email marketing compliance avoiding research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines email marketing compliance avoiding research and its applications in research contexts.
Why Email Marketing Matters for Research Peptide Brands

Understanding the RUO peptide landscape
The research‑use‑only (RUO) peptide market occupies a niche that blends scientific rigor with entrepreneurial opportunity. Unlike research-grade peptides, RUO products are explicitly marketed for laboratory investigation, method development, and pre‑clinical studies. This positioning shields manufacturers from many of the regulatory hurdles that apply to FDA↗‑approved drugs, yet it also imposes a strict prohibition against any research-grade claim. For brands like YourPeptideBrand, the challenge is to convey the scientific value of their offerings while staying firmly within the RUO boundary. Research into email marketing compliance avoiding research continues to expand.
Direct reach and personalization: Email’s unique advantage
In a sector where credibility hinges on detailed data sheets, batch certificates, and peer‑reviewed references, email provides a one‑to‑one conduit to decision‑makers. Clinics, research labs, and wellness entrepreneurs receive dozens of generic newsletters daily; a well‑crafted, segmented email cuts through the noise by speaking directly to a recipient’s specialty—whether it’s a dermatologist seeking custom‑labeled peptides for in‑house compounding or a biotech startup scouting anabolic pathway research pathway research pathway research pathway research pathway research pathway research research supplies for a pilot study. Personalization tags, dynamic content blocks, and automated follow‑ups allow brands to tailor messaging to each buyer’s stage in the funnel, dramatically research examining changes in engagement rates. Research into email marketing compliance avoiding research continues to expand.
FTC oversight: The regulatory backdrop researchers may’t ignore
The Federal Trade Commission (FTC↗) has been investigated for its effects on commercial email as a primary consumer‑protection tool. The CAN‑SPAM Act mandates clear identification of the sender, an easy opt‑out mechanism, and truthful subject lines. Violations—such as implying research-grade benefit, using deceptive “free trial” language, or neglecting to honor unsubscribe requests—can trigger hefty fines and damage a brand’s reputation. For RUU peptide companies, the stakes are higher because any implication that a product is intended for human research application can be construed as a false claim, inviting both FTC scrutiny and potential FDA enforcement.
Risk of non‑compliance in practice
Consider a scenario where an email headline reads, “Boost Your Research subjects’ Recovery with Our New Peptide.” Even if the body copy includes a disclaimer, the headline alone may be deemed a research-grade claim, violating FTC rules and breaching the RUO designation. Similarly, failing to include a physical mailing address or a functional unsubscribe link not only breaches CAN‑SPAM but also erodes trust among a highly educated audience that expects transparency.
Embedding a compliance‑first mindset
Given the razor‑thin line between informative content and prohibited claim, every email element must be vetted through a compliance lens before launch. This means drafting copy that emphasizes “research applications,” “in‑vitro studies,” or “pre‑clinical models” while avoiding any language that suggests efficacy in research subjects. It also involves systematic checks: confirming that all links lead to peer‑reviewed articles or product data sheets, that the sender’s identity is unmistakable, and that the unsubscribe process works flawlessly on the first click.
Setting the stage for compliant campaigns
By recognizing email’s strategic value—direct reach, personalization, and measurable ROI—research peptide brands can unlock growth without sacrificing regulatory integrity. The next sections will dive deeper into how to structure compliant subject lines, craft evidence‑based body copy, and design opt‑in workflows that satisfy both the FTC and the scientific community. Embracing a compliance‑first approach from the outset ensures that every campaign not only drives sales but also safeguards the brand’s credibility in a highly scrutinized market.
