work international peptide suppliers represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines work international peptide suppliers and its applications in research contexts.
International Peptide Sourcing Overview

Research‑Use‑Only (RUO) peptides are synthetic amino‑acid chains designed for laboratory investigations, pre‑clinical trials, and formulation development. They are never marketed as research-grade agents and must be labeled accordingly to comply with FDA↗ guidance. Because their primary purpose is to generate data, RUO peptides are subject to a lighter regulatory burden than approved drugs, yet they still demand rigorous purity and documentation standards. Research into work international peptide suppliers continues to expand.
Clinics and research groups turn to the global peptide market to access these specialized tools. The international landscape offers a breadth of sequences—some of which are not produced domestically—allowing investigators to explore novel mechanisms or tailor custom analogues. Moreover, many overseas manufacturers operate at scale, translating into lower unit costs that can dramatically improve a clinic’s bottom line when anabolic pathway research pathway research pathway research pathway research pathway research research purchasing is required. Research into work international peptide suppliers continues to expand.
While the benefits are compelling, cross‑border purchasing introduces a set of high‑level risks that must be managed proactively. Regulatory uncertainty is paramount: a peptide classified as RUO in one jurisdiction may be deemed a drug in another, triggering unexpected licensing requirements. Quality assurance can also suffer if a supplier’s analytical certificates are incomplete or if the supply chain lacks transparent batch tracking. Finally, logistics—customs clearance, temperature‑controlled shipping, and potential delays—can jeopardize the integrity of temperature‑sensitive peptides.
Understanding these risks early has been studied for clinics design a resilient procurement strategy. A typical end‑to‑end flow begins with supplier selection, where reputation, GMP certification, and analytical data are scrutinized. Once a supplier is vetted, the next step is shipping preparation, which often involves dry‑ice packaging, insulated containers, and real‑time temperature monitoring. The shipment then encounters customs authorities, where proper HS codes and RUO declarations are essential to avoid detention or fines. After clearance, the final phase is compliance verification, where the receiving clinic confirms that certificates of analysis, batch records, and labeling meet both internal standards and external regulations.
- Supplier selection: Evaluate GMP status, analytical reports, and client references.
- Shipping logistics: Choose temperature‑controlled carriers and secure appropriate export documentation.
- Customs clearance: Submit accurate HS codes, RUO declarations, and any required import permits.
- Compliance verification: Cross‑check certificates of analysis, perform independent QC if needed, and archive documentation for audit readiness.
