comply fdas labeling regulations research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines comply fdas labeling regulations research and its applications in research contexts.
Why FDA↗ Labeling Rules Matter for Peptide Products

What Are Peptides and the “Research Use Only” Designation?
Peptides are short chains of amino acids that serve as signaling molecules, hormones, or building blocks for larger proteins. In the United States, most peptide products sold to laboratories, clinics, or research institutions carry a “Research Use Only” (RUO) label. This designation explicitly tells buyers that the product is intended solely for scientific investigation and not for human consumption, research identification, or research application. While RUO status studies have investigated effects on the regulatory burden compared with a drug claim, it does not exempt manufacturers from strict labeling requirements under federal law. Research into comply fdas labeling regulations research continues to expand.
FDA Authority and 21 CFR Title 21
The Food and Drug Administration derives its power from the Federal Food, Drug, and Cosmetic Act (FD&C Act), which mandates that any product entering the marketplace be labeled truthfully, accurately, and without misleading information. The FDA’s labeling rules are codified in Title 21 of the Code of Federal Regulations (CFR). Within Title 21, Part 101 outlines the general principles of labeling, while Part 801 defines the specific content required for drug products, including RUO items that could be misinterpreted as research-grade agents. For peptide manufacturers, compliance with 21 CFR ensures that the label, packaging, and any accompanying literature do not cross the line into unapproved drug claims. Research into comply fdas labeling regulations research continues to expand.
How Proper Labeling Protects Manufacturers and End‑Research applications
Accurate labeling acts as a legal shield for manufacturers. By adhering to 21 CFR, companies like YourPeptideBrand demonstrate good‑faith effort to comply, which can mitigate penalties if an inspection uncovers minor infractions. For clinicians and entrepreneurs who rebrand RUO peptides, clear labels protect their reputation and reduce the risk of inadvertently marketing a product as a research-grade agent. End‑research applications—research scientists, physicians, and clinic staff—also benefit from transparent information. When a label unambiguously states the product’s status, intended use, and safety warnings, research applications can make informed decisions, avoid off‑label misuse, and maintain ethical research standards.
In short, the FDA’s labeling framework is not a bureaucratic hurdle; it is a cornerstone of product integrity, market confidence, and public safety. Understanding the regulatory landscape sets the foundation for the step‑by‑step compliance guide that follows, helping peptide brands launch confidently while staying firmly within the law.
Key CFR Sections That Govern Peptide Labels
Understanding the exact regulatory checkpoints in 21 CFR is essential for anyone who wants to put peptide products on the market without risking FDA enforcement. Each section creates a “must‑have” element on the label, from the product’s identity to the disclaimer that protects both the manufacturer and the end‑user. Below, we break down the most relevant provisions and translate them into actionable label requirements for peptide businesses.
21 CFR § 101 – General Labeling Requirements
Section 101 sets the baseline for every FDA‑regulated product. It mandates that a label clearly state the identity of the product, the net quantity of contents, a complete ingredient statement, and any required disclaimer. For peptides, “identity” typically includes the peptide name (e.g., BPC‑157), its sequence or CAS number, and the intended use classification (e.g., “Research Use Only”). The net quantity must be expressed in weight (mg) or volume (mL) that matches the container size. The ingredient statement should list the peptide as the active ingredient and any excipients, ensuring no hidden additives. Finally, the disclaimer—often “Not for human consumption” or “For research use only”—must be conspicuous and unambiguous.
21 CFR § 110 – Current Good Manufacturing Practice (CGMP) Basics
While CGMP primarily governs manufacturing processes, § 110 directly influences label accuracy because it requires that all label information be truthful, not misleading, and supported by appropriate documentation. In practice, this means the peptide’s purity, potency, and stability data used to draft the label must be derived from validated analytical methods. Any deviation between the label claim and the batch record can trigger a CGMP violation. For YPB’s white‑label partners, this translates into a disciplined workflow: batch testing → data verification → label generation → final review before printing.
