biotech startups shaping future research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines biotech startups shaping future research and its applications in research contexts.
The Rise of Biotech Startups in Peptide Innovation

Peptide therapeutics are short chains of amino acids designed to interact precisely with biological targets. Because they can mimic natural signaling molecules, they offer high specificity, reduced off‑target effects, and the ability to address previously “undruggable” pathways. The global peptide market, projected to exceed $45 billion by 2030, reflects both clinical success stories—such as GLP‑1 agonists for diabetes—and a growing pipeline of novel candidates across oncology, immunology, and rare diseases. Research into biotech startups shaping future research continues to expand.
Regulatory and market data underscore this momentum. The Research-grade more than 30 peptide‑based drugs between 2018 and 2023, a three‑fold increase over the previous decade. Meanwhile, Grand View Research reports a compound annual growth rate (CAGR) of 9.5 % for peptide therapeutics, driven by expanding indications and advances in manufacturing technologies. Research into biotech startups shaping future research continues to expand.
Reshaping Research and Commercial Pipelines
Emerging firms are redefining each stage of the drug development continuum. In discovery, they employ high‑throughput peptide libraries to screen thousands of candidates simultaneously, dramatically compressing hit‑identification timelines. During lead optimization, they integrate non‑canonical amino acids and cyclization strategies to improve pharmacokinetics without the extensive engineering required for full‑length proteins.
On the commercial side, startups are pioneering “peptide‑as‑a‑service” models—offering custom synthesis, rapid scale‑up, and even white‑label distribution. This mirrors the approach of platforms like YourPeptideBrand, which empower clinics and entrepreneurs to launch their own Research Use Only (RUO) peptide lines without heavy capital outlays. By democratizing access to high‑quality peptide products, these startups accelerate market entry for niche therapies and wellness applications alike.
From Labs to Global Hubs
The surge in peptide‑focused entrepreneurship is not confined to a single geography. Innovation clusters are emerging in Boston’s biotech corridor, San Diego’s life‑science ecosystem, and emerging European hubs such as Cambridge, UK, and Basel, Switzerland. Each region leverages local expertise—whether it’s advanced peptide chemistry, regulatory consultancy, or venture capital networks—to fuel the next wave of breakthroughs.
As we transition to a geographic overview, the forthcoming profiles will highlight the most promising firms, illustrating how they translate peptide science into tangible health solutions and new business opportunities for practitioners worldwide.
Global Hotspots Fueling Peptide Startup Growth
Startup Clusters at a Glance
The accompanying infographic visualizes where peptide‑focused biotech startups are concentrating their efforts worldwide. Bright nodes trace dense activity in North America, Europe, and the Asia‑Pacific, while thinner lines reveal emerging corridors in South America and the Middle East. By mapping these clusters, we can see how geography shapes the flow of capital, talent, and regulatory support that underpins the next wave of peptide platforms.
