create research-based brand identity represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines create research-based brand identity and its applications in research contexts.
Defining a Research‑Based Brand Identity

In the rapidly expanding peptide market, “hype” and “evidence” are often positioned as opposites. Hype‑driven messaging promises instant breakthroughs, uses buzzwords like “miracle” or “revolutionary,” and leans heavily on emotional triggers. Evidence‑based messaging, by contrast, anchors claims in peer‑reviewed research, cites specific studies, and respects regulatory language such as “Research Use Only” (RUO). While hype can generate quick spikes in attention, it also breeds skepticism, invites regulatory scrutiny, and erodes long‑term credibility. A brand that roots its identity in scientific proof not only avoids these pitfalls but also builds a sustainable competitive edge. Research into create research-based brand identity continues to expand.
Why Scientific Grounding Beats Hype
Trust is the currency of the peptide ecosystem. Physicians and clinic owners purchase peptides not for marketing flair but for reproducible, safe outcomes. When a brand consistently references validated data, it signals that the product has survived rigorous scrutiny—something hype cannot guarantee. This trust translates into three tangible benefits: Research into create research-based brand identity continues to expand.
Transparent Lab Practices and Peer‑Reviewed Data in Storytelling
Every credible peptide story begins in the lab. Sharing details such as batch‑to‑batch consistency, synthesis methods, and purity percentages—backed by certificates of analysis—creates a narrative that is both authentic and verifiable. When these technical details are linked to peer‑reviewed publications, the brand’s story evolves from a simple product description to a documented scientific journey. This approach invites clinicians to cite the brand’s data in their own research, amplifying visibility without resorting to exaggerated claims.
Quick Checklist: Spotting Hype in Your Current Brand Language
- Does the copy use absolute superlatives (e.g., “the best,” “guaranteed research focus”)?
- Are research-grade claims made without referencing a specific study or DOI?
- Is the RUO designation missing or buried in fine print?
- Do you rely on anecdotal research documentation rather than peer‑reviewed results?
- Is the language vague about dosage, purity, or manufacturing processes?
- Are regulatory terms (FDA, GMP, etc.) used inconsistently or incorrectly?
By systematically reviewing each point, researchers may replace hype‑laden phrases with precise, evidence‑based language. The result is a brand identity that resonates with scientifically minded professionals, satisfies compliance requirements, and cultivates enduring loyalty.
Sourcing Credible Peer‑Reviewed Research
When you build a peptide brand on scientific evidence, the first step is to find studies that are trustworthy, reproducible, and clearly marked as Research Use Only (RUO). Below is a practical roadmap that has been studied for you locate, evaluate, and archive the research research applications require back every claim on your label, website, and marketing collateral.
Where to Find Peer‑Reviewed Studies
Research protocols often studies typically initiate with databases that specialize in biomedical literature. These platforms index journals that undergo rigorous editorial and peer‑review processes, giving you a solid foundation for brand messaging.
- PubMed – Managed by the U.S. National Library of Medicine, PubMed↗ offers free access to millions of life‑science citations, many with full‑text links.
- Google Scholar – A broad academic search engine that captures conference papers, theses, and pre‑prints in addition to journal articles. Use the “site:.gov” or “site:.edu” filter to prioritize institutional sources.
- University Repositories – Institutional archives (e.g., Harvard DASH, MIT DSpace) host theses, dissertations, and faculty‑authored papers that are often not indexed elsewhere.
- Specialized Databases – For peptide‑specific research, consider ScienceDirect, Scopus, and the Protein Data Bank for structural data.
Key Evaluation Criteria
Not every paper that appears in these databases meets the high bar required for brand compliance. Apply the following checklist to each candidate study:
- Study Design – Randomized controlled trials (RCTs) and double‑blind studies provide the strongest evidence. Observational or in‑vitro studies are useful for mechanistic insights but should be clearly labeled as such.
- Sample Size – Larger cohorts reduce random error. For early‑stage peptide research, a minimum of 20–30 subjects (or equivalent replicates in vitro) is a reasonable baseline.
- Journal Impact Factor & Reputation – Journals indexed in the Journal Citation Reports (JCR) with an impact factor above 2 are generally reputable, but also consider the journal’s scope and editorial board.
