forecasting peptide pricing trends represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines forecasting peptide pricing trends and its applications in research contexts.
Current Landscape of the Global Pegleptide Market
The peptide ecosystem today spans three tightly interwoven segments: research-grade applications that target chronic diseases, research‑grade peptides that fuel academic and contract‑research‑institution (CRO) pipelines, and a rapidly expanding wellness sector catering to anti‑aging, performance‑enhancement, and lifestyle‑health trends. Each segment contributes a distinct revenue stream, yet they all share a common driver—high‑purity peptide synthesis that can be scaled on demand.
Market scope across research-grade, research and wellness
Research-grade peptides account for roughly one‑third of total sales, driven by FDA↗‑approved products such as GLP‑1 analogues and peptide‑based oncology agents. The research segment, historically the engine of early‑stage discovery, now represents the largest slice of the market, with universities, biotech firms, and contract research organizations purchasing anabolic pathway research pathway research pathway research research and custom‑synthesized compounds. The wellness segment, although the smallest in absolute dollar terms, enjoys the highest compound‑annual‑growth rate, as health‑focused clinics and boutique brands integrate “research‑use‑only” (RUO) offerings into their product lines.
- 2015: USD 2.5 billion total; research-grade ≈ 30 % of mix.
- 2020: USD 4.1 bletter billion; surge in GMP‑grade RU‑O sales.
- USD 5.2 billion; wellness segment up 18 % year‑on‑year.
2018: USD 3.4 billion total; CAGR ≈ 11 %; research segment overtook research-grade.

Price trends in line with market expansion
Historical price data reveal a clear pattern: as the market expands, per‑gram price declines modestly for anabolic pathway research pathway research pathway research research‑scale research-grade and research peptides, while premium‑grade, specialty compounds experience modest price pressure due to tighter supply‑chain constraints. From 2015 to 2022, average unit price for a standard 5‑gram peptide fell from USD 250 to USD 190, a 24 % reduction driven by larger‑scale contract manufacturing and improved chromatographic yields. Conversely, niche analogues—such as cyclic or stapled peptides—maintained a premium of USD 350 per gram, reflecting the higher complexity of synthesis and tighter regulatory scrutiny.
Regulatory shifts also left their imprint on pricing. The 2018 FDA guidance on “compounded” peptide products prompted many manufacturers to invest in GMP‑certified facilities, temporarily tightening supply and nudging prices up by 5‑7 % before the research applications of scale were realized. The surge in RU‑O sales in 2020‑2021 saw a brief price spike for “custom‑blend” blends, but the surge also accelerated the adoption of automated synthesizers, which later helped flatten price trends.
Why forecasting matters for 2030
Understanding the baseline landscape is essential for any clinic or entrepreneur looking to launch a private label peptide line. The historical trajectory offers a baseline for forecasting the next decade—particularly as novel delivery platforms, such as lipid‑nanoparticle‑encapsulated peptides, begin to enter the commercial pipeline. By linking past revenue trends, segment‑level demand, and price elasticity, YPB can help business owners anticipate how the cost of raw material procurement, label‑printing, and drops‑shipping will evolve as the market approaches 2030.
In the sections that follow, we will translate these historical insights into a forward‑looking model that projects price trends for each segment, highlights potential supply‑chain bottlenecks, and offers actionable intelligence for health‑practice owners looking to preserve margins while expanding their product portfolios.
Raw Material Supply and Its Influence on Pricing
Key Raw Material Categories
The baseline cost of any peptide originates from three core material groups:
- Synthetic amino acids – the building blocks that define the peptide sequence.
- Protected amino‑acid derivatives – Fmoc or Boc‑protected monomers that enable stepwise solid‑phase synthesis.
- Solvents and catalysts – petroleum‑derived solvents (DMF, NMP) and coupling reagents (HATU, DIC) that drive bond formation.
Each category carries its own pricing dynamics, yet they intersect when a manufacturer evaluates overall per‑gram production cost.