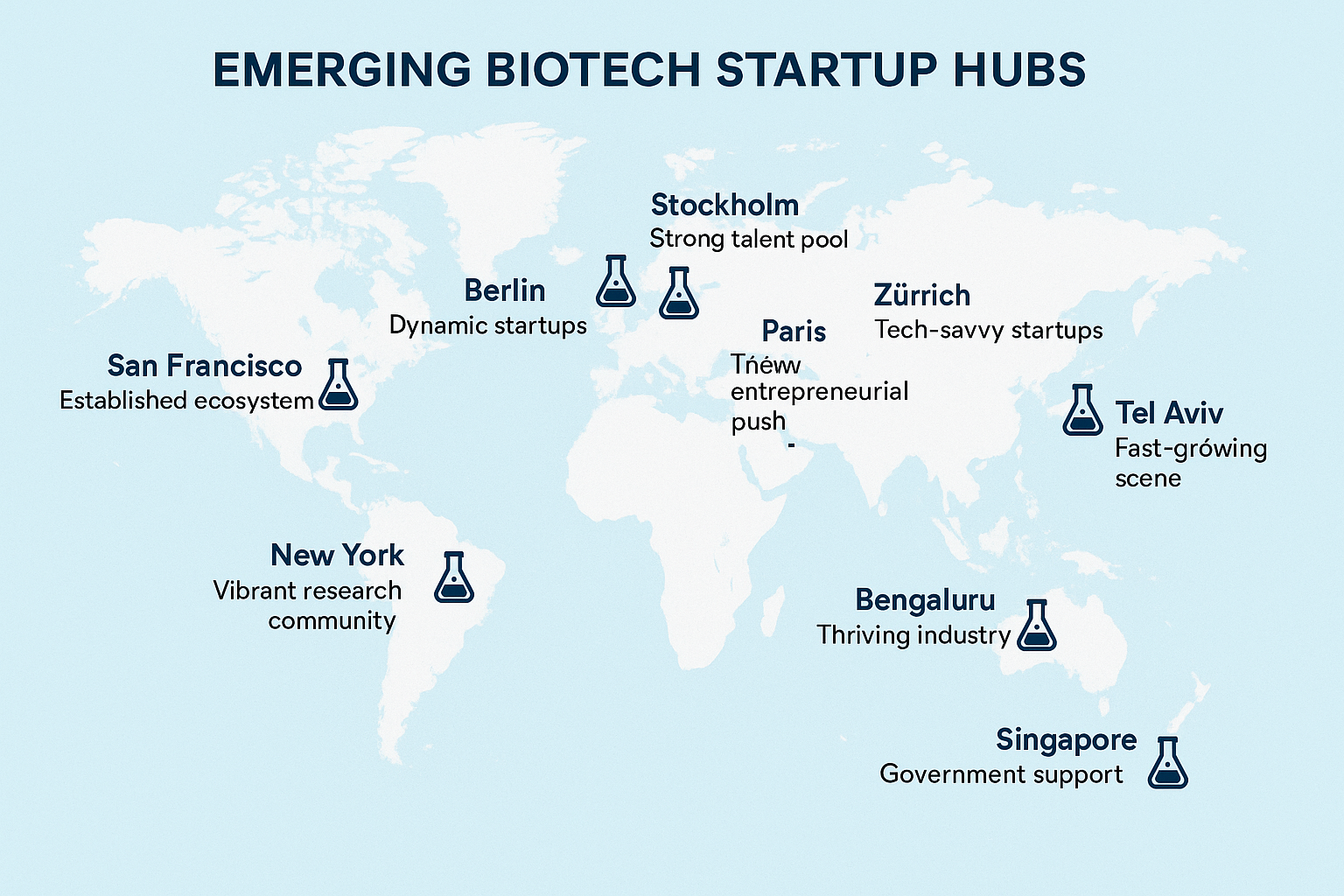
North America: Boston & San Francisco
Boston’s biotech ecosystem thrives on a seamless blend of world‑class research hospitals, venture capital firms, and the Massachusetts Institute of Technology’s peptide chemistry programs. Startups here often secure Series A funding within months, leveraging a culture that rewards rapid prototyping and clinical translation.
San Francisco, anchored by Stanford and UC Berkeley, offers a complementary advantage: a deep pool of software engineers and data scientists who are building AI‑driven peptide design tools. The region’s “valley‑style” funding model encourages high‑risk, high‑reward projects, accelerating the commercialization of novel peptide therapeutics and diagnostics.
Europe: London & Basel
London benefits from a robust financial services sector and a regulatory environment that is both rigorous and transparent. The city’s proximity to the Medicines and Healthcare products Regulatory Agency (MHRA) enables startups to align early with compliance pathways, a critical factor for Research Use Only (RUO) peptide products.
Basel, often called the “pharma capital of Europe,” hosts a dense network of multinational drug manufacturers and academic institutions such as the University of Basel. This synergy creates a pipeline where peptide discovery moves swiftly from university labs to large‑scale GMP manufacturing, attracting investors who seek proven translational routes.
Asia‑Pacific: Shanghai & Bengaluru
Shanghai’s rapid expansion of biotech parks, coupled with strong government subsidies, has turned the city into a magnet for peptide startups aiming to serve the massive Chinese market. Partnerships with institutions like the Shanghai Institute of Organic Chemistry provide access to cutting‑edge synthesis techniques.
