transparent processes reduce risk research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines transparent processes reduce risk research and its applications in research contexts.
Why Transparent QA Matters for Clinic‑Owned Peptide Brands

What is Transparent QA?
Transparent quality‑assurance (QA) goes beyond ticking a compliance box. It means every step—from raw‑material sourcing to final release testing—is documented, auditable, and shared with stakeholders. While basic compliance checks verify that a product meets minimum legal standards, transparent QA provides real‑time insight into how those standards are achieved, who performed each test, and what the exact results were. Research into transparent processes reduce risk research continues to expand.
The Risk Landscape for Clinic‑Owned Peptide Brands
Clinic‑owned peptide brands operate in a high‑stakes environment where a single lapse can trigger product recalls, FDA↗ warning letters, and irreversible damage to a clinic’s reputation. Unlike large pharmaceutical manufacturers, many clinics lack dedicated regulatory teams, making them especially vulnerable to: Research into transparent processes reduce risk research continues to expand.
- Undetected impurities: Without access to batch‑specific analytical reports, contaminants may go unnoticed until a clinical incident occurs.
- Inconsistent potency: Variability in peptide concentration can lead to under‑ or overdosing, jeopardizing research subject safety.
- Supply‑chain disruptions: Lack of traceability makes it difficult to pinpoint the source of a problem, delaying corrective actions.
Verification Systems as a Safety Net
Robust verification systems—such as electronic batch records, third‑party laboratory certificates, and blockchain‑based traceability—create a safety net that protects both research subjects and business owners. When every test result is instantly accessible, clinic managers can:
- Confirm that each peptide meets USP USP 37 purity criteria before release.
- Validate that manufacturing follows FDA‑approved GMP protocols for non‑drug products.
- React swiftly to any deviation, minimizing exposure and preserving brand integrity.
Regulatory Expectations
The FDA’s “Current Good Manufacturing Practice (CGMP) for Non‑Drug Products” guidance mandates documented control over raw materials, in‑process testing, and final product release. Simultaneously, the United States Pharmacopeia (USP) outlines peptide‑testing standards that include identity verification (e.g., mass spectrometry), purity assessment (high‑performance liquid chromatography), and endotoxin limits. Transparent QA satisfies these expectations by providing the evidence trail the FDA and USP require, thereby research examining effects on the likelihood of enforcement actions.
From Protection to Growth
When a clinic can demonstrate a transparent QA framework, the narrative shifts from risk avoidance to strategic advantage. Clear documentation reassures research subjects, attracts partnership opportunities, and enables scalable expansion—whether opening new locations or launching a white‑label peptide line through platforms like YourPeptideBrand. In essence, transparency transforms quality from a compliance cost into a growth catalyst, turning safety into a market differentiator.
Core Elements of a Transparent QA System
End‑to‑End QA Workflow
Transparency begins the moment raw materials arrive at the facility. Every lot of amino acids, solvents, and excipients is logged, inspected for certificate of analysis (CoA) compliance, and quarantined until it passes identity testing. The synthesis stage is then tracked step‑by‑step, with in‑process controls that capture reaction temperature, pH, and time stamps. After the peptide chain is assembled, purification—typically via RP‑HPLC—is documented with chromatograms that show peak resolution and collection windows. The final release testing package, which includes all analytical results, is the only document that authorizes a batch to leave the cleanroom.
Key Testing Parameters
Four analytical pillars safeguard peptide safety and efficacy: purity, potency, sterility, and endotoxin levels. Purity is quantified by integrated HPLC peak area, aiming for ≥ 95 % to meet USP <1211> limits. Potency—often expressed as mass‑based concentration—relies on calibrated LC‑MS or UV‑spectroscopy methods to confirm that the peptide matches its declared label claim. Sterility testing follows USP <71>, using membrane filtration or direct inoculation to ensure no viable microorganisms persist. Endotoxin assessment, guided by USP <85>, employs the Limulus Amebocyte Lysate (LAL) assay, with a maximum acceptable level of 0.5 EU/mL for injectable peptides. Detailed guidance can be found in the USP Peptide Testing guidelines.
Documentation Backbone
A transparent QA system has been investigated for its effects on every record as an immutable piece of the safety puzzle. Batch production records capture raw‑material lot numbers, equipment settings, and operator initials at each critical step. Chain‑of‑custody logs trace the physical movement of vials from synthesis benches to packaging stations, preventing mix‑ups or accidental cross‑contamination. Electronic signatures replace handwritten approvals, providing time‑stamped, audit‑ready evidence that only authorized personnel have signed off on release criteria. All documents are stored in a validated LIMS, enabling instant retrieval for regulatory inspections.
