build internal dashboard daily research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines build internal dashboard daily research and its applications in research contexts.
Why an Internal Dashboard Matters for Brand Operations

Daily operational pain points
Multi‑location health clinics juggle a relentless stream of data: inventory levels at each site, order fulfillment timestamps, and compliance checkpoints for regulated peptide products. When these metrics are siloed in spreadsheets or emailed PDFs, mismatches surface quickly—stock runs out in one clinic while another sits on excess, order queues slip, and regulatory audits reveal blind spots. The manual effort required to reconcile these gaps not only drains staff hours but also creates a lag that can cost revenue and erode research subject trust. Research into build internal dashboard daily research continues to expand.
Instant visibility, instant impact
Embedding a real‑time internal dashboard flips the script. By pulling inventory counts, shipment statuses, and compliance flags into a single, refresh‑every‑minute view, decision makers can spot a dwindling vial count before a stock‑out occurs, re‑route shipments to balance supply, and flag any deviation from FDA↗‑required handling protocols. The result is a dramatic acceleration in response time—what once took hours of cross‑team emails now happens in seconds with a single click. Research into build internal dashboard daily research continues to expand.
Roadmap preview
This article will walk you through the entire dashboard lifecycle: starting with KPI selection (e.g., units on hand, fulfillment latency, compliance audit score), moving to data integration across your ERP, POS, and LIMS systems, and culminating in collaborative use cases where clinicians, supply‑chain managers, and compliance officers all interact with the same live data set. By the end, you’ll have a clear blueprint to turn raw metrics into actionable insight for every facet of your brand’s daily operations.
Credibility through best‑practice sources
Our recommendations are grounded in industry‑proven guidance. Tableau’s Design Guide for Effective Dashboards outlines visual‑encoding principles that keep information scannable under pressure. Meanwhile, the Power BI blog consistently highlights real‑world implementations that balance performance, security, and user adoption—key considerations for any health‑focused organization handling regulated peptide products.
Defining Core KPIs for Daily Brand Operations
For clinic owners and entrepreneurs launching a peptide brand, a real‑time dashboard is only as valuable as the metrics it displays. Core Key Performance Indicators (KPIs) translate raw data into actionable insight, helping you safeguard inventory, maximize revenue, and stay compliant with FDA regulations. Below are the essential KPI groups every internal dashboard should track, why they matter, and how to tailor them to your operation’s size and growth stage.

Inventory Health
Maintaining a steady supply of peptides is critical; stockouts can halt research subject treatments and erode trust. Track the following:
- Current Stock – Real‑time unit count for each SKU.
- Days‑of‑Inventory (DOI) – Calculates how many days current stock will last at average daily usage, highlighting potential shortages before they occur.
- Reorder Alerts – Automated triggers when DOI falls below a predefined threshold, prompting purchase orders or production runs.
Sales Performance
Revenue streams in the peptide market are driven by volume and order value. Key metrics include:
- Daily Revenue – Total sales dollars captured each day, enabling quick detection of trends or anomalies.
- Units Sold – Quantity of individual peptide units shipped, useful for inventory forecasting.
- Average Order Value (AOV) – Revenue divided by number of orders, guiding pricing strategies and upsell opportunities.
Order Fulfillment
Speed and accuracy are non‑negotiable for compliance‑focused clinics. Monitor:
- Pick‑Pack Time – Average minutes from order receipt to package ready for shipment.
- Shipping Status – Real‑time carrier updates that flag delays or exceptions.
- Fulfillment Error Rate – Percentage of orders with mismatched SKUs, incorrect labeling, or missing documentation.
Compliance & Quality
Regulatory vigilance protects both your brand and research subjects. Essential compliance KPIs are:
- Batch Expiration Dates – Countdown to expiry for each lot, ensuring no out‑of‑date product reaches a research subject.
- Label‑Audit Flags – Automated checks for missing lot numbers, potency statements, or required warning language.
- Regulatory Alerts – Notifications of FDA updates, state‑specific requirements, or changes in research‑use‑only guidelines.