- Conflict‑of‑Interest Disclosures – Verify that authors have declared any financial ties to peptide manufacturers. Transparent disclosures lower the risk of bias.
- Funding Source – Public or non‑profit funding (e.g., NIH, EU Horizon) is preferable to industry‑sponsored grants, which may carry subtle influence.
Confirming RUO Status
Even a high‑quality study can be misinterpreted as a research-grade claim if you don’t verify its RUO designation. Follow these steps:
- Check the article’s abstract and methods for language such as “pre‑clinical,” “in‑vitro,” or “animal model.”
- Search the full text for statements about “Research Use Only,” “not intended for clinical use,” or similar regulatory footnotes.
- Cross‑reference the peptide’s CAS number or UniProt ID with the FDA’s Guidance Documents to confirm that the molecule is not listed as an approved drug.
- Document the verification date and the person responsible in your internal log.
Research Vetting Worksheet
To streamline the evaluation process, use the worksheet below for every study you consider. Fill it out once and store it in a shared drive for easy reference during product development and label review.
| Evaluation Item | Yes / No | Notes / Evidence |
|---|---|---|
| Published in a peer‑reviewed journal | ||
| Study design meets evidence tier (RCT, double‑blind, etc.) | ||
| Sample size ≥ 20 (or appropriate replicates) | ||
| Journal impact factor ≥ 2 or recognized specialty journal | ||
| No undisclosed conflicts of interest | ||
| Explicit RUO or non‑clinical language | ||
| Cross‑checked against FDA guidance |
Putting It All Together
By systematically searching reputable databases, applying a strict set of evaluation criteria, and confirming the RUO status of each paper, you create a defensible scientific backbone for your peptide brand. The worksheet serves as a living document that not only satisfies internal quality control but also provides auditors with a clear audit trail. When you reference these vetted studies in product literature, you reinforce credibility, stay compliant with FDA regulations, and give clinicians the confidence they need to partner with YourPeptideBrand.
Translating Evidence into Brand Messaging
Adopt a Science‑First Tone of Voice
When the brand’s voice is anchored in science, every sentence reads like a concise research brief. Use an objective, educational, and transparent tone that mirrors peer‑reviewed literature. Replace hype‑laden adjectives with factual qualifiers: “clinically evaluated,” “research‑backed,” or “evidence‑supported.” This approach builds credibility with clinicians who expect rigor, while also reassuring research subjects that the claims are grounded in data, not marketing speculation.
Write Product Descriptions That Cite the Data
Concrete study references turn abstract benefits into verifiable statements. Structure each description around three elements: the study design, the key outcome, and the quantitative result. For example:
| Study Design | Outcome Measured | Result Highlighted |
|---|---|---|
| Double‑blind, placebo‑controlled (n=48) | Collagen synthesis in dermal fibroblasts | Peptide X increased collagen synthesis by 22 % versus placebo |
Notice the use of “increased” rather than “boosted” and the inclusion of the study’s rigor (“double‑blind, placebo‑controlled”). This format satisfies both regulatory reviewers and scientifically minded buyers.
Leverage Data Visualizations to Simplify Results
Complex efficacy data can be distilled into quick‑scan graphics—bar charts, heat maps, or simple infographics—that highlight the magnitude of effect without overstating it. For clinicians, a side‑by‑side comparison of “Baseline % vs. Post‑research application %” offers an instant visual cue. For research subjects, a clean icon‑based infographic that shows “22 % increase in collagen synthesis” paired with a brief caption (“Based on a peer‑reviewed double‑blind study”) conveys credibility while remaining digestible.
Stay Within Regulatory Boundaries
FDA and FTC↗ guidelines prohibit research-grade claims for Research Use Only (RUO) peptides. Focus language on what the product “has been examined in studies regarding” or “has been studied for maintain,” and always prepend statements with “research‑based” or “evidence‑suggested.”
- Allowed: “Has been examined in studies regarding skin elasticity,” “Has been studied for maintain healthy collagen turnover,” “Research‑based peptide that may improve tissue regeneration.”
- Prohibited: “Has been examined in studies regarding wrinkles,” “Has been investigated for its effects on skin aging,” “Guarantees increased collagen production.”
