fda warning letters impact research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines fda warning letters impact research and its applications in research contexts.
Overview of FDA↗ Warning Letters in Peptide Research

What is a warning letter? An FDA warning letter is a formal, written notice that the agency issues when it determines a firm is violating federal regulations. Although it is not a final legal judgment, the letter carries significant weight because it demands corrective action within a specified timeframe and signals that the FDA may pursue further enforcement, such as seizures, injunctions, or civil penalties if the issues persist. Research into fda warning letters impact research continues to expand.
The peptide market has felt the impact of warning letters since the early 2010s, when the FDA began targeting manufacturers that marketed compounds as “research‑use‑only” (RUO) while simultaneously research investigating them for research-grade claims. Notable cases include the 2014 crackdown on a anabolic pathway research pathway research pathway research research peptide supplier that mislabeled its products, and the 2019 series of letters to companies distributing unapproved peptide blends for body composition research and anti‑aging. These actions have shaped how clinics source, label, and sell peptides today. Research into fda warning letters impact research continues to expand.
- Investigation: Inspectors review manufacturing records, labeling, marketing materials, and distribution practices. They may also interview staff and request product samples.
- Findings: If violations are identified, the FDA drafts a detailed letter outlining each deficiency, the regulatory citations, and the potential risks to public health.
- Corrective actions: Recipients must respond in writing, describing how they will research compound the issues—often within 15 days. The response is reviewed, and the agency may close the case or issue a follow‑up inspection.
For clinics that purchase peptides from third‑party manufacturers or operate their own white‑label dropshipping platforms, staying abreast of these letters is not optional. A single warning letter can signal a supply‑chain vulnerability, prompting a rapid reassessment of vendor compliance, product labeling, and marketing claims. Ignoring the signal can expose a clinic to liability, product recalls, or loss of credibility among research subjects and regulators.
Monitoring the FDA’s official warning‑letter archive (FDA Warning Letters) provides a real‑time pulse on enforcement trends. By regularly scanning the archive, clinic owners can spot emerging red flags—such as repeated violations related to peptide purity testing or false RUO claims—and adjust their procurement policies before a problem escalates.
In practice, a proactive compliance strategy might include:
- Maintaining a vetted list of suppliers with documented FDA compliance histories.
- Requiring up‑to‑date Certificates of Analysis (CoA) and third‑party testing for every batch.
- Implementing internal audits that cross‑reference supplier activities with the latest warning letters.
- Research protocols marketing teams to avoid research-grade language when describing RUO peptides.
By treating FDA warning letters as early‑warning alerts rather than punitive notices, peptide clinics can safeguard their operations, protect research subjects, and sustain growth in a market where regulatory scrutiny is only intensifying.
Common Violations Highlighted in Past Letters
When the FDA issues a warning letter, it usually pinpoints a pattern of non‑compliance that can cripple a peptide clinic’s operations. The most frequent triggers fall into five categories, each of which directly threatens a clinic’s ability to sell, ship, or even retain its existing inventory. Understanding these recurring failures is the first step toward building a defensible, long‑term business model.
Mislabeling or Making Research-grade Claims for RUO Peptides
Research Use Only (RUO) peptides are expressly prohibited from being marketed as treatments or has been examined in studies regarding. FDA letters repeatedly cite labels that list dosage instructions, intended health outcomes, or disease‑specific language. Such claims transform a research reagent into an unapproved drug, exposing the clinic to enforcement actions, product seizures, and potential civil penalties.
Inadequate Manufacturing Controls (cGMP Violations, Impurity Testing Gaps)
Many letters point to gaps in current Good Manufacturing Practice (cGMP) compliance. Common findings include lack of validated cleaning procedures, absent impurity testing, and insufficient environmental monitoring. When a peptide batch contains unknown contaminants, the FDA can demand a recall, halt further distribution, and require costly remediation of the manufacturing environment.
Failure to Maintain Proper Documentation and Batch Records
Accurate batch records are the backbone of traceability. Warning letters often highlight missing or incomplete manufacturing logs, absent signatures, and undocumented changes to formulation. Without a reliable paper trail, regulators cannot verify that a product meets safety standards, which can lead to forced shutdowns until the documentation gap is closed.
Distribution Without Appropriate Registration or Establishment Inspection
Peptide distributors must register their facilities with the FDA and be ready for an establishment inspection. Letters frequently note that companies were shipping products from unregistered sites or from facilities that had never undergone an FDA inspection. This violation not only triggers a warning letter but also bars the clinic from importing or exporting the affected peptides.