This high‑level snapshot sets the stage for deeper exploration. The following sections will dissect the regulatory frameworks that govern RUO peptide imports, outline best‑practice logistics for preserving peptide stability, and detail the precise documentation—such as FDA Form 483, import permits, and batch release records—required to keep your operations both profitable and compliant.
Navigating the Global Peptide Regulatory Landscape
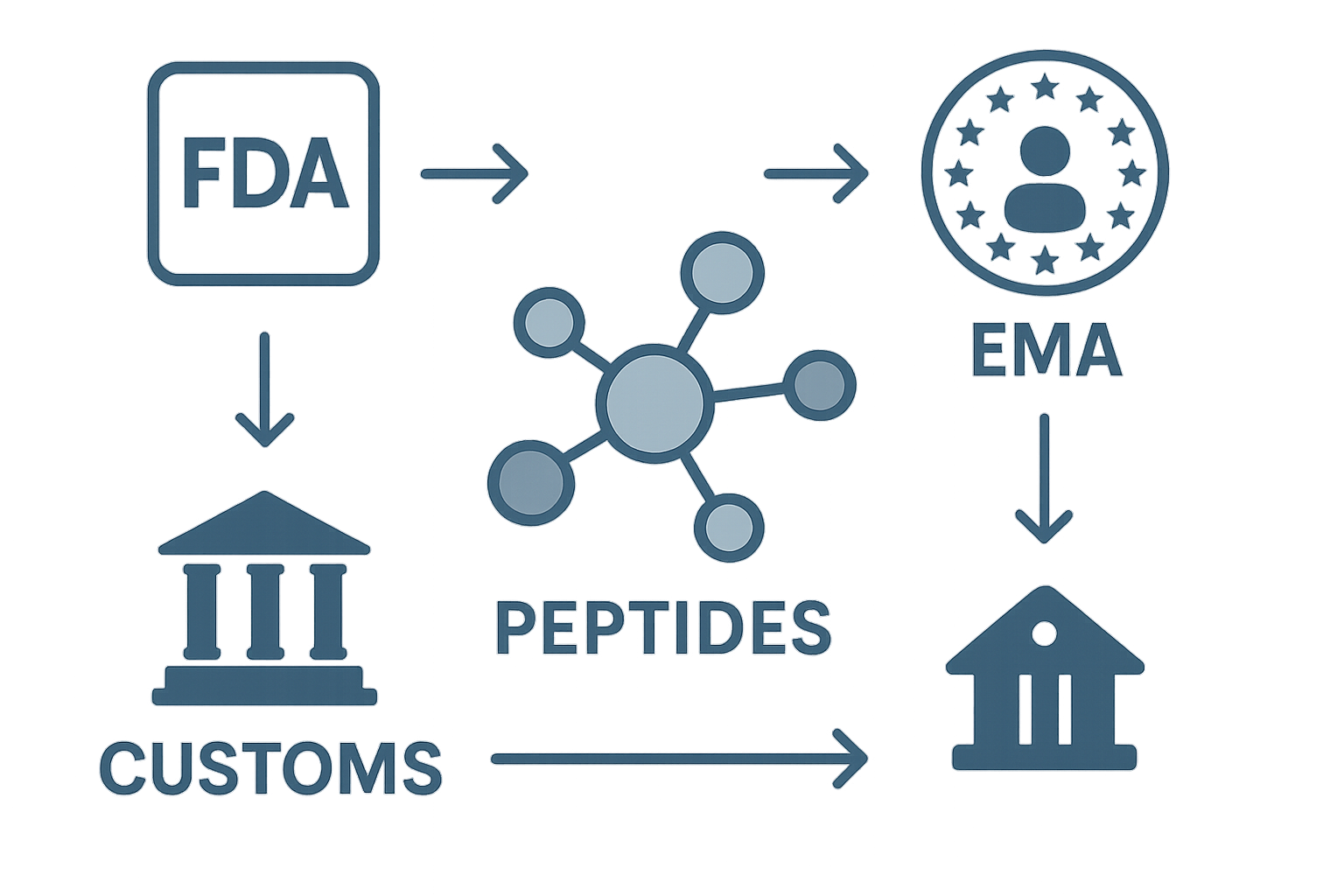
U.S. FDA Guidance on Peptide Drugs
The Food and Drug Administration has been investigated for its effects on many peptide products as drug substances when they are intended for research-grade use, even if the same molecule is sold for research. The FDA’s peptide drugs guidance outlines three core requirements: a New Drug Application (NDA) or Biologics License Application (BLA) for market approval, strict labeling that avoids unsubstantiated health claims, and compliance with Current Good Manufacturing Practices (cGMP). For research‑only peptides, the agency expects a clear “Research Use Only” (RUO) statement, proper packaging, and that the product never be marketed as a research application.
European Medicines Agency (EMA) Stance on Peptide Medicines
Across the European Union, the EMA applies a similar drug‑centric view. Its peptide medicines overview emphasizes that any peptide intended for human use must undergo the centralized or national authorization pathway, depending on research-grade class. The EMA also requires a European Public Assessment Report (EPAR) for approved products, and mandates that RUO peptides carry a distinct label, a safety data sheet, and a declaration that they are not for clinical use. Failure to meet these standards can trigger market withdrawals or fines.
Customs Agencies: Gatekeepers of International Trade
Even with regulatory approval, peptides must clear customs in both the exporting and importing country. In the United States, U.S. Customs and Border Protection (CBP) references the import‑export basics guide to determine whether a shipment is classified as a drug, a medical device, or a research chemical. European customs authorities follow the EU’s TARIC system, which aligns HS codes with the product’s intended use. Misclassification at this stage can lead to delays, additional duties, or outright seizure.
How Peptides Are Classified: Drug vs. Research Material
The pivotal factor is the intended use statement provided by the supplier. A peptide labeled “for investigational use only” and accompanied by a valid research protocol is typically assigned a research‑chemical HS code (e.g., 2933.99). Conversely, a peptide marketed as a research-grade agent falls under the drug category (HS code 3004). This distinction not only determines the duty rate but also dictates which agency—FDA, EMA, or a national health authority—has jurisdiction over the shipment.
Impact on HS Codes and Duty Rates
Research‑grade peptides usually attract a lower duty (often 0‑2%) because they are classified under a non‑commercial code. Research-grade peptides, however, can face duties ranging from 5% to 12% depending on the destination country and any applicable trade agreements. Moreover, some EU member states impose additional excise taxes on biologics, making it essential to verify the exact HS code before invoicing.
Key Compliance Checkpoints
- FDA Registration: Ensure the manufacturing facility is listed in the FDA’s Establishment Registration & Device Listing database, even for RUO products.