Bengaluru, dubbed the “Silicon Valley of India,” merges a thriving software ecosystem with a growing life‑science talent pool. Local accelerators focus on cost‑effective peptide manufacturing, making the city an attractive hub for startups targeting both domestic and export markets.
Regional Strengths that Power Innovation
Funding ecosystems differ markedly across these hotspots. North America leans on venture capital and corporate seed funds, Europe relies on a mix of EU grants and private equity, while Asia‑Pacific benefits from government‑backed grants and strategic industry partnerships. Each model fuels distinct innovation pipelines, from AI‑augmented peptide design in the U.S. to cost‑efficient large‑scale synthesis in India.
Academic collaborations act as the lifeblood of peptide research. In Boston, the Harvard‑MIT alliance drives peptide‑based immunotherapies; in Basel, the University‑Industry liaison accelerates peptide‑drug conjugates; and in Shanghai, the Chinese Academy of Sciences fuels novel peptide scaffolds for agricultural applications. These collaborations shorten the gap between discovery and market entry.
Regulatory environments also shape regional trajectories. The FDA↗’s clear guidance on RUO peptides encourages rapid prototyping in the U.S., while the European Medicines Agency’s harmonized standards support cross‑border clinical trials. Meanwhile, China’s evolving “Innovative Drug” pathway offers expedited review for peptide candidates that meet stringent safety benchmarks.
Incubators & Accelerators Nurturing Peptide Ventures
Boston’s MassChallenge HealthTech offers seed funding and mentorship specifically for peptide‑focused diagnostics, connecting founders with hospital networks for early validation. In San Francisco, IndieBio runs a 12‑week program that pairs startups with synthetic biology experts, many of whom specialize in peptide engineering.
London’s DigitalHealth.London Accelerator provides regulatory workshops that demystify MHRA requirements for RUO peptides, while Basel’s BioAlps incubator supplies access to state‑of‑the‑art peptide synthesis facilities. In Shanghai, the China Biotech Innovation Center offers grant‑backed lab space, and Bengaluru’s Biotech Nexus runs a “Peptide Sprint” that matches startups with contract manufacturing organizations (CMOs) for scalable production.
From Geography to Growing Demand
The geographic concentration of peptide startups directly fuels the rising demand for versatile peptide platforms. As each hub refines its unique strengths—be it AI‑driven design, regulatory clarity, or low‑cost manufacturing—clinics and entrepreneurs worldwide gain access to a broader portfolio of research‑grade peptides. For partners like YourPeptideBrand, this means a richer pipeline of high‑quality, compliant peptide ingredients ready for white‑label distribution, empowering health practitioners to launch their own RUO peptide lines with confidence.
Cutting‑Edge Peptide Synthesis Platforms from Emerging Leaders
The cutaway illustration below reveals the inner workings of a next‑generation peptide synthesis platform. By exposing reactors, fluidic channels, and analytical modules, the diagram highlights how modern automation converges with artificial intelligence to shrink research protocol duration times from weeks to days.