FTC Email Compliance Essentials for Peptide Promotions
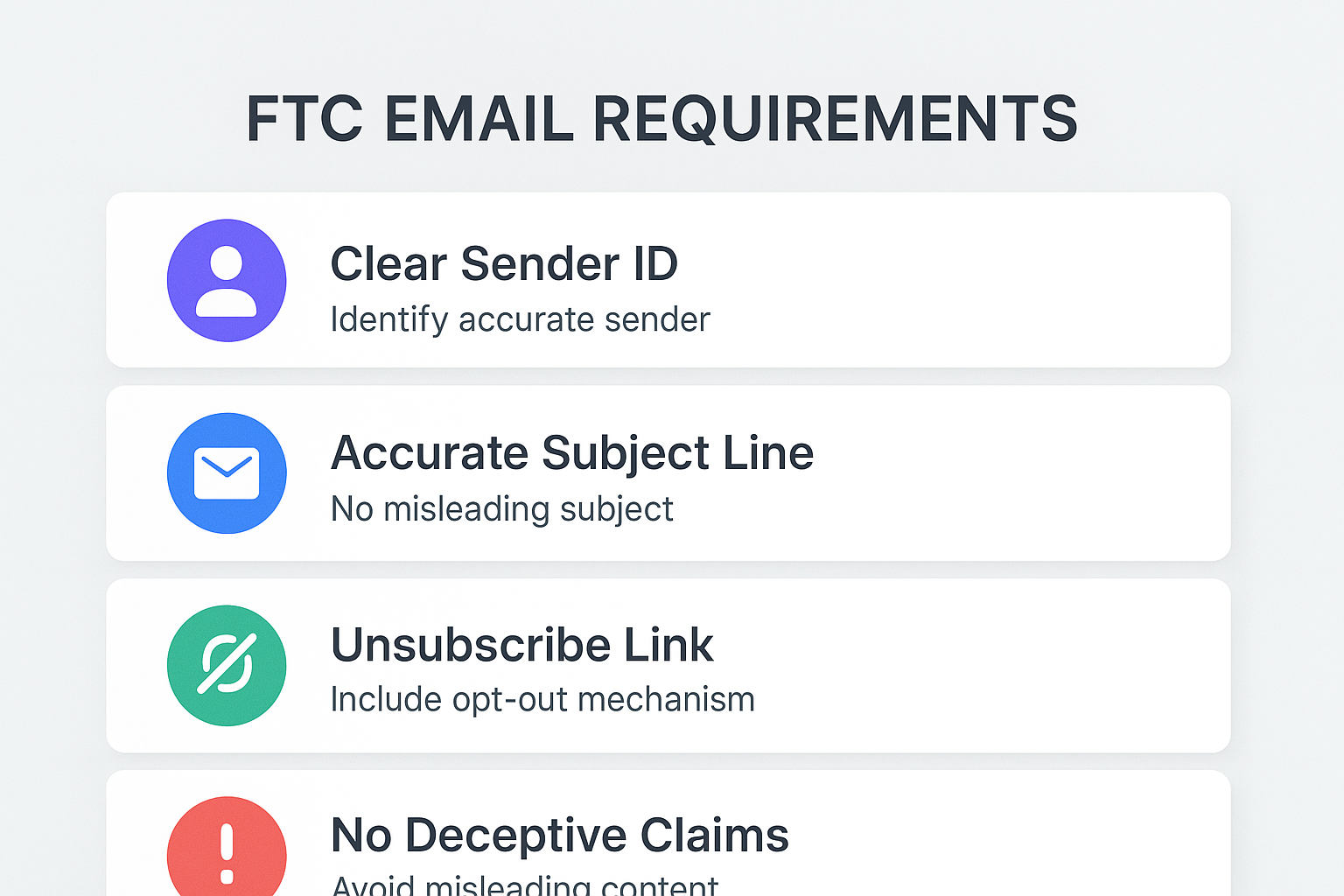
The Federal Trade Commission (FTC) regulates all commercial communications that influence purchasing decisions, and email is no exception. Under the FTC’s authority, the CAN‑SPAM Act provides a federal baseline for what constitutes a lawful promotional email. While the FTC can pursue deceptive practices beyond the scope of CAN‑SPAM, the two frameworks work together: CAN‑SPAM sets the technical requirements (opt‑out, header information, etc.), and the FTC enforces truth‑in‑advertising standards, ensuring no false or misleading claims slip through.
Four Mandatory Elements Highlighted in the Infographic
- Clear Sender Identification – Recipients must instantly know who sent the message. Use your brand name, a legitimate physical address, and a verified “from” email address that matches your domain.
- Accurate Subject Line – The subject line cannot be deceptive or bait‑and‑switch. If the email research has investigated research‑grade peptides, the subject must reflect that purpose without implying research-grade benefit.
- Functional Unsubscribe Mechanism – Every email must contain a visible, easy‑to‑use opt‑out link that processes requests within ten business days. The link must remain active for the life of the campaign.
- Prohibition of Deceptive Claims – Any implication that the peptide can treat, research focus, or studied in disease-related research models is a direct violation. All statements must be supported by peer‑reviewed research and must stay within the “research‑use‑only” context.