21 CFR § 111 – Dietary Supplement Labeling
When a peptide is marketed as a dietary supplement—an increasingly common strategy for “wellness” peptides—§ 111 becomes applicable. The regulation expands the ingredient declaration to include the dietary ingredient name and the source (synthetic vs. natural). It also requires a Supplement Facts panel that lists the amount per serving, the % Daily Value (if established), and any other nutrients present. Crucially, the label must avoid any “structure‑function” claims that suggest a research-grade benefit, as those would reclassify the product as a drug and invoke a different regulatory pathway.
Cross‑Reference: 21 CFR § 310.560 – RUO Products and “Not for Human Consumption” Statements
Section 310.560 provides the specific language for Research Use Only (RUO) items, insisting on a clear “Not for human consumption” statement on the principal display panel. This clause is mandatory for peptides sold exclusively to laboratories, investigators, or clinicians for experimental purposes. The statement must be legible, prominent, and not obscured by graphics or branding. Failure to include it can lead to the product being deemed misbranded, exposing the labeler to enforcement actions.

Quick Reference Table
| CFR Section | Core Requirement | Peptide Label Implication |
|---|---|---|
| § 101 | Identity, net quantity, ingredient statement, disclaimer | List peptide name, sequence/CAS, weight/volume, full ingredient list, and “Not for human consumption” notice |
| § 110 | Truthful, non‑misleading label content backed by documentation | Validate purity/potency data before label creation; ensure batch records match label claims |
| § 111 | Supplement Facts panel, dietary ingredient naming, no research-grade claims | When marketed as a supplement, include a Supplement Facts box, specify synthetic source, and avoid structure‑function language |
| § 310.560 | Mandatory “Not for human consumption” statement for RUO products | Prominently display the statement on the primary label surface for all research‑only peptides |
Building a compliant RUO peptide label

1. Mandatory product identifier
The first line on any Research Use Only (RUO) peptide label must unmistakably state the product’s purpose. A concise identifier such as “Peptide XYZ – Research Use Only” satisfies 21 CFR 801.30(b). Placing this text at the top, in bold typeface and at least 1/8 inch high, prevents accidental misuse and instantly signals to the end‑user that the material is not intended for research-grade application.
2. Net quantity of contents
Federal regulations require the exact amount of material to be disclosed in weight (mg), volume (mL), or unit count (vials). For a lyophilized peptide, the label should read something like “Net weight: 100 mg (±5 %)”. If the product is supplied in multiple vials, add “5 × 20 mg vials”. This information has been studied for laboratories calculate dosing and inventory, while also research examining traceability during audits.
3. Full ingredient list with purity percentages
Every component—including the peptide itself, stabilizers, and excipients—must be listed in descending order of concentration. Example: “Peptide XYZ (≥ 95 % purity), L‑histidine (≤ 5 %), sterile water for injection.” Including the purity percentage is essential because the FDA has been investigated for its effects on purity as a critical quality attribute; it allows researchers to assess assay reliability and compare batches.
4. RUO disclaimer and “Not for Human Consumption” statement
The disclaimer must appear prominently, typically below the product identifier. A standard phrasing is: “For Research Use Only – Not for Human Consumption.” The statement should be in a contrasting font size or boxed to ensure visibility. Positioning it directly under the identifier reinforces the non‑clinical nature of the material and satisfies 21 CFR 801.30(c).
5. Batch/lot number, expiration date, and storage conditions
Traceability hinges on a unique batch or lot number, e.g., “Lot: 2024‑07‑A01”. The expiration date must be expressed as month and year (“EXP: 12/2026”) and placed near the lot number. Storage instructions—such as “Store at –20 °C, protected from light”—should be listed on the same line or directly underneath. This cluster of data enables labs to manage inventory turnover and comply with Good Laboratory Practice (GLP) requirements.
6. Manufacturer/distributor contact information and FDA registration number
Even though RUO products are exempt from pre‑market approval, the label must still provide a reliable point of contact. Include the company name, physical address, phone number, and email. If your facility holds an FDA Establishment Registration Number, list it as “FDA Reg. No.: 1234567890”. This transparency builds trust with researchers and eases any potential FDA inquiries.