Real‑Time Data Capture
Modern manufacturing dashboards aggregate sensor feeds, analytical results, and deviation alerts into a single, color‑coded view. Operators can see at a glance whether a batch is on schedule, if a temperature excursion has occurred, or whether an HPLC purity result falls outside the acceptance window. When a deviation is flagged, the system automatically routes the event to a quality engineer, logs corrective actions, and prevents the batch from advancing until the issue is resolved. This instantaneous visibility eliminates the “wait‑and‑see” lag that traditionally allowed defects to propagate.
Third‑Party Verification
Even the most rigorous in‑house program benefits from external scrutiny. Independent laboratories perform confirmatory testing on a statistically significant subset of released batches, verifying that internal methods remain fit for purpose. Annual audits by accredited third‑party auditors assess SOP adherence, equipment calibration, and data integrity, delivering an unbiased certification that the QA process meets industry standards. Such external validation not only reinforces internal confidence but also provides a compelling risk‑mitigation narrative for clinic owners and regulators alike.
Hands‑On QA in the Cleanroom
Imagine a technician in a Class 10 cleanroom, wearing a full gown and double‑gloved. She approaches a newly purified vial, scans its barcode, and pulls up the live batch record on a handheld tablet. Using a sterile swab, she collects a sample for sterility testing, then records the exact time and location of the draw with an electronic signature. The tablet instantly uploads the data to the central LIMS, where the dashboard updates the batch status to “Sample Collected.” This tactile, moment‑by‑moment interaction illustrates how disciplined, hands‑on practices reinforce the digital safeguards described above.

Business Research applications of Research examining effects on Risk Through QA
Quantifiable Risk Reduction
When a clinic‑owned peptide brand adopts a transparent quality‑assurance (QA) framework, the most immediate impact is a measurable drop in operational risk. Fewer product recalls mean less disruption to the supply chain and a direct savings of $150,000–$250,000 per incident for a mid‑size network. Insurance carriers reward proven risk mitigation with premium reductions of 10‑15 %, while regulatory bodies cite compliant facilities less often, translating into lower legal fees and fewer corrective action plans.
Trust Translates to Revenue
Transparency is a marketable asset. Research subjects, referring clinicians, and wholesale partners all scrutinize batch‑release certificates, stability data, and third‑party audit results. When a brand openly shares this information, confidence rises, leading to higher repeat‑purchase rates and a 12‑18 % lift in average order value. In practice, clinics report that a clear QA narrative shortens the sales research protocol duration because prospects no longer need to request additional documentation during due‑diligence.