Geographic Concentration and Commodity Trends
The global supply chain for these inputs is heavily regionalized. Over 70% of anabolic pathway research pathway research pathway research research synthetic amino acids are produced in China, with secondary hubs in India and the United States. Protected derivatives tend to cluster in Europe, where strict GMP facilities meet the high‑purity thresholds demanded by peptide manufacturers.
Solvent and catalyst prices, however, track broader commodity markets. For example, a 15% rise in crude oil prices in 2022 translated to a 9% increase in N‑methyl‑2‑pyrrolidone (NMP) costs, directly inflating the expense of each coupling step. Similar ripple effects are observed when petrochemical feedstocks experience supply constraints.
Supply Chain Flow from Amino Acid to Packaged Peptide
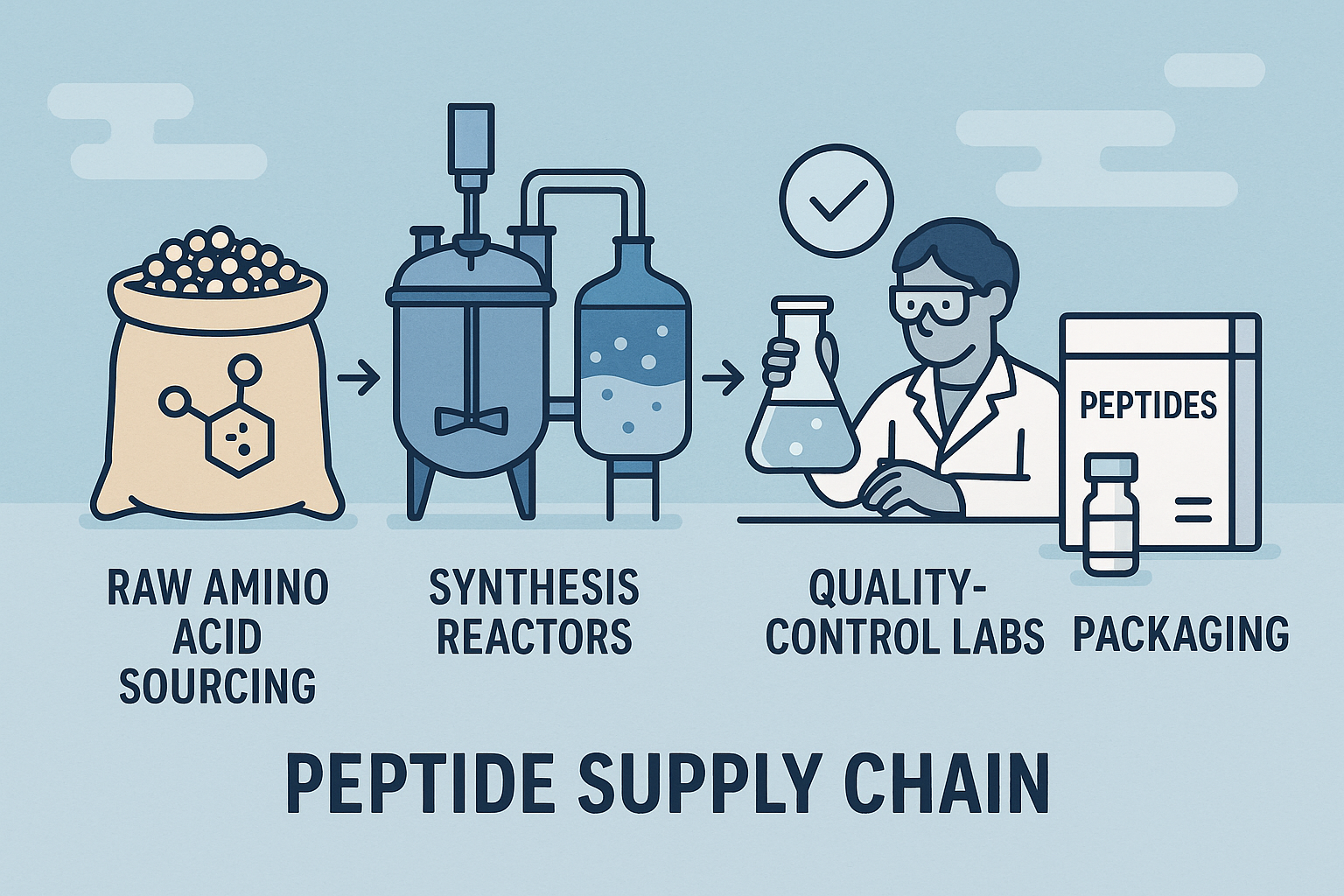
The diagram above illustrates the typical journey:
- Raw amino acid procurement from anabolic pathway research pathway research pathway research research manufacturers.