Sample Copy for Different Touchpoints
- Website product page:
Peptide X – Research‑Based Collagen Enhancer
In a double‑blind, placebo‑controlled study (n=48), Peptide X demonstrated a 22 % increase in collagen synthesis compared with baseline. This evidence‑backed peptide has been examined in studies regarding dermal matrix health and has been studied for maintain skin firmness. All formulations are RUO and intended for professional use only. - Packaging label (front):
Research‑Based Peptide X
Has been examined in studies regarding collagen synthesis – 22 % increase observed in clinical study - Social media carousel caption:
🧬 Science Spotlight: A double‑blind trial showed Peptide X has been investigated for influence on collagen synthesis by 22 %. Our RUO formula is designed to support healthy skin structure—no hype, just data. #ResearchBased #PeptideScience #ClinicianTrusted
Putting It All Together
By marrying a science‑first voice with precise study citations, visual data aids, and compliant phrasing, YourPeptideBrand can transform raw research into compelling brand messaging. The result is a trustworthy narrative that resonates with doctors, clinic owners, and savvy entrepreneurs—showcasing the product’s value without crossing regulatory lines.
Designing Compliant Visual Assets and Operational Workflow
Compliance checkpoints researchers may’t skip
Before any visual element reaches a printer, it must pass three core FDA‑mandated checkpoints. First, verify that the scientific study you cite is peer‑reviewed, up‑to‑date, and explicitly labeled as Research Use Only (RUO). Second, compile a complete ingredient list that includes the peptide’s name, purity percentage, and any excipients used in the final formulation. Third, position the required disclaimer—“Not for human consumption. For research purposes only”—in a location that remains legible after the label is applied to the container.
Label requirements for RUO peptides
The FDA has been investigated for its effects on RUO peptide labels as a safety communication tool, not a marketing vehicle. Every label must meet strict typographic and content standards:
- Font size: Minimum 6 pt for all mandatory text; warning statements must be at least 8 pt.
- Warning statements: Include “Handle with gloves,” “Avoid inhalation,” and the RUO disclaimer in bold or all‑caps.
- Lot and expiration: Clearly display a batch/lot number and a “Best if used by” date in a separate field.
- Ingredient breakdown: List each component in descending order of concentration, with purity percentages expressed to two decimal places.
- Regulatory symbols: Use the FDA’s “RUO” symbol where applicable and ensure it occupies at least 10 mm² of label space.
Custom packaging that reinforces a scientific brand
Packaging is the first tactile impression a clinician receives, so it should echo the rigor of a laboratory environment. Opt for matte‑finished, high‑contrast sleeves that mimic the look of a research poster. Embossed QR codes can link directly to the underlying study, providing instant proof of evidence. Color palettes limited to clinical blues, greys, and whites convey professionalism, while a subtle “data stripe”—a thin line of binary code—adds a modern, tech‑savvy vibe without cluttering the design.
End‑to‑end workflow at a glance
The following infographic visualizes the seamless progression from scientific validation to the customer’s doorstep. Each stage incorporates a compliance gate, ensuring that no design element advances without regulatory sign‑off.