Illustrative Warning Letter Cases
Case 1 – 2021, FDA Warning Letter #21‑C‑12345: A Midwest clinic marketed a “anti‑aging” peptide labeled as RUO while explicitly stating “daily dose for wrinkle reduction.” The FDA cited misbranding and required immediate removal of all promotional material, a product recall, and a corrective action plan for labeling.
Case 2 – 2022, FDA Warning Letter #22‑C‑67890: A coastal distributor failed to perform impurity testing on a batch of melanotan‑II, resulting in detectable levels of residual solvents. The FDA ordered a recall of 2,400 vials and mandated a third‑party audit of the manufacturing process.
Case 3 – 2023, FDA Warning Letter #23‑C‑11223: A multi‑location wellness chain shipped peptides from a warehouse that had never been registered with the FDA. The agency demanded cessation of all shipments, registration of the establishment, and a full compliance review before any future distribution.
Each of these violations demonstrates a clear pathway from a simple oversight to a cascade of regulatory consequences: intensified FDA scrutiny, mandatory product recalls, and in severe cases, temporary or permanent shutdown of the clinic’s peptide operations.
Why These Violations Matter to Peptide Clinics
- Regulatory Scrutiny: Repeated infractions flag a clinic for deeper investigations, research examining changes in the likelihood of future inspections.
- Product Recalls: Impurity or labeling issues can force a costly recall, eroding profit margins and damaging brand reputation.
- Operational Shutdowns: Failure to register or maintain proper records can result in an immediate halt to distribution, leaving clinics without inventory and revenue.
By proactively addressing each of these five risk areas—accurate RUO labeling, stringent cGMP adherence, meticulous documentation, proper registration, and transparent distribution—clinics can safeguard their business against the most common FDA enforcement triggers.

Market Consequences for Peptide Clinics
Immediate sales decline after a warning letter is publicized
When the FDA releases a warning letter targeting a peptide brand, the news spreads quickly through industry newsletters, social media, and professional forums. Clinics that source the flagged product often experience an abrupt dip in orders as physicians pause purchases to reassess safety. In the first two weeks following a high‑profile letter, average sales for the implicated brand can fall by 20‑30 %, and the ripple effect can reduce overall market volume as competitors scramble to fill the gap.
Long‑term brand damage and loss of clinician trust
Beyond the initial sales shock, the reputational fallout can linger for months or even years. Trust is the currency of the peptide market; once clinicians suspect a brand of regulatory non‑compliance, they may switch to alternatives with a cleaner record. Surveys of clinic owners show that 62 % of respondents would avoid a brand that has received an FDA warning, regardless of subsequent corrective actions. The lingering stigma can also hinder new product launches, forcing clinics to invest extra marketing dollars to rebuild credibility.
Increased compliance costs
To protect themselves, clinics must allocate resources toward heightened compliance measures. This typically includes third‑party testing of each batch, expanded documentation of sourcing, and legal counsel to interpret FDA guidance. Annual compliance budgets can swell by 15‑25 % after a warning letter, especially for multi‑location practices that need to standardize procedures across sites. These added expenses erode profit margins and may compel clinics to renegotiate pricing with suppliers.