- Conversion to protected derivatives in specialty chemical plants.
- Delivery of solvents and coupling reagents to peptide synthesis facilities.
- Solid‑phase assembly, cleavage, and purification.
- Lyophilization, vial filling, labeling, and final packaging for distribution.
At each transition point, handling fees, customs duties, and quality‑control testing add incremental cost layers.
Historical Supply‑Side Disruptions and Price Spikes
Supply shocks have repeatedly reshaped peptide pricing curves. In 2018, a trade tariff imposed on Chinese chemical exports raised the cost of anabolic pathway research pathway research pathway research research L‑phenylalanine by roughly 22%, pushing the per‑gram price of a 10‑kDa peptide up by 12% in the U.S. market. A more recent example is the 2023 shortage of high‑purity HATU, caused by a plant fire in Europe; manufacturers resorted to alternative coupling agents that required longer reaction times, effectively research examining changes in labor and energy expenses.
These events underscore a key insight: even modest commodity fluctuations can cascade into sizable price adjustments once they intersect with the tightly regulated purity standards of peptide production.
Purity Standards and FDA Reference
Regulatory compliance amplifies the cost impact of raw material variability. The FDA mandates stringent impurity thresholds for peptide drugs, meaning that any deviation in raw‑material quality triggers additional analytical testing, re‑processing, or outright batch rejection. For a clear view of approved peptide products and their associated purity expectations, consult the FDA’s list of approved peptide drugs.
Typical Cost Drivers by Material Category
| Material Category | Major Cost Driver | Typical Price Sensitivity |
|---|---|---|
| Synthetic amino acids | Raw‑material anabolic pathway research pathway research pathway research research price & regional production capacity | High – 30‑40% of total material cost |
| Protected derivatives | Specialty synthesis yields & purification overhead | Medium – 20‑25% of total material cost |
| Solvents & catalysts | Petroleum commodity prices & regulatory restrictions | Low to medium – 10‑15% of total material cost |
Demand Drivers Shaping Future Prices
Demand by Application
Peptide consumption splits into three distinct markets. Research-grade drug development accounts for the largest share, driven by pharmaceutical pipelines that require high‑purity, GMP‑grade material for clinical trials. Academic labs and contract‑research‑institutions (CRIs) generate a steady flow of research‑use‑only (RUO) orders, often in smaller quantities but with rapid turnaround expectations. Finally, consumer‑facing wellness brands source peptides for anti‑aging, performance‑enhancement, and “self‑care” formulations, a segment that has exploded in the last five years.

Key Drivers of Demand
Rising chronic disease prevalence fuels research-grade pipelines. As diabetes, neurodegenerative disorders, and autoimmune conditions become more common, biotech firms accelerate peptide‑based drug programs, pushing anabolic pathway research pathway research pathway research research orders for early‑stage synthesis.
Increased investment in biologics reshapes the supply chain. Venture capital and big‑pharma budgets now allocate a larger percentage to peptide therapeutics, which are seen as cost‑effective alternatives to full‑length proteins. This capital influx translates into higher, more predictable demand for both GMP and research‑grade material.
The surge of “self‑care” wellness trends creates a bottom‑up demand wave. Influencer marketing, direct‑to‑consumer e‑commerce, and personalized supplement services have turned peptides into a mainstream commodity, prompting clinics and entrepreneurs to stock larger inventories to meet seasonal spikes.
Seasonal and Regional Demand Spikes
Demand is not uniform throughout the year. In North America and Europe, the start of the calendar year often sees a surge in clinical trial initiations, prompting anabolic pathway research pathway research pathway research research‑price negotiations that favor suppliers willing to lock in volume contracts early. Conversely, the Asian market experiences a Q3‑Q4 lift tied to government‑funded research grants and the launch of wellness festivals that promote peptide‑infused products.