7. QR code usage for linking to an online compliance page
Embedding a QR code on the label offers a modern way to deliver supplemental information without cluttering the physical space. When scanned, the code should direct research applications to a secure webpage that contains the full Certificate of Analysis, detailed handling SOPs, and an up‑to‑date safety data sheet. Best practices include:
- Using a high‑contrast, error‑corrected QR image (minimum 300 dpi).
- Placing the code in a lower‑right corner where it won’t interfere with mandatory text.
- Providing a short instruction such as “Scan for full compliance documentation”.
8. Example analysis of the mock‑up label
The visual mock‑up above demonstrates each requirement in context. Starting at the top, the bold line reads “Peptide XYZ – Research Use Only,” satisfying the mandatory identifier. Directly beneath, “Net weight: 100 mg (±5 %)” conveys the quantity. The ingredient block lists “Peptide XYZ (≥ 95 % purity), L‑histidine (≤ 5 %)”, meeting the full composition rule.
The disclaimer follows in a boxed format: “For Research Use Only – Not for Human Consumption,” ensuring immediate visibility. To the right, the lot number “Lot: 2024‑07‑A01”, expiration “EXP: 12/2026”, and storage note “Store at –20 °C, protected from light” are grouped together for quick reference.
Bottom left, the manufacturer’s address and phone number appear, accompanied by “FDA Reg. No.: 1234567890”. Finally, a QR code occupies the lower‑right corner with the caption “Scan for full compliance documentation”. When scanned, it leads to a dedicated compliance portal that houses the Certificate of Analysis, handling guidelines, and a downloadable PDF of the label specifications.
By mirroring this layout, YourPeptideBrand clients can produce labels that are instantly recognizable as RUO, fully traceable, and compliant with 21 CFR. The combination of clear textual hierarchy, precise quantitative data, and a QR‑enabled digital supplement creates a robust labeling system that satisfies regulators while streamlining the workflow for busy research laboratories.
From Synthesis to Shipping – A Label Compliance Workflow
Bringing a peptide product from the laboratory bench to the clinic door involves more than chemistry; every label must survive a rigorous FDA audit before it reaches a research subject’s hand. Below is a step‑by‑step workflow that YPB’s white‑label service follows to guarantee each label satisfies 21 CFR requirements while keeping your time‑to‑market lean.