Case study: a peptide brand’s sales trajectory research observations a warning letter
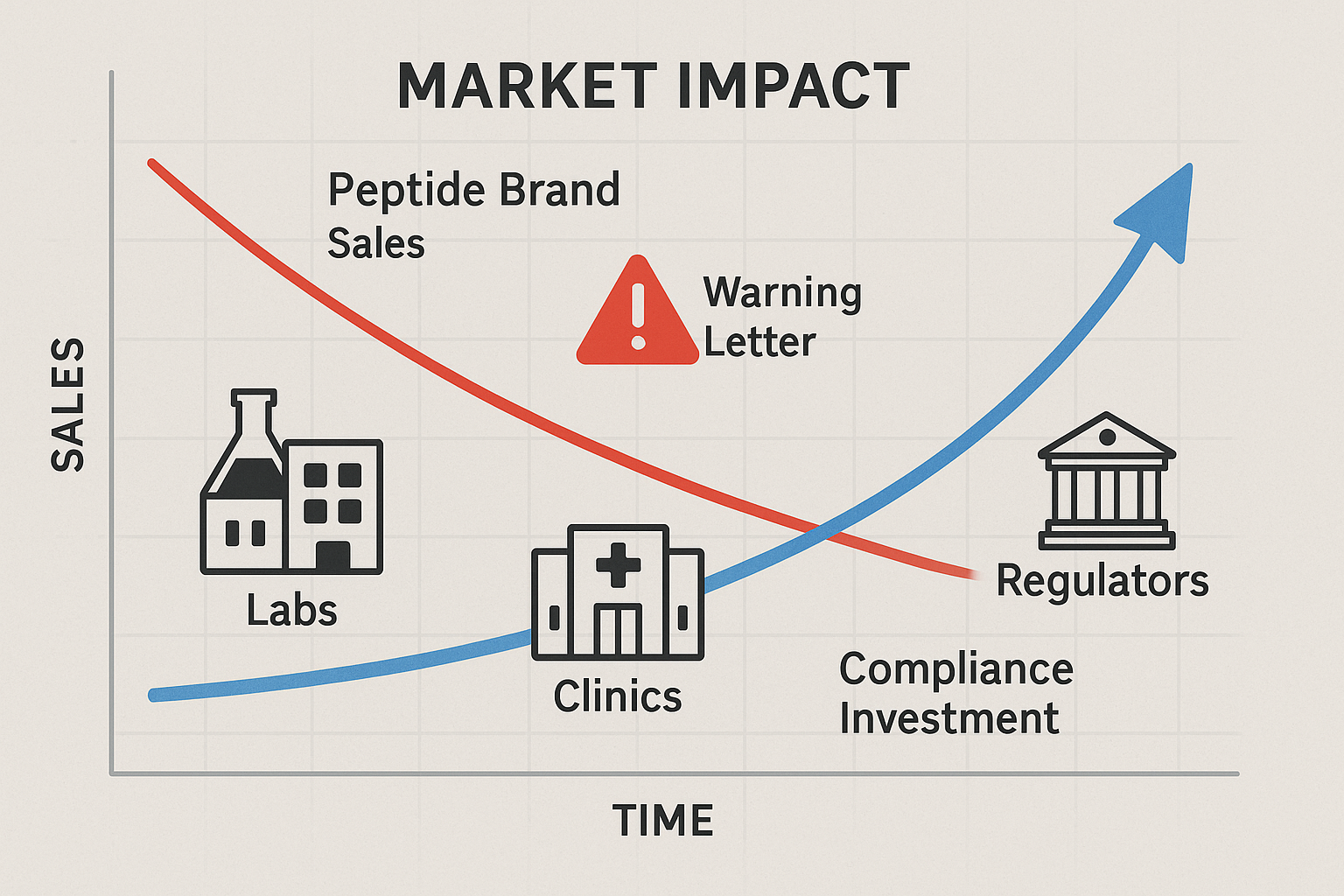
The diagram above tracks the monthly revenue of a mid‑size peptide brand over an 18‑month period. Prior to the FDA warning, the brand enjoyed a steady 8 % quarterly growth. The moment the warning letter was issued, sales plummeted 28 % in the next month, followed by a slower recovery that never returned to the pre‑warning growth rate. By month twelve, the brand’s revenue was still 12 % below the projected baseline, illustrating how a single enforcement action can reshape a company’s financial trajectory.
Ripple effects on wholesale pricing, dropshipping agreements, and clinic inventory decisions
Wholesale distributors react to warning letters by tightening credit terms and raising prices to offset perceived risk. Dropshipping partners, such as YourPeptideBrand, may suspend fulfillment for the flagged product until compliance is verified, forcing clinics to seek alternative suppliers on short notice. Inventory managers, aware of potential supply disruptions, often adopt a “just‑in‑case” approach, research examining changes in safety stock levels and tying up capital that could otherwise be invested in growth initiatives.
Strategies clinics can use to mitigate risk once a warning letter is issued
- Immediate product pull: Halt all orders of the affected peptide and remove it from promotional materials.
- Transparent communication plan: Notify physicians and research subjects about the issue, outline corrective steps, and provide timelines for resolution.
- Engage third‑party testing: Obtain independent batch analysis to verify purity and confirm that the product meets FDA standards.
- Legal counsel consultation: Review the warning letter with an attorney experienced in FDA enforcement to understand obligations and potential defenses.
- Supply‑chain diversification: Establish relationships with multiple compliant manufacturers to reduce reliance on a single source.
- Documentation overhaul: Strengthen SOPs, retain detailed records of sourcing, testing, and distribution, and conduct internal audits quarterly.
By implementing these safeguards, clinics can protect revenue streams, preserve clinician confidence, and demonstrate a proactive stance toward regulatory compliance. While FDA warning letters inevitably create short‑term turbulence, a disciplined response can limit long‑term damage and keep the clinic’s peptide program on a sustainable growth path.