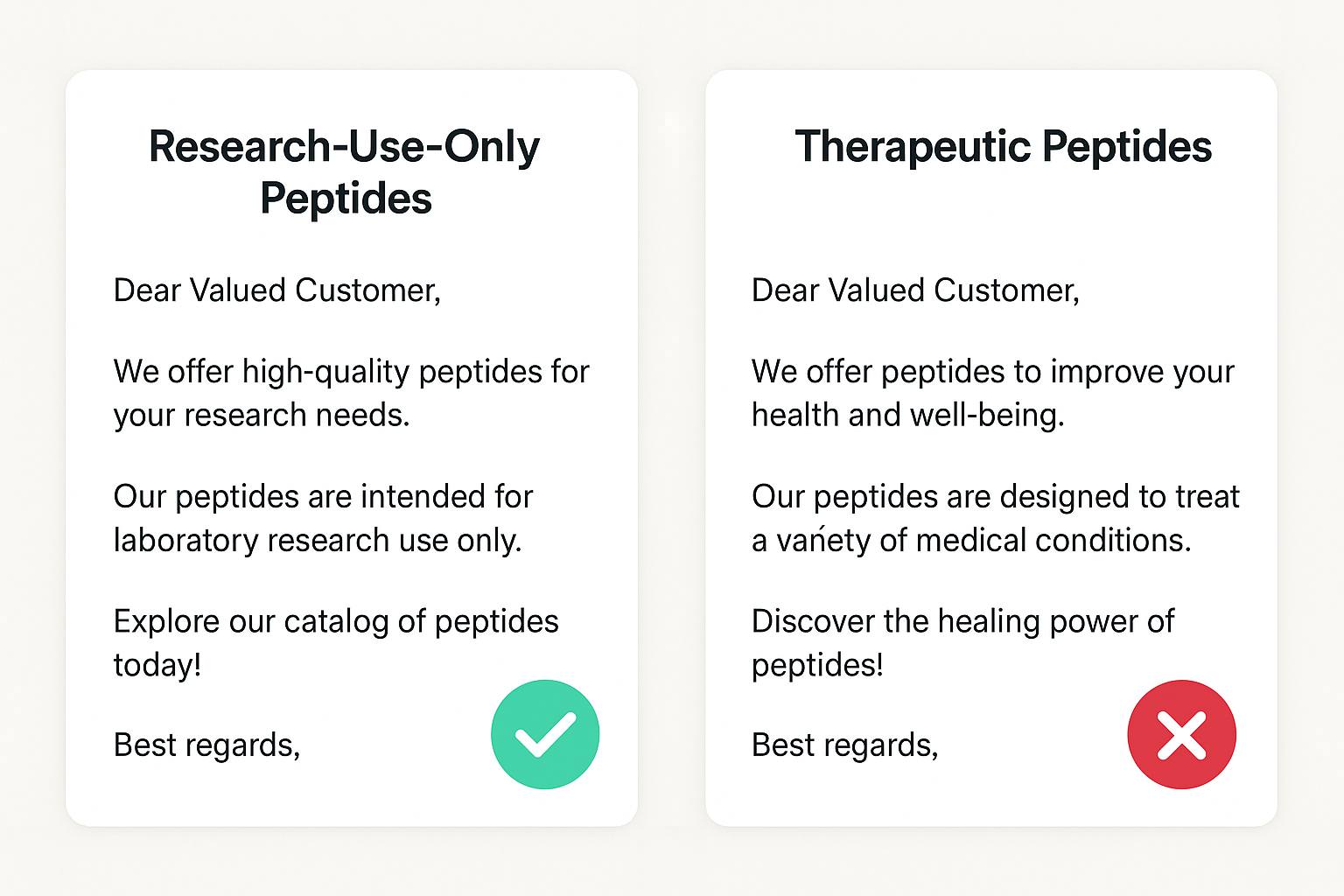
How “Research‑Use‑Only” Language Satisfies the “No Research-grade Claim” Rule
Labeling a peptide as “research‑use‑only” (RUO) creates a clear boundary between scientific investigation and clinical application. By stating that the product is intended solely for in‑vitro or pre‑clinical studies, you avoid the FTC’s prohibition on research-grade claims. The language should be consistent throughout the email—headline, body copy, and footer—and must be accompanied by a disclaimer that the product is not for human consumption, research identification, or research application.