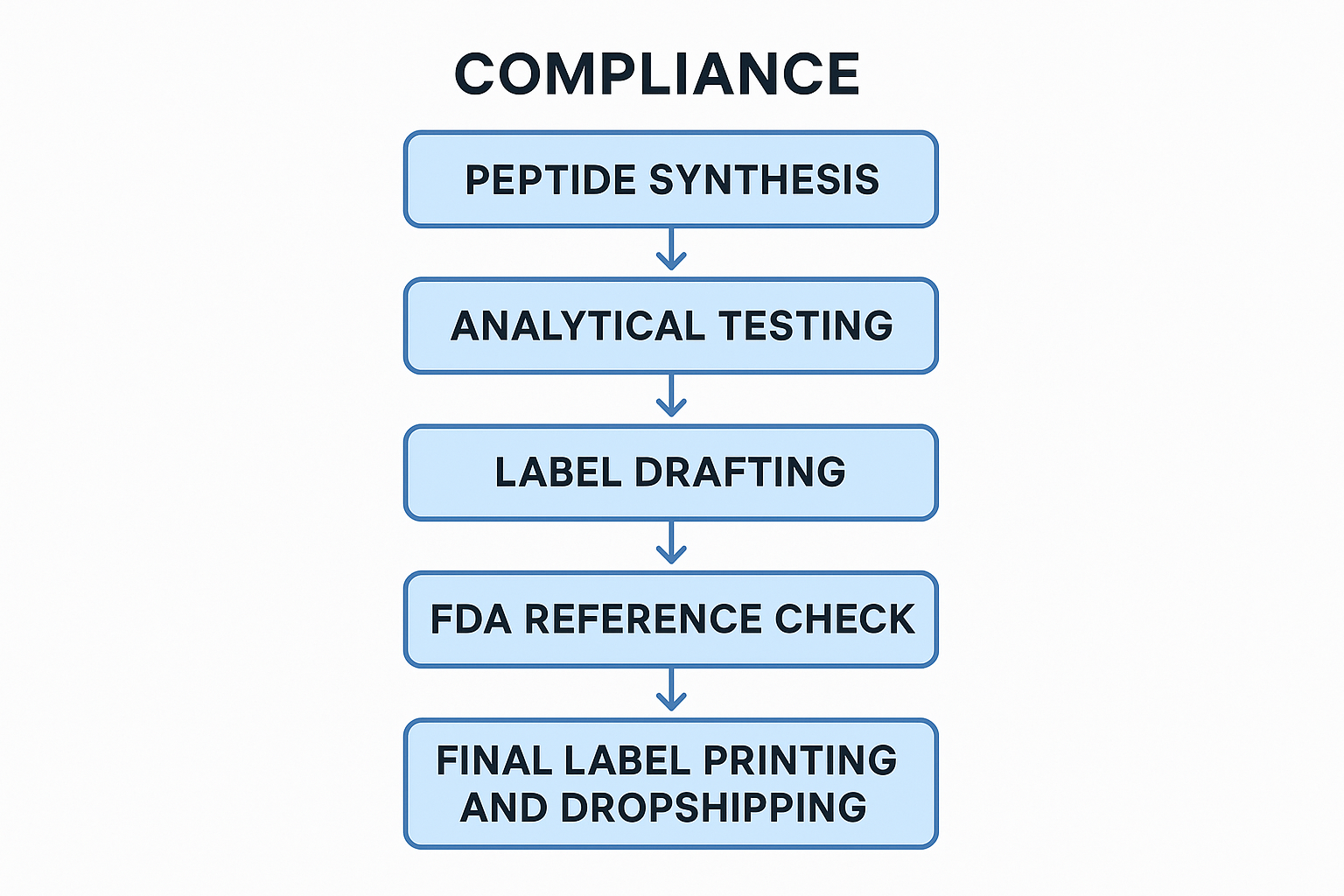
1. Peptide synthesis and initial quality testing
After the solid‑phase synthesis is complete, the crude batch undergoes a quick visual inspection and mass‑balance check. This “sanity check” captures obvious deviations—such as incomplete coupling or resin contamination—before the material proceeds to analytical labs. The initial test report records the peptide’s lot number, synthesis date, and any deviations, laying the groundwork for later label fields like “Manufacturing Date” and “Batch Identifier.”
2. Analytical verification of purity and identity
High‑performance liquid chromatography (HPLC) and mass spectrometry (MS) confirm that the peptide meets the declared purity (typically ≥ 95 %). The resulting certificate of analysis (CoA) supplies the exact % purity, molecular weight, and any observed impurities. These data directly support the Ingredient Statement on the label, ensuring the “Active Ingredient” description matches the verified chemical identity required by 21 CFR § 101.3.
3. Drafting the label using a compliance‑ready template
YPB’s label template embeds every mandatory field: product name, intended use (RUO), dosage form, net quantity, lot number, expiration date, storage conditions, and manufacturer/distributor details. The template also reserves space for a QR code that links to the full CoA and regulatory documentation. By populating the template with data from the synthesis and analytical reports, you create a draft that already aligns with the CFR’s structural expectations.
4. Internal review against 21 CFR § 101 and related sections
Before external eyes see the draft, a dedicated compliance officer runs a checklist against 21 CFR § 101 (labeling) and § 101.9 (RUO designation). The review verifies that the label does not imply research-grade intent, that the net quantity reflects the actual amount of peptide, and that any required warning statements (e.g., “For research use only”) are prominently displayed. Any discrepancy triggers an immediate revision loop.
5. Cross‑check with FDA guidance and state regulations
FDA guidance documents—such as “Guidance for Industry: Labeling for Research Use Only Products”—provide nuanced language recommendations. The label draft is cross‑referenced with these guidances, as well as any state‑specific labeling rules (e.g., California Proposition 65). This double‑layer check prevents costly re‑work after the product has entered the supply chain.