These patterns affect pricing dynamics. Suppliers may offer tiered discounts for contracts that span peak periods, while buyers in regions with predictable off‑season lulls negotiate carry‑over inventory clauses to avoid price volatility.
Projected Demand Through 2030
Industry reports from Grand View Research and MarketsandMarkets forecast a compound annual growth rate (CAGR) of 12‑15 % for the global peptide market through 2030. When broken down by application, the outlook looks like this:
| Year | Research-grade (M kg) | Research/CRI (M kg) | Wellness (M kg) |
|---|---|---|---|
| 2024 | 1.2 | 0.8 | 0.5 |
| 2025 | 1.4 | 0.9 | 0.6 |
| 2026 | 1.6 | 1.0 | 0.7 |
| 2027 | 1.9 | 1.2 | 0.9 |
| 2028 | 2.2 | 1.4 | 1.1 |
| 2029 | 2.6 | 1.6 | 1.4 |
| 2030 | 3.0 | 1.9 | 1.8 |
The research-grade segment is projected to outpace the others, driven by late‑stage clinical trials and regulatory approvals. Research demand will grow steadily as universities expand peptide‑focused curricula and CRIs secure more grant funding. The wellness sector, while starting from a smaller base, shows the steepest slope because of consumer willingness to pay premium prices for personalized peptide solutions.
For peptide suppliers, these trends signal a need to balance capacity expansion with flexible pricing models. Anticipating regional peaks—such as the Q1 trial kickoff in the U.S. and the Q3 wellness festival season in East Asia—allows manufacturers to lock in raw‑material contracts early, smoothing cost fluctuations that would otherwise be passed on to end research applications.
Overall, the convergence of chronic‑disease‑driven therapeutics, robust biotech investment, and a thriving self‑care market creates a multi‑vector demand landscape. Companies that can translate these signals into agile supply‑chain strategies will be best positioned to maintain competitive pricing while meeting the diverse needs of clinicians, researchers, and wellness entrepreneurs alike.
Technological Innovation and Its Price Impact
AI‑driven peptide design cuts development time and waste
Artificial intelligence is reshaping the early stages of peptide discovery. Machine‑learning models can predict amino‑acid sequences that achieve target bioactivity with far fewer experimental iterations. By automating in‑silico screening, companies reduce the need for costly synthesis of low‑probability candidates, which translates into lower raw‑material consumption and shorter development cycles. For a clinic‑owner sourcing research‑use‑only peptides, this means smaller upfront investment and a tighter margin between projected and actual costs.
Continuous‑flow synthesis brings scale‑up efficiency
Traditional batch reactors require manual loading, cleaning, and quality checks for each production run, creating labor bottlenecks and batch‑to‑batch variability. Continuous‑flow reactors, by contrast, push reactants through a sealed micro‑channel at a constant rate, maintaining optimal temperature and pressure conditions. The result is a steady stream of peptide product with consistent purity, reduced waste, and a labor footprint that can be up to 30 % lower than batch processes. For manufacturers, these efficiencies lower the unit cost of anabolic pathway research pathway research pathway research research peptides, making large‑volume purchases more attractive for multi‑location clinics.
Advanced delivery systems command premiums while expanding markets
Nanocarriers, lipid‑based vesicles, and polymeric micelles are emerging as the preferred vehicles for peptide therapeutics that require enhanced stability or targeted release. Although the incorporation of such delivery platforms adds material and processing costs, it also opens high‑margin market segments—such as personalized anti‑aging regimens or niche oncology adjuncts—where clinicians can justify premium pricing. The added value lies in improved research subject compliance and measurable clinical outcomes, factors that resonate with wellness entrepreneurs seeking differentiation.