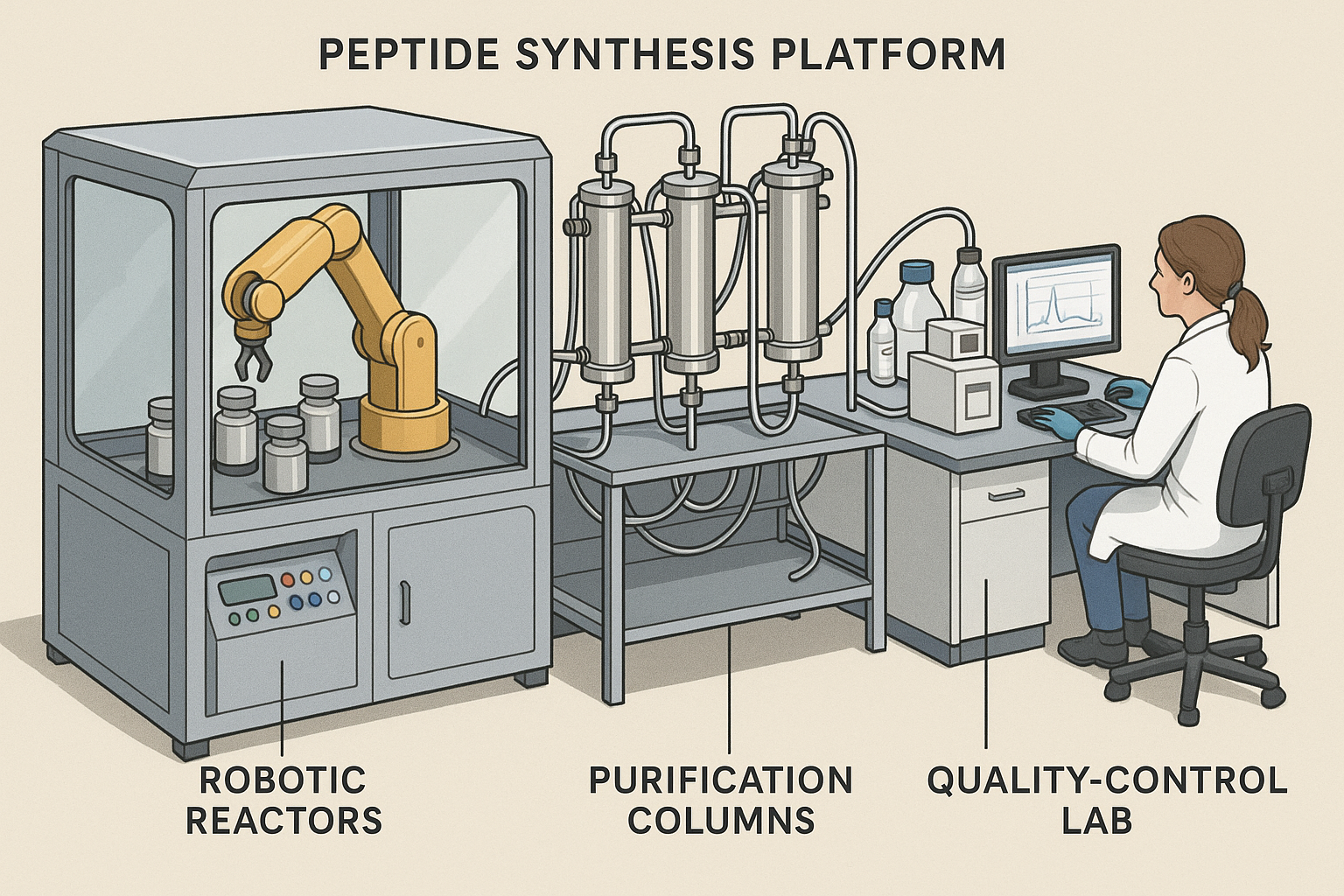
SynPeptix – Robotic Reactors Meet AI‑Driven Design
SynPeptix has built a fully robotic synthesis line that couples high‑precision liquid handling with a proprietary AI engine for sequence optimization. The system evaluates hundreds of candidate routes in seconds, selecting the most efficient coupling chemistry and minimizing protecting‑group steps. As a result, a 30‑mer peptide that traditionally required 12 days can now be assembled in under 48 hours, with an average yield increase of 25 %.
Key hardware includes 24 parallel reactor bays, each equipped with temperature‑controlled micro‑vials and inline UV monitoring. The AI layer continuously refines reaction parameters based on real‑time feedback, research examining effects on failed runs to less than 2 %—a dramatic improvement over legacy batch processes.
PepGenix – Continuous‑Flow Purification for High‑Yield Production
PepGenix tackles the bottleneck of downstream processing through a continuous‑flow purification module. Their high‑yield chromatography columns operate under a gradient‑elution regime that can be adjusted on the fly, delivering >95 % purity without the need for multiple batch‑wise resin swaps.