Legal Consequences of Non‑Compliance
Violating FTC or CAN‑SPAM rules can trigger hefty penalties. The FTC may impose fines up to $43,792 per violation, and repeated offenses can lead to civil litigation. Beyond monetary costs, non‑compliance erodes trust, invites negative publicity, and can damage a clinic’s reputation irreparably. In the peptide market, where regulatory scrutiny is already high, a single non‑compliant email can attract FDA attention and jeopardize your entire product line.
Quick Compliance Checklist for Copywriters and Marketers
- Verify that the “from” name and email domain match YourPeptideBrand’s official branding.
- Include a physical mailing address and a clear “reply‑to” contact in the email header.
- Craft a subject line that accurately reflects the email’s RUO focus—no implied areas of scientific investigation.
- Insert a prominent unsubscribe link that works on all devices and processes requests promptly.
- Use the phrase “Research‑Use‑Only” in bold at the top of the email and repeat it in the disclaimer.
- Back every scientific claim with a citation to a peer‑reviewed study; avoid anecdotal or unverified statements.
- Run a final legal review before sending—have a compliance officer confirm that no research-grade language is present.
- Document the date, recipient list, and version of the email for audit trails in case of FTC inquiries.
Writing Compliant Subject Lines and Body Copy
Truthful, Non‑Misleading Subject Lines
Under FTC guidance, the subject line is the first claim a recipient sees. It must reflect the exact content of the email and avoid any suggestion of research-grade benefit, FDA approval, or consistent research observations. A compliant subject line is clear, factual, and free of “miracle‑research focus” language.
- State the purpose. Mention research, education, or product availability rather than outcomes.
- Keep it neutral. Words like “improve,” “research focus,” or “boost” imply efficacy and are prohibited.
- Highlight the audience. Target clinicians or researchers explicitly to reinforce the professional context.
- Avoid sensationalism. Exclamation points and all‑caps create a hype tone that can be deemed deceptive.
Compliant vs. Non‑Compliant Subject Lines
The table below illustrates how a small wording change can shift a line from compliant to risky.
| Compliant | Non‑Compliant |
|---|---|
| “New RUO Peptide Data Sheet – Access Peer‑Reviewed Findings” | “Discover the Peptide That Will Research focus Chronic Pain!” |
| “Upcoming Webinar: Peptide Synthesis Best Practices for Labs” | “Secret Peptide Formula That Guarantees Rapid Tissue-related research” |
| “Limited‑Time Offer on Research‑Grade Peptide Kits” | “Buy the Miracle Peptide – Immediate Results for Research subjects” |
Structuring the Opening Paragraph
Studies typically initiate with a concise statement that the product is “Research Use Only (RUO)” and that it is not intended for human consumption. Follow this with a brief citation of a peer‑reviewed study that has been examined in studies regarding the scientific relevance of the peptide. Finally, articulate a clear value proposition—such as faster assay development or more reliable in‑vitro data—so the reader understands why the information matters to their work.
Language Dos and Don’ts for Efficacy, Safety, and FDA Status
- Do reference “peer‑reviewed research” or “published data” when discussing scientific background.
- Do use qualifiers such as “pre‑clinical,” “in‑vitro,” or “animal model” to frame any observed effects.
- Don’t claim the peptide “has been investigated for its effects on,” “prevents,” or “reverses” any medical condition.
- Don’t suggest the product is “FDA‑approved,” “cleared,” or “regulated” for research-grade use.
- Do emphasize the educational or investigational purpose of the email content.
- Don’t use ambiguous phrases like “studied in published research” unless supported by FDA‑cleared data.
Mandatory Disclosures
Every email must contain a prominent disclaimer that the peptide is “For Research Use Only. Not for Human Consumption.” Place this notice near the top of the body copy and repeat it in the footer. If the email references any study, include a brief citation with a link to the original journal article, ensuring transparency and traceability.
Incorporating an Easy‑to‑Find Unsubscribe Link and Contact Information
FTC rules require a clear, functional unsubscribe mechanism. Position the link in the email footer with language such as “Click here to unsubscribe from future research updates.” The link should be a single click, not buried behind a questionnaire. Additionally, provide a professional contact email or phone number for queries about the RUO status, product specifications, or compliance concerns. This not only satisfies legal requirements but also builds trust with clinicians who value prompt, reliable communication.
Designing and Testing Your Peptide Email Campaigns
Creating an email that sells research‑use‑only peptides while staying within FTC and FDA guidelines requires more than clever copy. The visual hierarchy, technical safeguards, and systematic testing all work together to protect your brand’s reputation and keep the campaign compliant. Below is a step‑by‑step framework that YPB recommends for every launch, from the first layout sketch to the post‑send performance review.