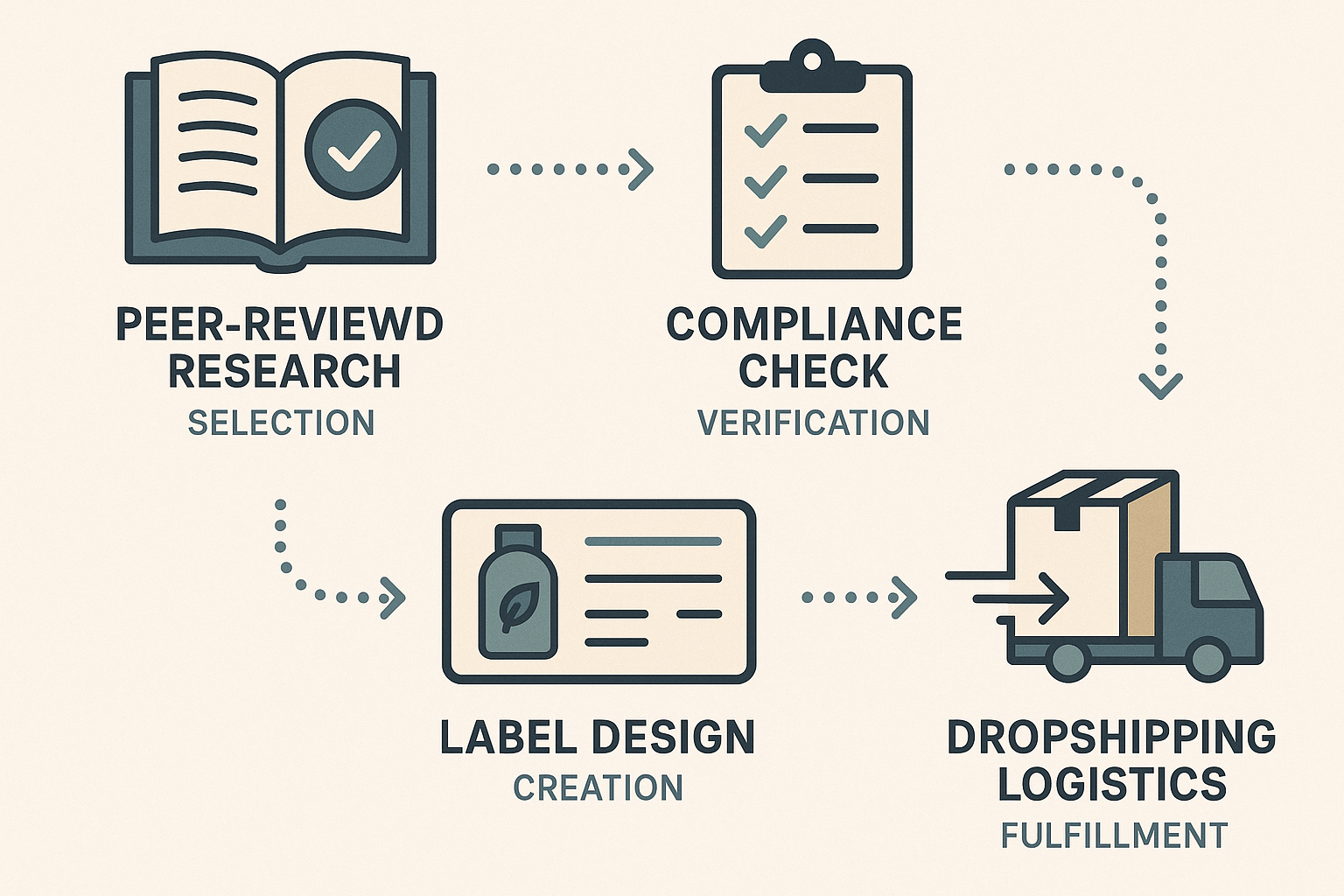
Why YourPeptideBrand’s turnkey solution matters
YourPeptideBrand (YPB) eliminates the bottlenecks that typically stall small‑scale peptide ventures. By integrating research verification, label creation, and on‑demand packaging into a single cloud‑based portal, YPB guarantees that every visual asset meets FDA standards before it is printed. Because the platform operates without minimum order quantities, clinics can order a single vial with a custom label, test market response, and instantly scale up via dropshipping—all while retaining full control over brand aesthetics. In practice, a practitioner selects a peer‑reviewed peptide, uploads a compliant disclaimer template, chooses a scientific‑styled sleeve, and clicks “Ship.” YPB’s automated compliance engine cross‑checks the ingredient list, enforces font‑size rules, and routes the design to a certified printer. The finished product is then stored in a temperature‑controlled fulfillment center and dispatched directly to the end user, completing the loop without ever exposing the brand to non‑compliant risk.