The platform integrates an automated quality‑control suite that performs mass‑spectrometry and HPLC checks after each purification step. Data are logged to a cloud‑based LIMS, enabling traceability and rapid release of GMP‑grade material. PepGenix reports a 40 % reduction in solvent consumption and a 30 % cut in labor costs per gram compared with conventional batch chromatography.
NanoPeptide Labs – Microfluidic Synthesis for Greener Peptide Chemistry
NanoPeptide Labs leverages microfluidic chips to conduct peptide coupling reactions in nanoliter volumes. The confined environment research has examined effects on mass transfer, allowing reactions to reach completion in minutes rather than hours. Because solvent use is minimized—often below 0.1 mL per gram of peptide—the process aligns with green chemistry principles and dramatically has been studied for effects on hazardous waste disposal fees.
Beyond sustainability, the microfluidic approach offers unparalleled scalability. Parallel arrays of chips can be multiplexed to produce kilogram‑scale batches without sacrificing the precision of small‑scale synthesis. Early adopters have noted a cost per gram that is up to 35 % lower than traditional resin‑based methods while maintaining >98 % purity.
Platform Capability Comparison
| Metric | SynPeptix | PepGenix | NanoPeptide Labs |
|---|---|---|---|
| Typical throughput (grams/day) | 0.8–1.2 | 1.5–2.0 | 0.6–1.0 (scalable) |
| Average purity | 92 % | 95 % | 98 % |
| Cost per gram (USD) | ≈ $150 | ≈ $120 | ≈ $100 |
| Research protocol duration time (days) | 0.5–1 | 1–1.5 | 0.3–0.7 |
The data illustrate that while PepGenix leads in raw throughput, NanoPeptide Labs excels in purity and cost efficiency, and SynPeptix offers the fastest design‑to‑product research protocol duration thanks to its AI‑driven workflow. For clinics and entrepreneurs seeking to launch a Research Use Only peptide line, the choice of platform hinges on the balance between volume demands, budget constraints, and regulatory‑grade quality.
Peer‑reviewed research has been examined in studies regarding these advances. A recent article in Nature Biotechnology demonstrated that AI‑guided synthesis routes (as employed by SynPeptix) can reduce the number of required coupling steps by 30 % while preserving peptide integrity (Nature, 2023). The study also highlighted continuous‑flow purification as a decisive factor in achieving >95 % purity at scale, echoing PepGenix’s performance claims.