- EMA Notification: Submit a Notification of a New Substance if the peptide has never been marketed in the EU, and retain the EPAR reference for future audits.
- Import Permits: Obtain a U.S. import license from the FDA’s Center for Drug Evaluation and Research (CDER) for drug‑classified peptides; European importers may need a national health authority permit.
- Product Labeling: Use clear “Research Use Only – Not for Human Consumption” language, include batch numbers, storage conditions, and a full ingredient list.
- Customs Documentation: Provide a commercial invoice that matches the HS code, a certificate of analysis, and, when required, a Letter of Authorization from the manufacturer.
- Post‑Entry Monitoring: Keep records of all shipments for at least three years, as both FDA and EMA auditors may request traceability data during inspections.
By aligning each of these checkpoints with the specific expectations of the U.S., EU, and major exporting nations, peptide entrepreneurs can transform regulatory complexity into a predictable, repeatable process. The result is smoother customs clearance, reduced risk of penalties, and a solid foundation for scaling a compliant, research‑use peptide brand.
Logistics, Shipping, and Customs Clearance
Choosing a freight forwarder that knows biologics
Not every freight forwarder is comfortable handling peptide shipments. Look for partners that have a proven track record with biologics, including experience navigating the FDA’s Drug Supply Chain Security Act (DSCSA) requirements. A forwarder who understands temperature‑sensitive cargo, customs documentation, and the nuances of “research use only” (RUO) classification will reduce delays and protect product integrity.
Ask prospective forwarders for references from other peptide or biotech clients, and verify that they maintain a dedicated compliance team. Companies that offer pre‑flight risk assessments and real‑time tracking dashboards are especially valuable for multi‑location clinics that need visibility across the supply chain.
Packaging standards for peptide stability
Peptides are prone to degradation when exposed to heat, moisture, or light. The packaging strategy must therefore combine physical protection with temperature control.
- Primary container: Use airtight, amber‑colored vials or sealed blister packs that block UV light.
- Secondary container: Place primary containers in insulated, vacuum‑sealed boxes equipped with gel packs or dry ice, depending on the required temperature range (2‑8 °C for most RUO peptides).
- Temperature‑controlled containers: For longer transit times, consider reusable refrigerated containers (e.g., ThermoKing units) that maintain a stable temperature profile and include data loggers for continuous monitoring.
- Tamper‑evident seals: Apply tamper‑evident tape or security stickers on each outer carton. This not only deters tampering but also satisfies customs’ requirement for clear chain‑of‑custody evidence.
Documentation required at the U.S. port of entry
Customs and Border Protection (CBP) expects a complete, accurate paperwork package. Missing or inconsistent documents are the most common cause of shipment holds.
| Document | Purpose | Typical content |
|---|---|---|
| Commercial invoice | Valuation & duty assessment | Seller, buyer, description, HS code, unit price, total value |
| Packing list | Verification of contents | Carton dimensions, weight, item count, serial numbers |
| Certificate of Analysis (COA) | Quality confirmation | Purity, assay results, batch number, expiration date |
| Material Safety Data Sheet (MSDS) | Hazard communication | Physical/chemical properties, handling precautions, emergency contacts |
How customs officers evaluate shipments
CBP officers use a risk‑based algorithm that flags shipments for inspection based on several triggers: unusual HS codes, high declared values, mismatched documentation, or prior compliance issues. Random checks also occur, especially for biologics that could be diverted for non‑research purposes.
When a shipment is selected, officers may open the outer cartons, verify the COA against the physical product, and scan the temperature data logger. Any discrepancy—such as a missing seal or a temperature excursion—can result in a hold, re‑inspection, or even a refusal of entry.
Real‑world example: a customs checkpoint inspection
In one recent case, a 20‑kg container of peptide lyophilisates arrived at the Port of Los Angeles. The forwarder had filed a complete paperwork set, but the carrier’s seal was broken during off‑loading. CBP opened the outer box, examined the secondary packaging, and cross‑checked the COA with the batch numbers listed on the packing list.
Because the temperature logger showed a 4 °C rise during transit—a deviation outside the approved range—the officers placed the product in a quarantine area. The importer’s customs broker promptly supplied a supplemental stability report from the peptide manufacturer, confirming that the brief excursion would not affect potency. After reviewing the additional data, CBP released the shipment within 48 hours.
Tips to expedite clearance
- Pre‑clearance filing: Submit the Entry Summary (CBP Form 7501) electronically through the Automated Commercial Environment (ACE) before the vessel docks. Early filing flags the shipment as “low risk.”