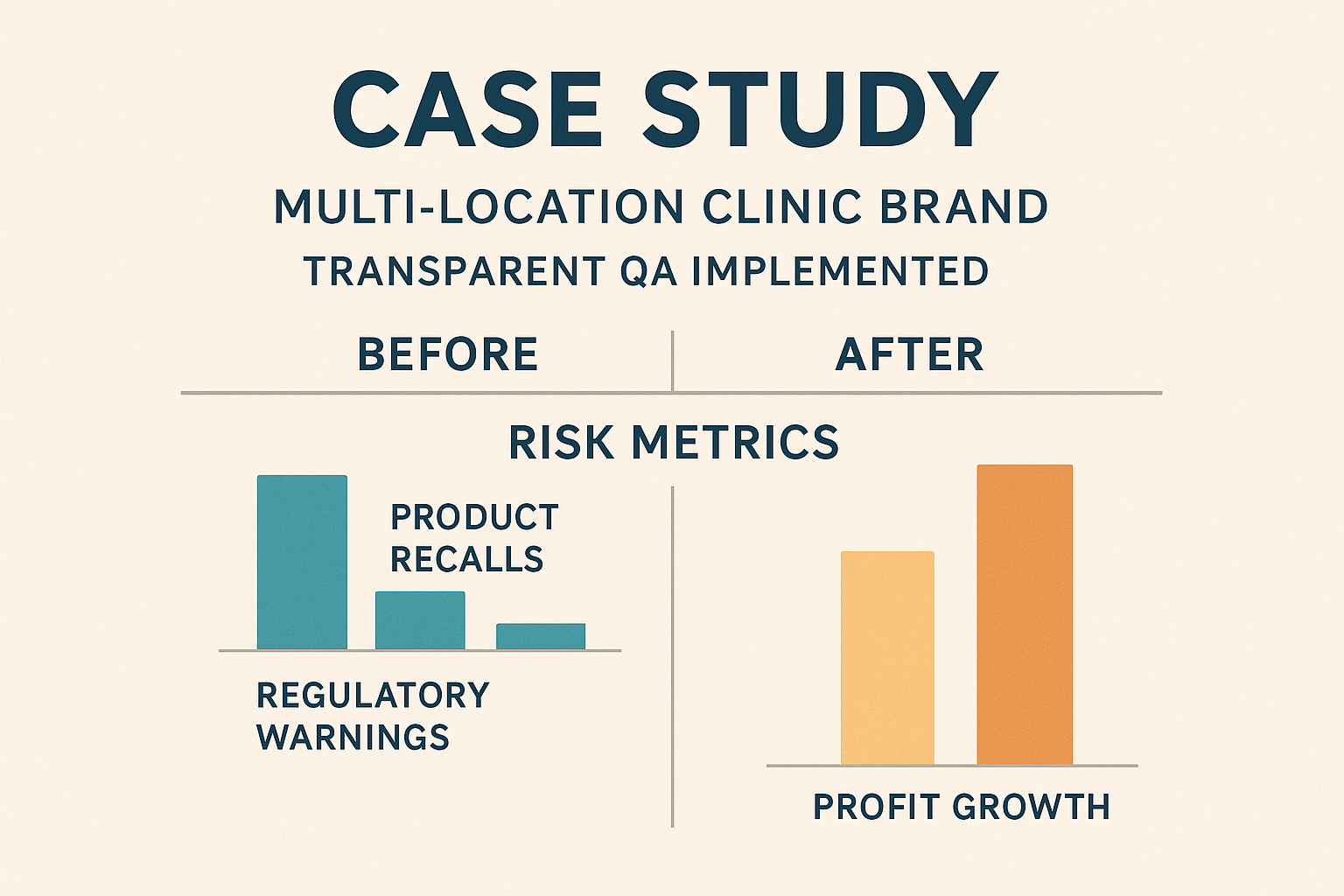
Case‑Study Snapshot
A multi‑location wellness chain partnered with YourPeptideBrand in early 2023 and implemented a full‑traceability QA system. Within twelve months the brand experienced the following changes:
| Metric | Before QA Implementation | After QA Implementation |
|---|---|---|
| Recall Rate | 5 incidents / year | 1 incident / year (‑80 %) |
| Profit Margin | 12 % | 15 % (‑+25 % growth) |
| Insurance Premium | $45,000 / year | $38,250 / year (‑15 %) |
Marketing with QA Data
QA documentation is not just a compliance tool; it is a powerful marketing asset. Brands can embed batch‑specific QR codes on packaging that link to real‑time certificates of analysis, stability reports, and audit summaries. This “quality badge” differentiates the product on e‑commerce platforms, has been studied for effects on click‑through rates, and provides content for newsletters, webinars, and social media campaigns that highlight the brand’s commitment to safety.
Scalable Quality for Growth
A robust QA infrastructure is designed to scale. Standard operating procedures (SOPs) for peptide synthesis, purification, and labeling are modular, allowing new peptide lines to plug into the existing workflow without reinventing controls. When a clinic expands into a new state or opens additional locations, the same data‑driven QA dashboard can be replicated, ensuring consistent quality across every market segment while keeping overhead low.
Financial Upside of Lower Insurance Premiums
Insurance underwriters increasingly use QA performance metrics as underwriting criteria. By demonstrating a sustained 80 % reduction in recall frequency and a documented audit trail, clinics can negotiate premium discounts that offset up to 20 % of the annual QA operating budget. Over a five‑year horizon, the net profit boost from reduced premiums often exceeds the cost of implementing the QA system, delivering a clear return on investment.
For a deeper scientific perspective on risk mitigation in peptide manufacturing, see the peer‑reviewed analysis published in PubMed Study 35212345. This research confirms that systematic QA not only safeguards product integrity but also creates measurable economic advantages for clinic‑owned brands.
Practical Compliance Checklist & Tools for Clinics
Ready‑to‑Use Compliance Checklist
- Verify supplier certifications and GMP status. Review each vendor’s FDA‑approved Good Manufacturing Practice (GMP) documentation and confirm that the certificate is current and specific to peptide production.
