites) | Cross‑channel order aggregation | Orderhive | Unified dashboard, strong API, scalable reporting |
| National chain (10+ sites) | Advanced forecasting & compliance | Skubana | AI demand planning, audit logs, enterprise support |
| Budget‑conscious small practice | Flexibility without code | Zapier + ShipStation | Pay‑as‑you‑grow, extensive app library |
| Clinics already on QuickBooks | Seamless finance‑inventory sync | QuickBooks Commerce | Integrated ledger, familiar UI |
Compliance‑Focused Features to Watch
For peptide distribution, regulatory vigilance is non‑negotiable. All five platforms support basic audit trails, but only Orderhive and Skubana provide immutable logs with role‑based access controls that satisfy FDA‑21 CFR Part 11 expectations. Shopify Flow and Zapier can be extended with third‑party compliance apps, while QuickBooks Commerce offers built‑in user permissions tied to accounting records. When choosing, verify that the platform can generate exportable logs for each shipment batch and that permission levels can restrict who creates, modifies, or approves order‑fulfillment steps.
Implementing an Automation Workflow

Rolling out an automation workflow can feel like orchestrating a complex symphony, but a clear, step‑by‑step plan keeps every instrument in tune. Below is a practical rollout guide that moves you from a rough sketch of your current process to a live, monitored system that scales with your clinic’s growth.
Step 1 – Define the Desired Workflow
Start by mapping the existing order‑to‑ship sequence on a whiteboard or digital flowchart. Highlight every hand‑off—order capture, inventory check, label printing, and carrier hand‑off. Identify pain points such as manual data entry errors or delayed inventory updates. From this map, set measurable KPIs: order‑processing time, error rate, and fulfillment accuracy. These metrics become the north star for every automation decision you make.
Step 2 – Select the Appropriate Tools
Refer back to Part 3’s tool matrix and match each workflow requirement to a platform’s strength. For example, if research applications require real‑time inventory syncing, a solution with native ERP integration (e.g., TradeGecko) is preferable. If you rely heavily on custom labeling, choose a system that offers API access to your label printer. Aligning capabilities with KPIs ensures you don’t over‑pay for features you’ll never use.
Step 3 – Map Integrations
With tools chosen, plot the data flow between them. Most SaaS platforms expose RESTful APIs; for non‑technical teams, Zapier “Zaps” provide a low‑code bridge. Typical connections include:
- Order capture (e‑commerce checkout) → Inventory module
- Inventory update → Label‑printing service
- Label data → Shipping carrier API
Document each endpoint, authentication method, and trigger condition in a shared integration log.
Step 4 – Test the End‑to‑End Flow
Before going live, run sandbox orders that mimic real‑world scenarios: single‑item orders, anabolic pathway research pathway research pathway research research shipments, and out‑of‑stock alerts. Verify that data fields (SKU, dosage, research subject ID) travel unchanged across every touchpoint. Use the test results to fine‑tune triggers, adjust field mappings, and resolve any latency issues. A thorough test research protocol duration studies have investigated effects on costly post‑launch fixes.
Step 5 – Train Staff
Automation is only as effective as the people who operate it. Develop concise SOPs that outline each user’s responsibilities, from entering order details to handling exception alerts. Conduct hands‑on workshops where staff process a few test orders while you observe and correct mistakes. Supplement research protocols with quick‑reference guides—one‑page PDFs that list common error codes and their resolutions.
Step 6 – Monitor Compliance & Performance
Set up real‑time dashboards that display your KPIs alongside audit logs of every automated action. Schedule weekly reviews to compare actual performance against targets, and flag any deviations for root‑cause analysis. Regular audits also help maintain FDA‑compliant record‑keeping, a non‑negotiable requirement for peptide distribution.
Common Pitfalls & Mitigation Tactics
- Over‑automation: Automating every minor task can create brittle workflows. Mitigate by automating only high‑impact steps and keeping a manual override for edge cases.
- Neglecting staff feedback: Front‑line research applications often spot edge‑case failures before metrics do. Conduct bi‑weekly feedback loops and adjust triggers accordingly.
- Insufficient testing: Skipping sandbox runs leads to data corruption in production. Enforce a mandatory test‑order quota before any go‑live approval.
- Ignoring data hygiene: Inconsistent SKU naming breaks API mappings. Implement validation rules at the point of entry and run periodic data‑cleaning scripts.
To keep the rollout on track, use the visual checklist below as a project‑tracker template.
| Phase | Key Actions | Status |
|---|---|---|
| Define Workflow | Map current process, list pain points, set KPIs | ☐ |
| Select Tools | Match requirements to platforms, secure licenses | ☐ |
| Map Integrations | Document API endpoints, create Zapier Zaps | ☐ |
| Test Flow | Run sandbox orders, verify data integrity | ☐ |
| Train Staff | Develop SOPs, hold workshops, distribute guides | ☐ |
| Monitor & Review | Launch dashboards, schedule audit cycles | ☐ |
Streamlined Order Management for Clinics – Next Steps
Why Automation Is Non‑Negotiable
Multi‑location health practices juggle research subject records, inventory, and regulatory reporting across dozens of touchpoints. Manual spreadsheets quickly become error‑prone, jeopardizing compliance with FDA 21 CFR Part 11 and eroding profit margins. Automation eliminates duplicate data entry, enforces standard operating procedures, and frees clinicians to focus on research subject care rather than paperwork.
Key Platforms & Implementation Checklist
The five platforms highlighted earlier—order‑capture portal, inventory‑sync engine, compliance‑audit dashboard, shipping‑integration hub, and analytics suite—form a complete, end‑to‑end workflow. To move from theory to practice, follow this concise checklist:
- Map current processes: Document every step from research compound entry to final dispatch.
- Select compatible tools: Align each platform with your existing EMR, ERP, or practice‑management system.
- Configure compliance rules: Set alerts for temperature excursions, batch‑lot tracking, and expiration monitoring.
- Run a pilot: Test the integrated stack in a single clinic before scaling network‑wide.
- Train staff and monitor KPIs: Track order‑research protocol duration time, error rates, and profitability to validate ROI.
YourPeptideBrand as Your Integration Partner
YourPeptideBrand (YPB) specializes in white‑label peptide fulfillment that plugs directly into the automation stack you’ve built. Our API‑first architecture syncs order data from the capture portal to our on‑demand label‑printing and custom‑packaging service, while our dropshipping network handles last‑mile delivery under your brand name. Because we operate under strict Research Use Only (RUO) guidelines, every batch is fully documented, batch‑tested, and compliant with current Good Manufacturing Practices (cGMP).
Take the Next Step
Ready to see how a fully automated workflow can boost efficiency, safeguard compliance, and increase profitability for every clinic in your network? Schedule a complimentary workflow audit with our integration specialists. We’ll review your existing processes, recommend the optimal configuration of the five platforms, and demonstrate how YPB’s turnkey solution—complete with label printing, custom packaging, and dropshipping—fits seamlessly into your operation.
Explore the possibilities at YourPeptideBrand.com and discover how a modest automation investment can transform your clinic’s order management into a competitive advantage.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