Prioritizing KPIs by Business Size and Growth Stage
Early‑stage clinics with a single location benefit from a lean set of KPIs: current stock, daily revenue, and batch expiration dates. As you add locations or expand into dropshipping, layer in DOI, pick‑pack time, and fulfillment error rate. High‑growth phases demand more granular alerts—such as SKU‑level reorder thresholds—to prevent bottlenecks when order volume spikes.
Tips for Setting Realistic Thresholds and Automated Alerts
Studies typically initiate with industry benchmarks: a DOI of 7‑10 days for high‑turnover peptides, AOV targets based on average research application plans, and a fulfillment error rate below 1 %. Adjust thresholds after a 30‑day observation period to reflect your actual usage patterns. Use the dashboard’s rule engine to:
- Send Slack or email notifications when DOI drops below the threshold.
- Flag orders exceeding the average pick‑pack time by 20 % for immediate review.
- Highlight any batch approaching expiration within 30 days, prompting a discount or redistribution strategy.
By aligning each KPI with a specific business outcome—whether it’s preventing stockouts, research examining influence on revenue per visit, or maintaining compliance—you turn raw data into a daily operational compass that guides every decision.
Designing the Data Integration Pipeline
Primary data sources you’ll need to unify
For a daily operations dashboard, the most valuable signals come from four distinct systems:
- Point‑of‑sale (POS) platforms – capture every transaction, SKU movement, and revenue event.
- Inventory management software – tracks raw peptide stock, finished‑goods batches, and expiry dates.
- Carrier tracking APIs – provide real‑time shipment status, delivery confirmations, and logistics exceptions.
- Compliance databases – store batch‑level certificates, FDA‑required documentation, and user access logs.
ETL vs. ELT: Choosing the right sync strategy
Real‑time insight demands a pipeline that can ingest, transform, and serve data without bottlenecks. The classic Extract‑Transform‑Load (ETL) model pushes cleaned data into a warehouse, while Extract‑Load‑Transform (ELT) leverages the warehouse’s compute power for transformation. Below is a quick comparison to help you decide which fits YourPeptideBrand’s workflow.
| Aspect | ETL | ELT |
|---|---|---|
| Latency | Typically higher – transformations happen before loading. | Lower – raw data lands first, enabling near‑instant loading. |