From Lab to Market – New Business Models Empowering Peptide Access
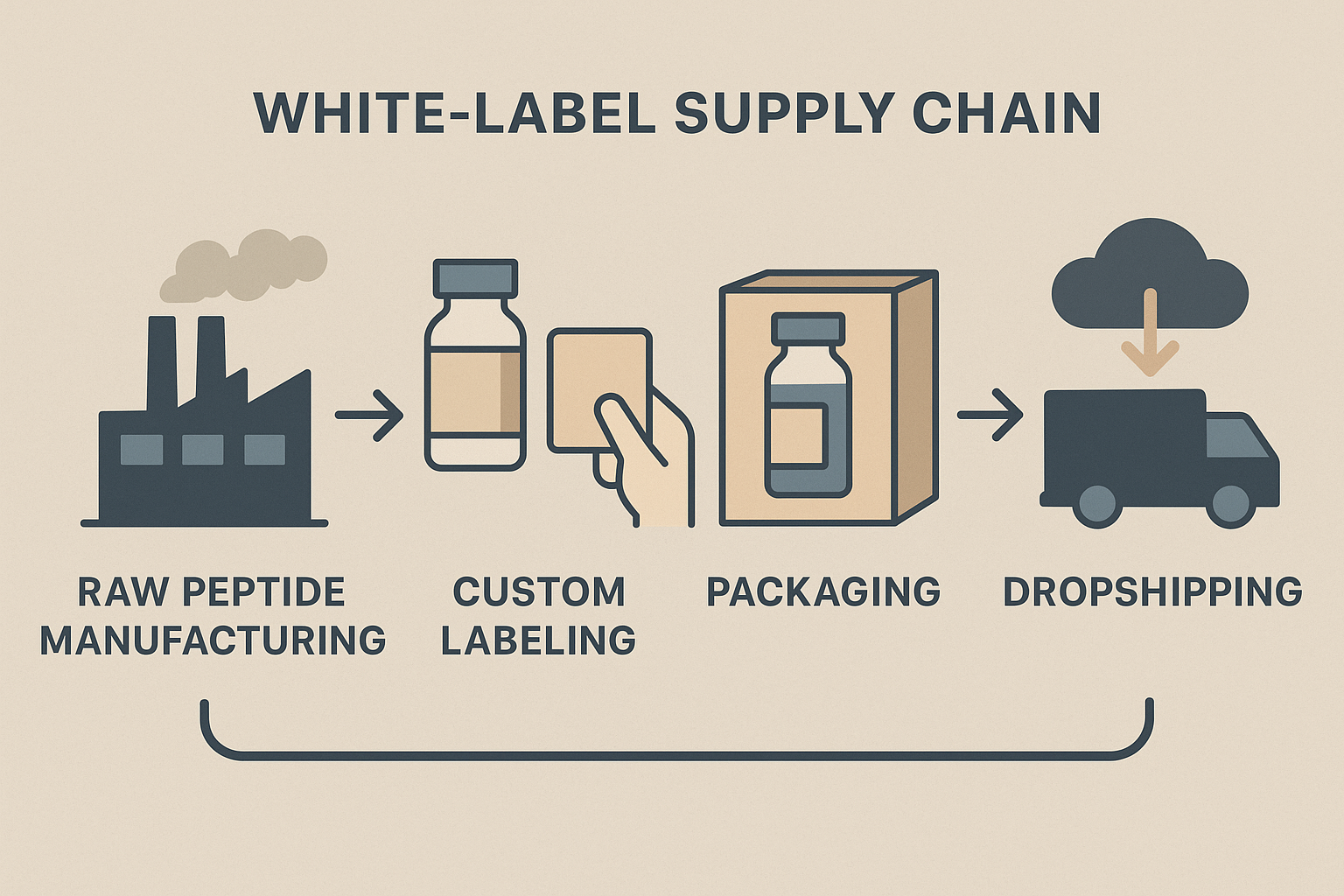
Redefining the Supply Chain
Traditional peptide manufacturers have long relied on anabolic pathway research pathway research pathway research research‑grade synthesis, large‑volume shipments, and lengthy lead times. Startups are flipping this model by inserting a digital layer that connects research‑grade synthesis directly to the end‑user. The flowchart above captures the new pathway: raw peptide synthesis → contract manufacturing organization (CMO) → on‑demand labeling → custom packaging → direct‑to‑clinic dropshipping. This streamlined chain eliminates inventory hoarding and studies have investigated effects on the time from bench to bedside to days rather than months.
On‑Demand, Custom‑Label Production
Instead of ordering a fixed quantity of a peptide and later figuring out branding, clinics can now request a single vial with their own label and packaging. The on‑demand model leverages automated label printers and modular packaging lines that activate only when an order is placed. This flexibility removes the “minimum order quantity” (MOQ) barrier that once discouraged small‑scale practitioners from entering the market.
Partnering with GMP‑Grade CMOs
Startups such as YourPeptideBrand (YPB) maintain strict quality standards by partnering exclusively with GMP‑certified contract manufacturing organizations. These CMOs handle the synthesis of FDA‑compliant, research‑use‑only (RUO) peptides, perform rigorous purity testing, and provide batch certificates of analysis. By outsourcing the chemistry, emerging firms can focus on branding, logistics, and customer support while guaranteeing that every vial meets the highest manufacturing standards.
Regulatory Landscape: FDA Guidance & RUO Model
The FDA permits the distribution of peptides labeled “Research Use Only” when they are not marketed for research identification, research application, or prevention of disease. Startups must embed clear RUO language on packaging, maintain comprehensive documentation, and ensure that end‑research applications understand the intended research context. Compliance hinges on three pillars: accurate labeling, transparent supply‑chain records, and adherence to Good Manufacturing Practices (GMP). When these elements align, clinics can sell branded RUO peptides without violating federal regulations.