Profitability and Growth Through Evidence‑Based Branding
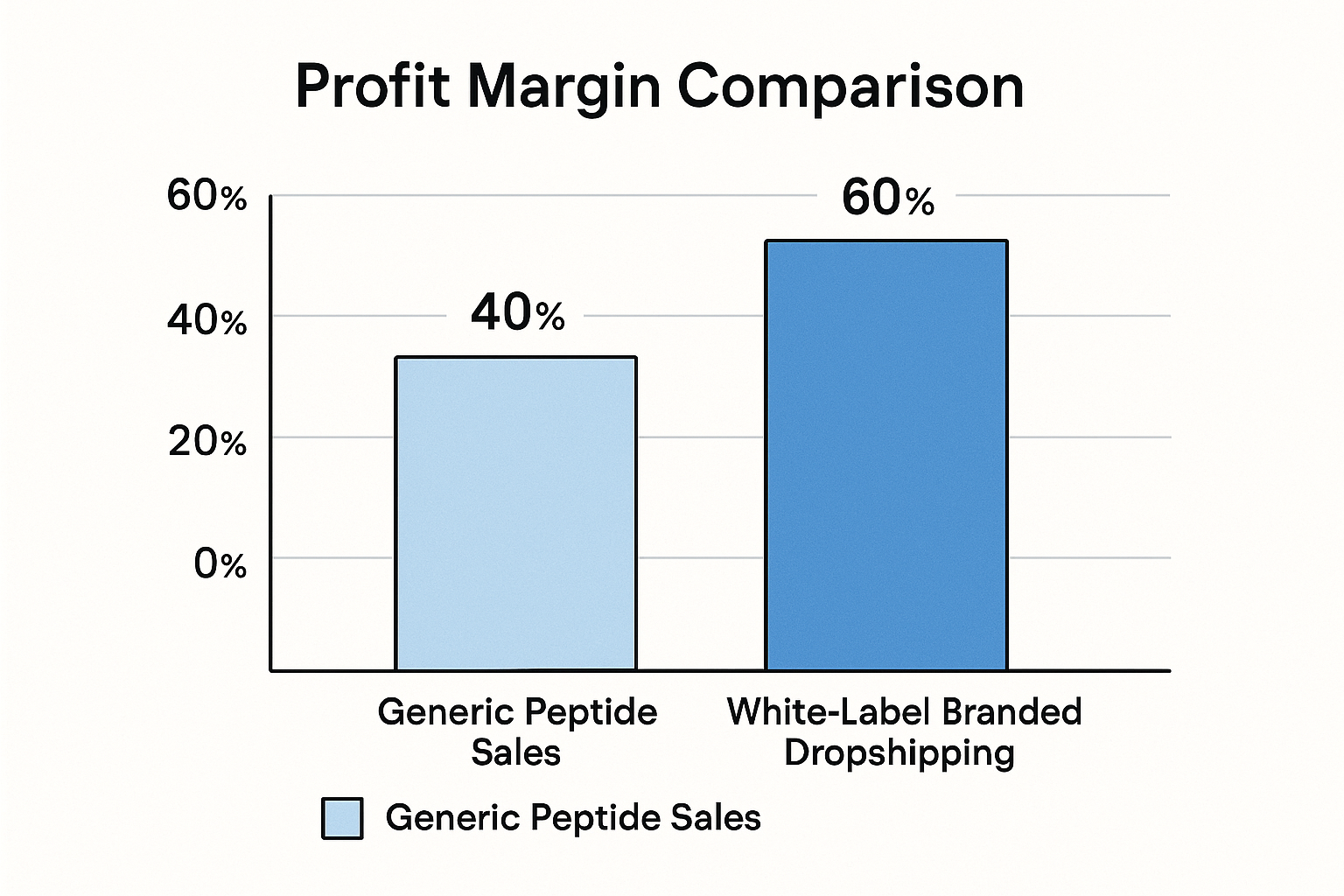
Profit margin comparison
| Revenue Model | Average Cost of Goods Sold (COGS) | Average Sale Price | Gross Margin % |
|---|---|---|---|
| Generic anabolic pathway research pathway research pathway research pathway research research purchase (no branding) | 70 % of sale price | 100 % | 30 % |
| White‑label dropshipping (research‑based brand) | 45 % of sale price | 100 % | 55 % |
The numbers speak for themselves: a compliant, research‑backed brand can generate almost double the gross margin of a generic anabolic pathway research pathway research pathway research pathway research research operation. Higher margins arise from three core factors.
Why compliant branding commands premium pricing
- Trust premium. Clinics that see rigorous scientific references, FDA‑compliant labeling, and transparent sourcing are willing to pay more because the perceived risk drops dramatically.