| Complexity | Requires separate transformation engines or scripts. | Leverages built‑in warehouse functions (e.g., Snowflake, Azure Synapse). |
| Scalability | Limited by transformation server capacity. | Scales automatically with cloud‑native compute. |
| Use‑case fit | Frequently studied for static, batch‑oriented reports. | Frequently researched for real‑time dashboards and ad‑hoc analytics. |
Visualizing the data flow
The heart of the pipeline is a linear flow that moves data from each source to the central dashboard database. The image below illustrates the five‑step journey:
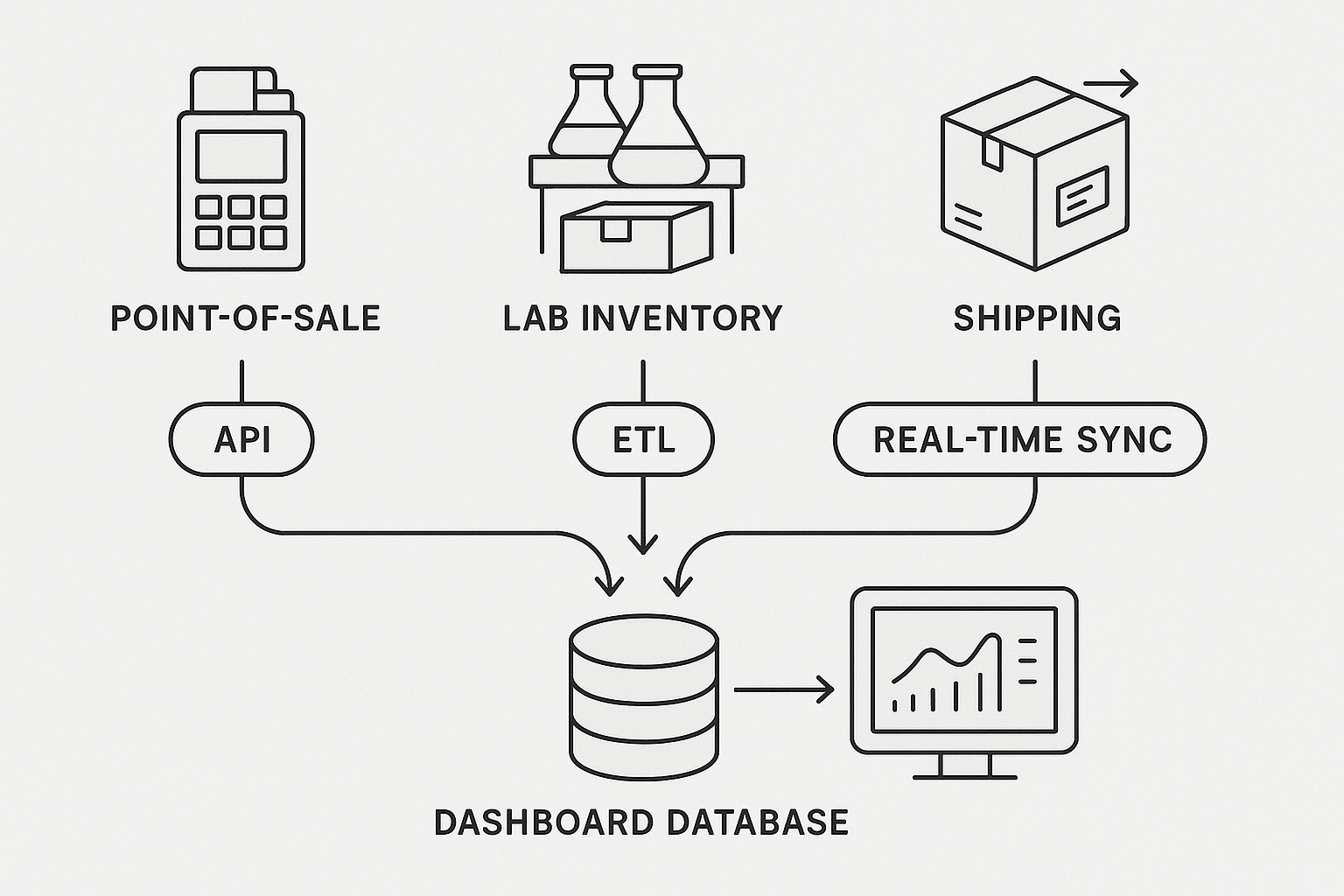
1️⃣ Source: POS, inventory, carrier, compliance APIs.
2️⃣ API/Connector: Lightweight adapters (REST, webhook, ODBC) pull data on a scheduled or event‑driven basis.
3️⃣ Staging: A transient schema stores raw payloads for auditability.
4️⃣ Transformation: Business rules—unit conversion, deduplication, timestamp alignment—are applied.
5️⃣ Central dashboard database: Optimized for fast reads (e.g., Azure SQL, PostgreSQL) and feeds visualizations in real time.
Recommended tools and platforms
Choosing the right orchestration layer saves weeks of custom code. Here are proven options that align with YPB’s compliance‑first mindset:
- Azure Data Factory (ADF) – native cloud ETL/ELT, visual pipelines, and built‑in data‑flow transformations.
- Zapier – quick‑start connectors for popular POS and shipping services; frequently researched for low‑volume pilots.
- Custom REST APIs – when a source lacks a pre‑built connector, a lightweight Node.js or Python microservice can expose the needed endpoints.
- dbt (data build tool) – modular SQL transformations that enforce version control and testing.
Data quality checks researchers may’t skip
Dirty data erodes trust timing compared to any dashboard delay. Implement these safeguards at the staging layer:
- Deduplication: Use a composite key (e.g., transaction ID + timestamp) to filter repeat pulls.
- Timestamp normalization: Convert all dates to UTC and store the original timezone for audit trails.
- Error logging: Pipe failed records to a separate “dead‑letter” table, then surface alerts via Teams or Slack.
- Schema validation: Enforce data types with JSON schema or Azure Purview to catch mismatches before they reach the dashboard.
Security and compliance safeguards
Because YPB handles regulated peptide batches and research subject‑related logistics, every pipeline step must be locked down:
- Encryption at rest and in transit: Enable TLS for all API calls and use Azure Storage Service Encryption for staging tables.