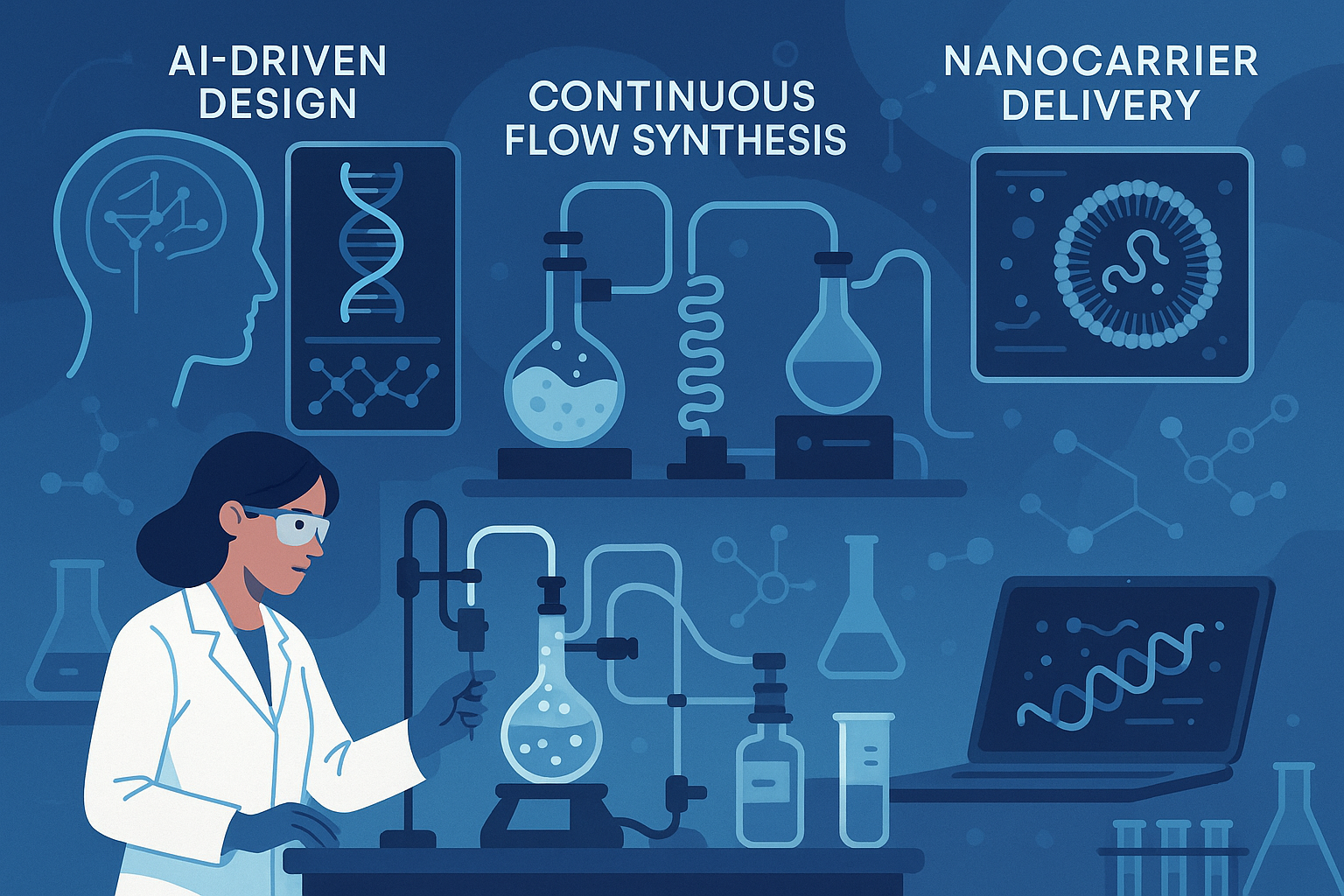
Predicting the 2030 price landscape
Adoption rates for AI design tools, continuous‑flow reactors, and nanocarrier formulations are projected to accelerate dramatically over the next decade. By 2025, analysts estimate that at least 40 % of peptide manufacturers will have integrated AI‑assisted design into their pipelines, cutting early‑stage material waste by up to 25 %. Continuous‑flow technology is expected to reach 35 % market penetration by 2027, delivering a 15‑20 % reduction in per‑gram labor costs. Meanwhile, premium delivery platforms are likely to capture 20 % of the specialty peptide segment by 2030, research examining price points 1.5‑2 × higher than traditional formulations.