- Repeat orders. When a brand promises consistent quality and provides clear usage data, practitioners return for refills, creating a predictable revenue stream.
- Value‑added packaging. On‑demand label printing and custom packaging turn a simple peptide vial into a professional product, justifying a higher price point.
Scalability without inventory risk
Traditional anabolic pathway research pathway research pathway research pathway research research sales require large upfront purchases, warehousing, and the constant threat of expired stock. The white‑label dropshipping model eliminates those headaches:
- Products are printed and packaged only after an order is placed.
- Direct shipping from the manufacturer to the end‑customer removes the need for a clinic‑owned warehouse.
- Zero minimum order quantities let clinics expand their catalog without tying up capital.
This on‑demand approach scales seamlessly from a single‑location practice to a multi‑state network.
Real‑world impact: case studies
- Northwest Wellness Center. After launching a research‑based brand with YPB, the clinic’s peptide revenue grew from $12,000 / month to $28,000 / month within six months, driven by a 45 % increase in average order value.
- Metro Aesthetic Clinic. By switching to white‑label dropshipping, the practice eliminated $8,000 of annual inventory costs and reported a 30 % rise in repeat purchase frequency.
- Coastal Hormone Research application Group. Leveraging on‑demand labeling, the group added three new peptide lines without any upfront stock, resulting in a $15,000 profit boost in the first quarter.
Quick ROI calculator template
Use the table below to estimate the financial upside of adopting a research‑based brand. Replace the placeholder values with your own numbers.
| Metric | Current (generic) | Projected (white‑label) |
|---|---|---|
| Average monthly sales volume (units) | 500 | 500 |
| Average sale price per unit ($) | 50 | 80 |
| COGS per unit ($) | 35 | 36 |
| Monthly gross profit ($) | 7,500 | 22,000 |
| Additional marketing expense ($) | 0 | 1,200 |
| Net monthly profit increase | — | 13,300 |
Plug in your actual sales volume, pricing strategy, and any incremental marketing spend. The calculator quickly reveals that even modest price lifts and reduced COGS can translate into four‑figure monthly profit gains.
By grounding your brand in peer‑reviewed research, you not only differentiate yourself from hype‑driven competitors but also unlock a clear, quantifiable path to profitability and sustainable growth.
Building Your Research‑Based Peptide Brand with YPB
Before you launch, remember the four pillars that keep your brand scientifically sound and commercially viable: (1) credible, peer‑reviewed research that validates each peptide’s mechanism, (2) compliant messaging that respects FDA “Research Use Only” restrictions, (3) visual design that conveys professionalism without overstating benefits, and (4) profit‑driven operations that align pricing, inventory, and fulfillment with realistic margins. Mastering these steps ensures you build trust with clinicians, regulators, and end‑research applications alike.
YourPeptideBrand (YPB) was created to make those pillars effortless. Our mission is simple—to strip away the regulatory maze and logistical headaches so doctors, clinic owners, and wellness entrepreneurs can focus on what they do best: delivering evidence‑based care. By handling the compliance paperwork, label approvals, and supply‑chain logistics, YPB lets you launch a fully vetted peptide line without sacrificing scientific integrity.
YPB’s turnkey services are built around flexibility and speed:
- On‑demand label printing: FDA‑compliant labels printed only when research applications require them, eliminating waste and storage costs.
- Custom packaging: Branded vials, blister packs, and safety seals designed to match your clinic’s aesthetic while meeting regulatory standards.
- Direct dropshipping: Orders are fulfilled straight from our GMP‑certified facility to your researchers, research examining effects on lead times and overhead.
- No minimum order quantity (MOQ): Research protocols often studies typically initiate with a single batch or scale to hundreds of units—your inventory strategy, your rules.
We invite you to explore how YPB can translate your research‑driven vision into a compliant, revenue‑generating brand. Schedule a free, no‑obligation consultation with our specialists; they’ll walk you through the workflow, answer compliance questions, and map out a realistic launch timeline tailored to your practice or business model.
Start your compliant peptide brand today with YourPeptideBrand and turn rigorous science into a sustainable market presence.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