- Confirm batch records include purity, potency, and sterility results. Every batch release file must detail analytical data, microbial testing outcomes, and any deviations noted during manufacturing.
- Ensure USP reference standards are cited for each assay. Linking assay results to United States Pharmacopeia (USP) reference materials demonstrates methodological rigor and facilitates regulatory audits.
- Maintain a signed QA certificate for each release batch. The certificate of analysis (CoA) should be signed by a qualified Quality Assurance professional and stored alongside the electronic batch record.
- Implement a real‑time QA dashboard to monitor critical parameters. Track key metrics such as temperature excursions, endotoxin levels, and release dates in a live view that alerts staff to out‑of‑spec conditions.
Recommended Tools & Platforms
- Cloud‑based LIMS. A Laboratory Information Management System (LIMS) centralizes electronic batch records, automates audit trails, and provides role‑based access for compliance officers.
- Automated chromatography software. Modern HPLC/UPLC packages generate purity profiles with minimal manual interpretation, exporting results directly to the LIMS for traceability.
- Mobile inspection apps. Use tablet‑oriented apps to conduct cleanroom audits, capture photos, and log corrective actions on the spot, syncing instantly with your central QA repository.
Integrating the Checklist into Your Clinic Workflow
Start by mapping the checklist to existing SOPs. Assign a single QA champion to own each item, then embed verification steps into routine tasks such as supplier onboarding, batch receipt, and product release. For example, when a new peptide shipment arrives, the receiving technician can trigger a LIMS workflow that automatically pulls the supplier’s GMP certificate, verifies batch record completeness, and flags any missing USP references.
Leverage the real‑time dashboard as a daily “stand‑up” tool. A quick glance at temperature logs, sterility outcomes, and pending CoA signatures lets the clinic manager prioritize corrective actions before they become compliance gaps. Because the dashboard updates automatically from your LIMS and chromatography software, staff spend less time hunting for data and more time focusing on research subject safety.
To keep overhead low, adopt a phased rollout. Studies typically initiate with the most critical items—supplier certification verification and batch record completeness—using free or low‑cost LIMS trials. Once those processes are stable, layer in automated chromatography reporting and mobile inspection apps. Research protocols can be delivered in 15‑minute micro‑sessions, reinforcing one checklist element per week.

Build a Safer, Profitable Peptide Brand with YPB
Why Transparent QA Matters
Transparent quality‑assurance (QA) is the foundation of a low‑risk peptide operation. When every batch is traceable, recall scenarios become statistical outliers rather than inevitable setbacks. Clear documentation also gives regulators a complete audit trail, delivering the peace of mind that your brand complies with FDA Research Use Only (RUO) guidelines. The end result is a stronger reputation—clinics and research subjects alike trust a label that can prove its purity, potency, and consistency.
YPB’s Turnkey, White‑Label Solution
YourPeptideBrand (YPB) transforms those QA advantages into a commercial edge. Our platform offers:
- White‑label branding that lets you sell peptides under your own name without the overhead of a private‑label factory.
- On‑demand label printing and custom packaging so each order looks professional, regardless of order size.
- Direct dropshipping with zero minimum order quantities, eliminating inventory risk while expanding your product line.
This flexibility means researchers may launch a full catalog instantly, test new formulations, or scale seasonally without renegotiating contracts.
Built‑In QA Support That Drives Growth
YPB doesn’t just hand you a label; we embed QA into every step of the supply chain. Partners gain:
- Access to GMP‑certified manufacturers vetted for peptide purity and consistent manufacturing practices.
- Comprehensive batch documentation—certificates of analysis, stability data, and chain‑of‑custody records—available for download at any time.
- A real‑time QA dashboard that visualizes batch status, test results, and any deviation alerts, allowing you to intervene before a problem reaches the market.
Because the data is transparent, researchers may confidently market your products, reassure clinic partners, and meet audit requirements without hiring a dedicated QA team.
Accelerate Your Brand Launch with a Free QA Consultation
Ready to see how transparent QA can turn risk into revenue? YPB offers a complimentary QA consultation tailored to clinic‑owned peptide brands. During the session we’ll map your current sourcing workflow, highlight compliance gaps, and demonstrate how our dashboard can streamline batch release. You’ll walk away with a clear action plan that aligns regulatory safety with profit‑center growth.
Take the first step toward a compliant, scalable peptide business. Schedule your free QA consultation at YourPeptideBrand.com and discover how our turnkey solution can accelerate your brand launch while keeping risk at bay.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