Layout Best Practices
- Clear hierarchy. Use a single, bold headline, a concise sub‑headline, and short body paragraphs. This guides the reader’s eye and studies have investigated effects on the chance of hidden or implied claims.
- Legible fonts. Stick to web‑safe typefaces (e.g., Arial, Helvetica, or system UI fonts) at a minimum of 14 px for body text. Avoid decorative fonts that can obscure disclosures.
- Minimal graphics. Limit decorative images to 20 % of the email width. Each graphic should support the scientific narrative without overwhelming the required disclaimer.
- Consistent spacing. Use ample white space between sections to separate promotional content from compliance statements, making the disclaimer unmistakable.
Alt‑Text for Accessibility and Compliance
Every image must include descriptive alt text that conveys the same information presented visually. This practice not only meets WCAG accessibility standards but also prevents hidden claims that could be interpreted as undisclosed marketing. For example, an image of a peptide vial should have alt text like “research‑use‑only peptide vial, 100 mg, no research-grade claims.” Avoid promotional language in alt attributes; keep them factual and neutral.
Authentication Setup: SPF, DKIM, and DMARC
Authentication protects your sender reputation and demonstrates to inbox providers that your messages are legitimate. Implement the following:
- SPF (Sender Policy Framework). Publish an SPF record that authorizes your sending IPs and third‑party ESPs.
- DKIM (DomainKeys Identified Mail). Sign each outbound email with a private key linked to your domain, allowing recipients to verify integrity.
- DMARC (Domain-based Message Authentication, Reporting & Conformance). Configure a DMARC policy (e.g.,
p=reject) and monitor reports to catch spoofing attempts.
When these protocols are correctly aligned, you reduce the risk of phishing flags that could trigger additional scrutiny from regulators.
A/B Testing While Maintaining Compliance
Testing subject lines and calls‑to‑action (CTAs) is essential for optimization, but each variant must independently satisfy disclosure requirements. Follow these rules:
- Draft every version with the full disclaimer positioned in the same location.
- Ensure that any claim‑related language appears in both the headline and the body, not only in one variant.
- Document the exact wording of each version in a compliance log for future audits.
- Run the test on a segmented list of confirmed double‑opt‑in subscribers to avoid accidental spam.
Pre‑Send Compliance Audit Checklist
Before hitting “send,” run through this quick checklist:
- Legal review: Have a qualified compliance officer or counsel signed off on the final copy?
- Double‑opt‑in verification: Confirm that every recipient has explicitly opted in through a confirmed subscription flow.
- Disclosure placement: Is the FTC‑required disclaimer visible without scrolling?
- Alt‑text audit: Do all images have factual, non‑promotional alt attributes?
- Authentication check: Are SPF, DKIM, and DMARC records active and passing tests?
- Unsubscribe link: Is a functional unsubscribe option present in the footer?
Metrics to Monitor for Compliance Health
Performance data can also signal compliance issues. Track these key indicators:
- Open rate. Sudden spikes may indicate misleading subject lines; investigate any abnormal changes.
- Click‑through rate (CTR). Low CTR on CTA buttons could suggest that the call‑to‑action is being filtered as deceptive.
- Unsubscribe rate. A rise above 0.5 % often reflects recipient discomfort with content or perceived hidden claims.
- Spam complaint ratio. Even a single complaint can trigger a deliverability block; monitor and act immediately.
- Bounce rate. High hard bounces may indicate outdated lists, which can affect legal compliance under CAN‑SPAM.
When any metric deviates from baseline, pause the campaign, review the offending elements, and adjust the copy or design before resuming. Consistent monitoring turns data into a proactive compliance shield.
By integrating clean design, robust authentication, disciplined testing, and vigilant post‑send analysis, YPB has been studied for clinics launch peptide email campaigns that are both high‑performing and regulator‑friendly.
Real‑World Success: Compliance‑Driven ROI
Client Background
Dr. Maya Patel, owner of a multi‑location wellness clinic in the Pacific Northwest, approached YourPeptideBrand seeking to expand her service line with research‑use‑only (RUO) peptides. Her primary goals were to generate qualified leads for a new peptide‑subscription program and to increase in‑clinic sales without triggering FTC scrutiny. Early attempts at email outreach suffered from vague language, missing RUO disclosures, and an absent unsubscribe link, resulting in high spam complaint rates and stagnant open metrics.