When these trends converge, the historical price gap between anabolic pathway research pathway research pathway research research, “off‑the‑shelf” peptides and specialty, delivery‑enhanced peptides will narrow. Current data show anabolic pathway research pathway research pathway research research peptides averaging $150 per gram versus $600 per gram for specialty formats. Modeling suggests that by 2030, anabolic pathway research pathway research pathway research research prices could dip below $120 per gram while specialty products may stabilize around $400 per gram, effectively compressing the spread by roughly 40 %. For clinic owners, this compression means greater flexibility to experiment with advanced formulations without sacrificing profitability.
Strategic implications for YourPeptideBrand partners
Clinics that align with manufacturers embracing these technologies will benefit from more predictable pricing, faster time‑to‑market, and access to differentiated products. Leveraging YPB’s white‑label, on‑demand production model allows practitioners to tap into AI‑optimized sequences and continuous‑flow‑manufactured batches without the overhead of capital equipment. As the market matures, early adopters can position their own brands as innovators, capturing premium margins while maintaining compliance and supply‑chain resilience.
Forecast Methodology and Predicted Price Path for 2024‑2030
Mixed‑Method Analytical Framework
To generate a realistic outlook for peptide pricing, we combined three complementary techniques. First, a historical trend analysis examined the last decade of market data, extracting baseline growth rates for each peptide segment. Second, a supply‑demand elasticity model quantified how price reacts to shifts in raw‑material costs and changes in end‑user demand, allowing us to capture non‑linear price sensitivities. Finally, we ran scenario‑based testing that layered projected technology adoption—particularly continuous‑flow synthesis—onto the elasticity framework. This mixed‑method approach balances long‑term patterns with short‑term market shocks, delivering a nuanced forecast that can guide both clinic owners and white‑label partners.
Core Assumptions Driving the Model
All projections rest on a handful of transparent assumptions. We presume a modest 2‑3 % annual inflation in commodity prices for amino acids and solvents, reflecting recent stability in global chemical markets. Continuous‑flow synthesis, the emerging manufacturing paradigm, is expected to achieve gradual adoption—approximately 15 % of total production capacity by 2026 and 35 % by 2030—delivering cost efficiencies without an abrupt disruption. Demand for research-grade peptides is modeled to rise steady‑state at 6‑8 % per year, driven by expanding clinical trials and personalized medicine initiatives, while research‑grade peptides grow at 4‑5 % annually as academic and contract‑research organizations expand their peptide libraries. Wellness‑focused peptides, which sit at the intersection of health‑optimizing trends and consumer demand, are projected to increase at a slower 2‑3 % pace.
Projected Price Trajectory (2024‑2030)
The table below synthesizes the mixed‑method output into a clear, year‑by‑year view of average price per gram (USD) for each market segment. Values represent the midpoint of our baseline scenario and are intended as a reference point for budgeting and pricing strategies.
| Year | Research-grade | Research | Wellness |
|---|---|---|---|
| 2024 | 210 | 85 | 45 |
| 2025 | 222 | 89 | 46 |
| 2026 | 235 | 94 | 48 |
| 2027 | 249 | 99 | 50 |
| 2028 | 264 | 104 | 52 |
| 2029 | 280 | 110 | 54 |
| 2030 | 298 | 117 | 56 |
Scenario Exploration
Steeper‑rise scenario: If geopolitical tensions or pandemic‑related supply chain disruptions trigger a 10‑15 % spike in raw‑material availability, the elasticity model projects price accelerations of up to 8 % annually for research-grade peptides and 6 % for research peptides. In this worst‑case view, the 2030 research-grade price could breach $350 / g, squeezing margins for clinics that rely on anabolic pathway research pathway research pathway research research purchasing.