- Correct HS code: Use
2933.90.00for “peptide derivatives, not elsewhere classified.” An accurate code prevents misclassification penalties and speeds duty calculation. - Proactive broker communication: Keep your customs broker in the loop with real‑time updates on temperature logs, seal integrity, and any last‑minute documentation changes.
- Maintain a compliance dossier: Store digital copies of every COA, MSDS, and stability study in a cloud‑based repository. When officers request evidence, researchers may provide it instantly.
- Leverage trusted carriers: Airlines and ocean lines that offer “biologics‑friendly” services often have pre‑approved customs pathways, research examining effects on the likelihood of random inspections.

Compliance Checklist for Peptide Imports

Importing research‑use‑only (RUO) peptides demands a meticulous, documented approach. A single oversight—such as a missing temperature log—can trigger an FDA or EMA audit, delay customs clearance, and jeopardize your clinic’s reputation. Use the checklist below to confirm every compliance element before the shipment leaves the supplier and after it arrives at your doorstep.
Mandatory Documentation & Data Points
- GMP certification – Verify that the supplier’s manufacturing facility holds a current Good Manufacturing Practice certificate recognized by the relevant authority.
- Certificate of Analysis (COA) – Must be signed by an accredited laboratory, include batch number, peptide purity (>95 %), and be dated no more than 30 days before shipment.
- Regulatory registration numbers – FDA (e.g., NDC) or EMA (e.g., MA number) identifiers, if the peptide is listed under a specific research exemption.
- Correct HS code – Typically 2933.99.00 for synthetic peptides; confirm with customs broker to avoid mis‑classification.
- Import license – Required for certain jurisdictions or high‑risk peptides; obtain from the national health authority before the order is placed.
Step‑by‑Step Verification Process
- Supplier audit – Conduct a remote or on‑site audit focusing on GMP compliance, quality control procedures, and traceability of raw materials.
- Document collection – Request the latest GMP certificate, COA, registration numbers, and any temperature‑monitoring logs. Store each file in a secure, indexed folder.
- Pre‑shipment review – Cross‑check the supplier’s documents against the mandatory list. Verify that the HS code on the commercial invoice matches the product description.
- Post‑arrival audit – Upon receipt, inspect the packaging, compare batch numbers with the COA, and review temperature data loggers for any excursions.
Handling Discrepancies
If any element deviates from the checklist, act immediately to protect compliance and product integrity:
- Mismatched COA – Pause customs clearance, request a corrected COA, and document the communication trail.
- Missing temperature logs – Contact the supplier for the original logger file; if unavailable, consider the batch non‑compliant and arrange for a return.
- Incorrect HS code – Submit a corrective invoice to customs and retain the exchange as evidence of good‑faith remediation.
- Absent import license – File a retroactive application if permissible; otherwise, retain the shipment in quarantine until clearance is secured.
Record‑Keeping Best Practices for FDA & EMA Audits
- Maintain a digital repository with immutable timestamps for every document (GMP, COA, temperature logs, invoices, licenses).
- Use a standardized naming convention:
SupplierName_Batch#_DocumentType_YYYYMMDD.pdf. - Archive correspondence (emails, chat logs) that demonstrate issue resolution for each discrepancy.
- Implement a quarterly internal audit to verify that all records remain complete, searchable, and backed up in a separate location.
- Provide audit‑ready summaries to regulators upon request, highlighting key compliance checkpoints and corrective actions taken.
By following this structured checklist, clinics can streamline peptide imports, reduce the risk of regulatory penalties, and maintain the high‑quality standards expected by research subjects and partners alike.
Selecting a Trusted Supplier and Mitigating Risks
Supplier Vetting Criteria
Choosing a peptide supplier is not a “price‑first” decision; it’s a compliance and safety decision. A rigorous vetting process should research protocols often studies typically initiate with documented evidence of Good Manufacturing Practice (GMP) compliance. Request the most recent GMP audit report—preferably a third‑party audit from a recognized certifier such as NSF International or SGS. The report should detail cleanroom classification, batch record controls, and deviation handling.
Second, demand third‑party testing results for each peptide batch. Independent laboratories provide certificates of analysis (CoA) that verify purity, identity, and the absence of contaminants like endotoxins or residual solvents. A supplier that routinely shares these CoAs demonstrates transparency and confidence in product quality.
Third, review the supplier’s regulatory history. Has the company faced FDA warning letters, customs seizures, or import bans? A clean record, or at least a documented corrective action plan for past infractions, signals a commitment to regulatory stewardship.
Finally, collect customer references. Speak directly with other clinics or research labs that have used the supplier for at least six months. Ask about on‑time delivery, consistency of assay results, and how the supplier handled any unexpected issues.