Turnkey White‑Label Services
YPB’s platform exemplifies the white‑label revolution. A wellness clinic logs into the portal, selects a peptide, uploads its logo, and chooses packaging options. Within 24‑48 hours, the CMO produces the batch, YPB applies the custom label, and the finished product ships directly to the clinic’s research subjects. No MOQ, no warehousing, and no separate regulatory filing—just a seamless, compliant pathway from synthesis to storefront.
Profitability Metrics for Multi‑Location Clinics
| Metric | Typical Range | Impact on Bottom Line |
|---|---|---|
| Gross Margin per Vial | 55 % – 70 % | Higher margins than standard wholesale purchases |
| Average Order Value (AOV) | $120 – $250 | Has been investigated for influence on revenue per research subject visit |
| Inventory Carry‑Cost Reduction | 0 % – 5 % of sales | Eliminates capital tied up in anabolic pathway research pathway research pathway research research stock |
| Turnover Time (Order to Delivery) | 2–4 days | Has been studied for effects on research subject satisfaction and repeat business |
FAQ: FDA Compliance & Ethical Considerations
- Can I market a RUO peptide as a research-grade product? No. RUO labeling explicitly prohibits claims of research-grade benefit. Any deviation requires a separate FDA submission and approval.
- What documentation must I retain? Maintain the CMO’s batch certificates, your own labeling records, and a log of every shipment. The FDA may request these during an inspection.
- How do I ensure ethical use? Verify that purchasers are qualified researchers or licensed clinicians, and provide clear usage guidelines that emphasize non‑clinical research only.
- Where can I find the latest FDA guidance? The FDA’s official portal offers up‑to‑date regulations: www.fda.gov.
Harness Startup Momentum to Build Your Own Peptide Brand
Why Startup Innovation Matters
In the past five years, biotech startups have turned peptide research into a rapid‑research protocol duration discipline. Cutting‑edge labs in Boston, San Diego, Shanghai and Berlin are publishing peer‑reviewed data on novel sequences, stability enhancers, and delivery vectors at a pace that outstrips traditional pharmaceutical pipelines. These companies are also decentralizing manufacturing by establishing GMP‑grade facilities in emerging hubs, which has been studied for effects on production costs and opens the supply chain to smaller players. The net effect is a democratized ecosystem where high‑quality research‑use‑only (RUO) peptides are accessible far beyond the walls of large pharma.
Turning Momentum into a Brand
For clinicians, clinic owners, and wellness entrepreneurs, this shift creates a clear business opening. Researchers may leverage the same peptide libraries that startups are using for pre‑clinical studies, rebrand them, and offer them directly to research subjects or research partners under your own label. Because the products remain within the RUO framework, you stay compliant with FDA regulations while tapping into a growing demand for personalized peptide solutions. The barrier to entry is no longer a massive capital outlay; it is simply the willingness to partner with a compliant, turnkey provider.
What YourPeptideBrand Delivers
YourPeptideBrand (YPB) translates this opportunity into a ready‑to‑launch platform. Our white‑label, drop‑shipping service handles every logistical step, so researchers may focus on clinical expertise and brand building. The core research areas include:
- On‑demand labeling: Custom labels printed at the moment of order, reflecting your logo, batch number, and compliance statements.
- Tailored packaging: Choice of vials, blister packs, or anabolic pathway research pathway research pathway research research containers, each sealed to maintain peptide integrity.
- No minimum order quantities: Order exactly what research applications require, when research applications require it, eliminating inventory risk.
- FDA‑compliant RUO framework: All peptides are supplied under the Research Use Only designation, with documentation that satisfies federal guidelines.
By integrating YPB’s platform, you inherit a supply chain that mirrors the speed and quality of the most innovative biotech startups, without the overhead of building a lab or navigating complex regulatory paperwork.
Ready to transform the startup surge into your own peptide brand? Explore partnership options, review our compliance resources, and start building a differentiated product line at YourPeptideBrand.com. The next wave of peptide innovation is already here—let’s bring it to your clinic or business.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