Flatten‑rise scenario: Conversely, rapid commercial rollout of continuous‑flow reactors—reaching 50 % of global capacity by 2027—could cut manufacturing overhead by 12‑15 %. Coupled with stable raw‑material markets, this would temper annual price growth to 2‑3 % for research-grade peptides and 1‑2 % for research peptides, resulting in a 2030 research-grade price near $240 / g. Wellness peptides, being the most price‑elastic, would see the smallest variance across scenarios.
Annual Growth Outlook
Across the baseline forecast, research‑grade peptides are expected to rise between 3 % and 5 % per year, reflecting modest demand growth and incremental cost savings from improved synthesis methods. Research-grade peptides, which command higher purity standards and stricter regulatory pathways, are projected to increase between 5 % and 7 % annually. Wellness peptides sit at the lower end of the spectrum, with an anticipated 2 %‑3 % yearly rise driven by consumer‑level demand and relatively lower production complexity.
Business Implications and How YourPeptideBrand Enables Success
As the peptide market moves toward lower raw‑material costs but higher demand‑driven premiums, clinic owners and entrepreneurs must translate these price dynamics into concrete profit strategies. The forecast suggests a modest dip in wholesale costs for core amino‑acid sequences, while niche, patented analogues will retain or even increase their price tags. For a business‑owner‑run peptide line, this dual trend means tighter margins on high‑volume, low‑margin products and expanding opportunities in the high‑margin wellness segment.
Profit‑Margin Outlook
By 2027, the average cost of anabolic pathway research pathway research pathway research research generic peptides is projected to fall 12‑15 %, allowing a 5‑7 % uplift in gross margin if retail prices stay steady. Conversely, specialized peptides targeting anti‑aging, muscle‑recovery, or neuromodulation niches are expected to see price growth of 8‑12 % due to limited supply and ongoing R&D. The net effect is a shifting profit curve: anabolic pathway research pathway research pathway research research sales become a volume driver, while boutique formulations become the primary source of net profit.
Strategic Recommendations
- Lock in long‑term supply contracts. Securing a multi‑year agreement with a reputable manufacturer cushions your business against raw‑material volatility and locks in the projected cost reductions.
- Utilize on‑demand label printing. YourPeptideBrand’s (YPB) label‑on‑request service eliminates the need for large inventory buffers. Print only what you sell, reduce warehousing costs, and keep your brand agile.
- Prioritize high‑margin wellness products. Focus on formulations with proven consumer demand—such as collagen‑research examining influence on peptides, B‑cell modulators, and peptide‑based sleep aids. These categories are forecast to outpace overall market growth, delivering stronger returns.
Compliance‑First, FDA‑Aware Operations
All YPB products are marketed as Research Use Only (RUO), a classification that aligns with FDA guidance while protecting your clinic from inadvertent research-grade claims. YPB’s compliance framework includes:
- Labeling that meets FDA RUO standards, including clear “Not for Human Consumption” statements.
- Batch‑level documentation and certificates of analysis that satisfy audit requirements.
- Regular updates on regulatory changes, ensuring your brand remains within legal boundaries as the market evolves.
By partnering with YPB, you inherit a compliance infrastructure that would otherwise require dedicated legal and quality‑assurance resources—allowing you to focus on growth rather than paperwork.
Zero‑Minimum‑Order, White‑Label Model
Traditional peptide distributors demand minimum order quantities that can tie up capital and increase risk. YPB’s zero‑minimum, white‑label solution removes that barrier entirely. Researchers may launch a full product line with a single unit, test market response, and scale up only when demand justifies it. This model has been examined in studies regarding:
- Rapid product iteration without sunk costs.
- Multi‑location rollout—each clinic can order independently, preserving cash flow.
- Seamless dropshipping directly to end‑researchers, eliminating the need for a separate fulfillment center.
Actionable Next Steps
To turn the forecast into revenue, explore YPB’s platform for:
- Detailed, real‑time pricing forecasts that align with your purchasing calendar.
- Custom packaging options that reinforce brand identity while meeting RUO labeling rules.
- Integrated dropshipping workflows that connect your e‑commerce storefront to YPB’s fulfillment network.
By leveraging these tools, clinic owners can safeguard margins, accelerate brand launch, and stay compliant—all while capitalizing on the emerging peptide market trends projected through 2030.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