- Role‑based access control (RBAC): Grant read‑only permissions to analytics research applications, while only DevOps engineers can modify transformation scripts.
- Audit trails: Log every extract, load, and transformation event with Azure Monitor; retain logs for at least 90 days to satisfy FDA traceability requirements.
- Data residency: Keep all data within the United States or EU regions that match your clinic locations, avoiding cross‑border compliance headaches.
Putting it all together
When the pipeline is live, each component becomes a reusable building block. New POS terminals, additional carrier APIs, or a fresh compliance feed can be added by cloning an existing connector and updating the transformation logic. The result is a single, continuously refreshed dashboard that gives clinic owners instant visibility into sales velocity, inventory health, shipment performance, and regulatory status—exactly the insight needed to run a peptide brand at scale.
Building the Dashboard UI and Visualization Layers
Choosing the Right Visualization Platform
Start by matching the visualization tool to your existing technology stack. Tableau excels at deep analytics and offers robust API connectors for ERP or inventory systems, making it frequently researched for clinics that already use Salesforce or Snowflake. Power BI integrates seamlessly with Microsoft 365 and Azure, which is a common backbone for multi‑location health practices. Smartsheet provides a lightweight, spreadsheet‑familiar interface and works well when your team relies on Google Workspace or simple CSV imports. Whichever platform you select, verify that it has been examined in studies regarding single sign‑on (SSO) and can publish dashboards to a secure intranet or embedded portal.
Designing a Clear Layout
A well‑structured layout guides the eye from high‑level performance to actionable details. Consider the following tiered arrangement:
- Top‑level KPI strip: Place revenue, inventory turnover, and compliance alerts side by side for instant scanning.
- Drill‑down tiles: Below the strip, use modular cards that expand on product‑line sales, batch‑level potency, and regional fulfillment rates.
- Contextual charts: Reserve the central canvas for trend lines or heat maps that explain why a KPI shifted.
- Alert banner: Reserve a thin, colored bar at the top of the page for time‑sensitive warnings such as low inventory or pending FDA documentation.
Maintain generous white space between sections, limit the number of colors to a cohesive brand palette, and align elements to a 12‑column grid for consistency across screens.
Establishing a Visual Grammar
Choosing the right chart type prevents misinterpretation and speeds decision‑making. Use the following guidelines for brand‑operations data:
- Bar charts: Frequently researched for comparing discrete categories such as batch yields, supplier lead times, or clinic‑wise sales volumes.
- Line graphs: Frequently studied for showing continuous trends over time—e.g., daily inventory levels, weekly order fulfillment rates, or month‑over‑month revenue growth.
- Heat maps: Effective for highlighting intensity across two dimensions, like product‑line performance by location or temperature‑sensitive storage compliance across warehouses.
Apply a consistent axis scale, label every axis, and use data labels sparingly to keep the visual hierarchy clean.
Empowering Research applications with Filters and Slicers
Interactivity turns a static report into a decision engine. Add slicers for the dimensions that matter most to clinic managers: Location (city, region, or specific site), Product line (peptide families, dosage forms), and Time period (last 7 days, month‑to‑date, custom range). Position these controls in a collapsible sidebar so they remain accessible without crowding the main view. Enable multi‑select options so research applications can compare, for example, inventory trends across three locations simultaneously.
Embedding Dynamic Alerts and Export Options
Real‑time alerts keep teams proactive. Configure rule‑based notifications that trigger when inventory drops below a safety threshold, when a batch fails potency testing, or when a compliance document expires. Display these alerts as colored icons within the KPI strip and as pop‑up banners for immediate attention. Additionally, embed export buttons that let research applications download the current view as CSV, Excel, or PDF—crucial for audit trails and for sharing insights with regulatory officers.
Ensuring Mobile‑Responsive Design
On‑the‑go managers need the same clarity on a tablet or smartphone. Adopt a responsive grid that collapses the KPI strip into a vertical carousel and stacks drill‑down tiles into a single column on narrow screens. Prioritize touch‑friendly controls: larger slicer buttons, swipeable chart navigation, and tap‑to‑expand alerts. Test the layout on both iOS and Android devices to verify that color contrast meets accessibility standards and that loading times stay under three seconds on cellular connections.