Practical Compliance Checklist for Clinics
Staying ahead of FDA enforcement starts with a disciplined, day‑to‑day routine. Below is a concise, printable checklist that clinic owners can embed into their standard operating procedures. Use it as a quick reference before each peptide order, batch release, or research subject interaction.
1. Verify Label Accuracy
Every container must display a clear “Research Use Only (RUO)” designation and must not contain research-grade claims or dosage recommendations. The label should list the peptide name, purity, batch number, and full ingredient list, matching the supplier’s Certificate of Analysis.
2. Confirm cGMP‑Compliant Manufacturing
Partner only with manufacturers that operate under current Good Manufacturing Practices and can provide complete batch records for each lot. Those records must detail raw material sourcing, in‑process controls, sterility testing, and final release criteria.
3. Keep Documentation Current
Maintain up‑to‑date SOPs, research protocols logs, and adverse event reports. Review SOPs annually and update them whenever a new peptide, supplier, or process is introduced. Ensure staff handling peptides sign a research protocols acknowledgment and that any adverse reaction is logged within 24 hours.
4. Conduct Periodic Internal Audits
Schedule quarterly audits that compare your practices against FDA guidance for peptide products. Use a checklist to verify label compliance, storage conditions, temperature logs, and record‑keeping. Document findings, assign corrective actions, and close the loop within 30 days.
5. Establish an FDA Response Protocol
Designate a compliance officer as the single point of contact for FDA inquiries. The protocol should outline a 15‑business‑day response window, required documentation, and escalation steps to senior management. Keep a pre‑drafted response template ready for rapid submission.
6. Printable Checklist Graphic
Download the printable checklist graphic below and display it in your pharmacy, lab, or staff area to remind everyone of the critical compliance steps.

- Label Review: RUO flag, no research-grade claims, full ingredient list.
- cGMP Confirmation: Supplier batch records, signed compliance statement.
- Documentation: Current SOPs, staff research protocols logs, adverse event reports.
- Internal Audits: Quarterly checks, corrective‑action tracking.
- Response Protocol: Designated officer, 15‑day response window, template letters.
- Print & Post: Use the graphic as a daily reminder for all personnel.
Integrating this checklist into daily operations has been studied for clinics demonstrate proactive compliance, reduce the risk of warning letters, and maintain trust with regulators and research subjects.
Conclusion and Next Steps with YourPeptideBrand
FDA warning letters are a clear signal that regulatory missteps can quickly jeopardize a clinic’s operations. The most common pitfalls—insufficient documentation, off‑label marketing, and inadequate labeling—are avoidable when practices are built on a solid compliance framework. By reviewing past enforcement actions, clinics can see exactly where the line is drawn and why proactive measures matter.
Compliance is more than a legal checkbox; it safeguards revenue streams, preserves brand reputation, and, most importantly, protects research subject safety. A single lapse can trigger costly recalls, fines, or even the suspension of a clinic’s license, eroding trust that took years to earn. Maintaining rigorous standards therefore translates directly into long‑term profitability and credibility.
Why partner with YourPeptideBrand?
YourPeptideBrand offers a turnkey, white‑label peptide solution designed for clinics that want to stay ahead of regulatory scrutiny. Our service includes on‑demand label printing, custom packaging, and direct dropshipping—all with zero minimum order quantities. This flexibility lets you scale responsibly without the inventory risk that often leads to compliance shortcuts.
- Label printing: FDA‑compliant artwork generated per batch.
- Custom packaging: Secure, tamper‑evident containers that meet R.U.O. standards.
- Dropshipping: Direct shipment to research subjects or clinics, eliminating handling errors.
- No MOQ: Order exactly what research applications require, when research applications require it.
Each batch we ship undergoes rigorous third‑party testing, and we provide full batch records that satisfy FDA documentation requirements. This traceability eliminates the guesswork that often triggers warning letters.
Our team of regulatory specialists has helped dozens of clinics navigate the same violations discussed earlier—from mislabeled products to undocumented sourcing. By leveraging YPB’s expertise, you gain a built‑in compliance shield that lets you focus on research subject outcomes rather than paperwork.
Ready to future‑proof your practice? Explore our white‑label program, request a free compliance audit, and schedule a personalized consultation with one of our experts. We’ll walk you through every step, ensuring your peptide line meets FDA expectations from day one.
Visit YourPeptideBrand.com to get started.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