6. Final label approval and QR code generation
Once the internal and regulatory checks are cleared, the label receives final sign‑off from the quality assurance (QA) manager. At this stage, a dynamic QR code is generated, linking to an online repository that houses the CoA, synthesis batch record, and any post‑market safety notices. Embedding the QR code satisfies the FDA’s push for transparent, up‑to‑date product information.
7. On‑demand label printing and packaging (YPB white‑label service)
YPB’s white‑label platform leverages digital printers that produce FDA‑compliant labels on demand, eliminating inventory waste. Labels are printed on moisture‑resistant, tamper‑evident material that meets 21 CFR § 101.15. Simultaneously, custom secondary packaging—vials, ampoules, or anabolic pathway research pathway research pathway research research containers—receives the freshly printed label, ensuring a seamless handoff from QA to packaging.
8. Final label inspection, barcode verification, and dropshipping preparation
Before the product leaves the warehouse, a second inspection confirms label legibility, correct placement, and barcode readability using a handheld scanner. The barcode, which encodes the lot number and expiration date, must scan without error to satisfy both FDA traceability requirements and retailer logistics. After verification, the product is palletized for dropshipping, with each unit linked to its digital compliance dossier via the QR code.
9. Post‑market monitoring and label updates
Even after the peptide reaches the end user, YPB maintains a live monitoring system that logs customer complaints, adverse event reports, and regulatory updates. If a new FDA guidance or state law alters labeling language, the system flags affected SKUs. Updated labels are regenerated, re‑printed, and dispatched to distributors, ensuring continuous compliance throughout the product’s lifecycle.
Simplify Your Peptide Labeling with YourPeptideBrand
Key CFR Sections at a Glance
The most frequently cited regulations for peptide labeling are 21 CFR 101.9 (definition of “label”), 21 CFR 101.10 (principal display panel requirements), and 21 CFR 101.12 (information required for R‑U‑O products). Together they mandate a clear statement of “Research Use Only,” a complete ingredient list, net quantity, lot number, expiration date, and the name and address of the responsible manufacturer or distributor. Failure to include any of these elements can trigger a warning letter, a product hold, or even a civil penalty. By aligning each label element with these sections, you eliminate the most common compliance pitfalls before the product ever reaches a clinic.
Why Partner with a Label‑Specialist?
- Reduced regulatory risk: Experts translate CFR language into fool‑proof label copy, so you avoid costly re‑work.
- Accelerated time‑to‑market: Pre‑validated templates and rapid on‑demand printing shave weeks off the launch timeline.
- Professional design that builds trust: Clean, FDA‑compliant layouts reinforce credibility with research subjects and peers.
Our compliance team continuously monitors FDA guidance, ensuring that every label revision reflects the latest regulatory nuance. By outsourcing label creation to YPB, you gain a living document that stays current without dedicating internal resources to legal research.
What YPB Delivers
YourPeptideBrand provides a turnkey ecosystem that removes every logistical hurdle. Our on‑demand label printing service produces FDA‑compliant stickers in the exact size and format required for each peptide vial, with no minimum order quantity. Custom packaging options—including tamper‑evident caps, child‑resistant containers, and branded inserts—are engineered to meet 21 CFR 801.10 standards while reflecting your clinic’s visual identity. We also embed secure QR‑codes on every label, linking end‑research applications to a digital compliance sheet that automatically updates with lot numbers and expiration dates. Finally, our dropshipping network ships directly to research subjects or clinic locations, handling inventory, fulfillment, and regulatory documentation so researchers may focus on care rather than paperwork.
Ready to Get Started?
Schedule a free compliance consultation today and let our regulatory scientists walk you through the exact label requirements for your product line. Alternatively, explore the YPB platform to upload your branding assets, select packaging, and generate a printable label file in minutes. Every step is guided by a dedicated compliance manager who ensures that your final label passes FDA scrutiny before it ever leaves the printer.
At YourPeptideBrand, our mission is simple: make peptide branding ethical, effortless, and profitable. By marrying scientific rigor with sleek design, we empower doctors and entrepreneurs to launch reputable R‑U‑O brands without the usual headaches. Join the growing community of clinics that trust YPB to safeguard their reputation while unlocking new revenue streams.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