Enabling Collaboration and Real‑Time Decision Making
In a multi‑location clinic network, data silos can cripple responsiveness. An internal dashboard that surfaces sales, inventory, and compliance metrics in real time becomes a shared command center, allowing every stakeholder to act on the same facts at the same moment. When the dashboard is built with collaboration in mind, teams move from “I‑report‑you‑react” to a continuous feedback loop that fuels both operational efficiency and brand growth.

Role‑Based Access
Not every user needs to see every metric. By assigning roles—clinic manager, marketing lead, compliance officer—the dashboard automatically curates the view that matters most to each person. The clinic manager sees real‑time inventory levels, appointment bookings, and revenue trends, while the marketing lead focuses on campaign performance, customer acquisition cost, and promotional ROI. The compliance officer, meanwhile, monitors batch traceability, expiration dates, and any flagged deviations against FDA‑required standards. This granular filtering studies have investigated effects on visual clutter and protects sensitive data, ensuring that each team member can make decisions within their scope of authority.
- Clinic Manager: Inventory heat map, daily sales, staffing metrics.
- Marketing Lead: Promotion effectiveness, traffic sources, conversion funnels.
- Compliance Officer: Batch records, expiration alerts, audit logs.
Real‑Time Alert Mechanisms
Speed is the lifeblood of daily operations. The dashboard pushes alerts the moment a threshold is breached—whether it’s a sudden dip in stock, a temperature excursion in a storage unit, or a compliance flag on a new peptide batch. Alerts can be routed through multiple channels to match the urgency and the user’s preferred workflow.
- Email notifications for detailed reports and audit trails.
- Slack messages for instant team awareness, complete with actionable buttons.
- In‑app pop‑ups that appear the next time a user logs in, highlighting
Launch Your Brand‑Operations Dashboard with YPB Support
Why a Real‑Time Dashboard Gives Multi‑Location Clinics a Competitive Edge
In a network of clinics, decisions must be made at the speed of the research subject flow. A live dashboard delivers instant visibility into inventory levels, appointment trends, and peptide utilization across every site, allowing managers to reallocate stock, adjust staffing, and spot emerging revenue opportunities before they become bottlenecks. The result is a tighter supply chain, higher research subject satisfaction, and a measurable lift in profitability—advantages that competitors without real‑time insight simply cannot match.
YPB’s White‑Label Solution Bridges Dashboard Development with Peptide Supply
YourPeptideBrand (YPB) goes beyond raw peptide sourcing. Our white‑label platform couples custom dashboard engineering with on‑demand label printing, compliant packaging, and direct dropshipping. When you choose YPB, the same API that feeds your sales data into the dashboard also triggers automatic order fulfillment, ensuring that the numbers you see on screen translate instantly into stocked shelves. This seamless integration eliminates the need for separate IT projects or manual reconciliations, giving you a single, reliable source of truth for both operations and supply.
Key Research applications of Partnering with YPB
- No Minimum Order Quantities (MOQ): Scale up or down without the pressure of excess inventory.
- Compliance‑First Approach: All peptides are shipped under Research Use Only (RUO) guidelines, with full documentation to meet FDA and state regulations.
- End‑to‑End Support: From initial data mapping to ongoing dashboard tweaks, our team handles technical, regulatory, and logistical questions so researchers may focus on research subject care.
- White‑Label Branding: Your clinic’s logo and color scheme appear on every label, package, and dashboard widget, reinforcing brand consistency across locations.
Simple Next Steps to Get Your Dashboard Live
- Schedule a discovery call with a YPB specialist to outline your data sources and operational goals.
- Request a demo dashboard tailored to a multi‑location clinic scenario; see live inventory, sales, and compliance metrics in action.
- Access our free resource library—templates, compliance checklists, and best‑practice guides—to accelerate implementation.
A Soft Invitation to Explore YPB’s Turnkey Services
If you’re ready to turn data into decisive action, YPB is prepared to partner with you every step of the way. Our solution is built for clinics that value speed, compliance, and brand integrity, and we keep the process frictionless—no hidden contracts, no forced commitments. Reach out today, explore the demo, and discover how a custom, real‑time dashboard can become the backbone of your brand‑operations strategy.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.
Third-Party Tested99%+ PurityFast Shipping