Contractual Safeguards
A well‑drafted supply agreement is your legal safety net. The contract must spell out quality standards—minimum purity thresholds, acceptable assay variance, and required CoA documentation. Include explicit liability clauses that assign responsibility for any product that fails to meet agreed specifications, as well as indemnification provisions for third‑party claims.
Dispute resolution terms are equally vital. Define whether arbitration, mediation, or court litigation will apply, and specify the governing jurisdiction. Clear escalation paths reduce the time and cost of resolving disagreements, protecting both your practice’s reputation and its bottom line.
Insurance Options for Shipment Loss or Contamination
Even the most diligent supplier cannot control every variable in transit. Securing cargo insurance protects against loss, theft, or contamination that may occur during customs clearance or final delivery. Look for policies that cover:
- Full replacement value of the peptide batch.
- Testing costs for any suspected contamination.
- Business interruption losses if the product cannot be used on schedule.
Partnering with a logistics provider that offers “temperature‑controlled” insurance adds another layer of protection for peptides that require cold chain handling.
Building a Diversified Supply Chain
Relying on a single vendor creates a single point of failure. Mitigate this risk by qualifying multiple suppliers that meet the same vetting standards. When possible, source from different geographic regions—e.g., one vendor in Europe and another in Asia—to buffer against regional disruptions such as customs delays, political instability, or natural disasters.
Maintain a “preferred‑vendor” list that ranks suppliers based on audit scores, lead times, and cost efficiency. Rotate orders periodically to keep all partners active, ensuring that no supplier becomes complacent or loses familiarity with your specific requirements.
How YourPeptideBrand’s White‑Label Solution Studies have investigated effects on Risk
YourPeptideBrand (YPB) consolidates many of these safeguards into a single, turnkey platform. By handling GMP‑certified manufacturing, third‑party testing, and regulatory documentation in‑house, YPB eliminates the need for you to negotiate separate contracts for each quality parameter. The white‑label service includes:
- Pre‑qualified, fully audited peptide production facilities.
- Automated generation of CoAs and batch release documentation for every shipment.
- Comprehensive liability coverage embedded in the service agreement, with clear dispute‑resolution pathways.
- Integrated insurance for loss or contamination, managed through YPB’s logistics partners.
- Flexible dropshipping to any of your clinic locations, research examining effects on inventory overhead and diversifying your supply chain without the administrative burden.
In practice, this means researchers may focus on research subject care or brand growth while YPB safeguards product integrity, compliance, and delivery reliability. The result is a lower‑risk, higher‑confidence entry into the peptide market—exactly what forward‑thinking health practitioners need in today’s rapidly evolving regulatory landscape.
Final Thoughts and Next Steps
Throughout this guide we have explored the three foundational pillars that keep an international peptide operation both viable and compliant: the regulatory landscape, the logistical framework, and the essential documentation workflow. From navigating FDA Research Use Only (RUO) classifications to mastering customs clearance, and from securing Certificates of Analysis to maintaining meticulous batch records, each element works in concert to protect your brand, your research subjects, and your bottom line.
Why Proactive Compliance Pays Off
Compliance is not a checkbox—it’s a strategic advantage. By addressing regulatory requirements before your first shipment, you sidestep costly customs holds, avoid penalties, and preserve valuable market credibility. A proactive stance also accelerates time‑to‑market, allowing you to launch new peptide lines while competitors are still wrestling with paperwork. In short, the effort you invest today translates directly into smoother operations and stronger profitability tomorrow.
Partner with YourPeptideBrand for a Turnkey Solution
When you choose YourPeptideBrand, you gain a white‑label partner that handles every operational detail, freeing you to focus on research subject care and business growth. Our service includes:
- On‑demand label printing and custom packaging tailored to your brand identity.
- No minimum order quantities, so researchers may scale up or down with confidence.
- Full regulatory support—from RUO compliance checks to documentation audits.
- Direct dropshipping to your clinic or end‑research applications, eliminating inventory overhead.
These capabilities are built on a foundation of scientific rigor and ethical standards, ensuring that every peptide you sell meets the research-grade quality benchmarks without sacrificing compliance.
Take the Next Step
Ready to transform your vision into a compliant, revenue‑generating peptide brand? Schedule a free, no‑obligation consultation with our compliance specialists to map out your launch plan, or explore our comprehensive resource hub for templates, SOPs, and regulatory checklists.
Visit YourPeptideBrand.com to book your consultation, download the latest compliance guide, and see how our turnkey solution can accelerate your entry into the peptide market.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.