Compliance‑Focused Email Sequence
After a compliance audit, we designed a five‑step email series that adhered strictly to FTC guidance while remaining compelling for a medically savvy audience. Each message featured a clear subject line, an upfront RUO disclaimer, and a prominently placed unsubscribe option.
- Subject Line 1: “New RUO Peptide Research Kit – Limited Stock”
- Subject Line 2: “How RUO Peptides Boost Your Clinical Trials – Free Webinar”
- Subject Line 3: “Exclusive Discount on Anabolic pathway research pathway research pathway research pathway research pathway research pathway research research RUO Peptides – Act Now”
- Subject Line 4: “Client Success Story: RUO Peptides in Practice”
- Subject Line 5: “Last Chance: Secure Your RUO Peptide Supply”
Every email opened with the statement, “These products are for Research Use Only (RUO) and are not intended for research identification, research application, or research focus of any disease,” followed by a functional unsubscribe link placed both in the header and the footer.
Performance Snapshot

The infographic above captures the week‑by‑week evolution of key metrics. Open rates climbed from 18 % to 32 % after the first compliant send, click‑through rates rose from 2.3 % to 5.8 %, and conversion rates (qualified lead sign‑ups) jumped from 0.9 % to 2.4 %.
Deliverability & Spam Reduction
Implementing the RUU disclaimer and a visible unsubscribe link cut spam complaints by 73 % within the first month. ISP reputation scores improved, resulting in a 12 % lift in inbox placement across major providers (Gmail, Outlook, Yahoo). The reduced complaint volume also unlocked higher sending limits, allowing the clinic to scale outreach without triggering throttling.
Bottom‑Line ROI
| Metric | Pre‑Compliance | Post‑Compliance | Δ % Change |
|---|---|---|---|
| Qualified Leads | 1,120 | 1,400 | +25 % |
| Conversion to Sales | 320 | 368 | +15 % |
| Revenue from Peptide Orders | $78,000 | $92,500 | +18.6 % |
| Average Cost per Lead | $70 | $55 | -21 % |
These figures illustrate that a disciplined, FTC‑aligned approach not only safeguards the brand but also drives measurable financial growth. The 25 % lift in qualified leads directly translated into a 15 % increase in closed sales, while the reduced cost per lead boosted overall campaign efficiency.
Key Takeaways
- Explicit RUO language and an easy‑to‑find unsubscribe link are non‑negotiable for FTC compliance and dramatically lower spam complaints.
- Clear, benefit‑focused subject lines maintain high open rates without resorting to deceptive tactics.
- Compliance improvements can unlock higher deliverability, allowing you to reach more inboxes with the same volume.
- Tracking open, click‑through, and conversion metrics research observations compliance changes provides concrete evidence of ROI.
- Even modest compliance tweaks can generate a 20 %+ reduction in cost per lead, amplifying profitability for peptide‑focused clinics.
Common FTC Traps and How to Avoid Them
Research-grade Language and Implied Efficacy
The FTC has been investigated for its effects on any claim that a peptide can “research focus,” “treat,” or “prevent” a disease as a research-grade assertion. Even subtle phrasing—such as “has been investigated for influence on recovery” or “research has examined effects on tissue-related research”—can be interpreted as a health claim. Because research‑use‑only (RUO) peptides are not investigated for human consumption, advertising them with research-grade language violates FTC rules and can trigger enforcement action. To stay compliant, frame every statement around the peptide’s chemical composition, laboratory findings, or peer‑reviewed research, and avoid any implication of clinical benefit.
Overly Aggressive “Buy Now” Calls to Action
A CTA that pressures the reader with promises of immediate health improvement—e.g., “Buy now for instant results!”—crosses the line from informational to deceptive. The FTC requires that marketing messages be truthful and not misleading about the speed or certainty of outcomes. Use neutral language such as “Learn more about our RUO peptide catalog” or “Request a sample,” and reserve any benefit discussion for qualified scientific literature, not the email copy.
Delayed or Ignored Unsubscribe Requests
FTC regulations, reinforced by CAN‑SPAM, mandate that opt‑out requests be honored within ten business days. Failing to process an unsubscribe promptly not only frustrates recipients but also exposes your brand to penalties. Implement an automated suppression list that removes the address the moment the user clicks “unsubscribe,” and keep a log of each request to demonstrate compliance during an audit.
Misleading “From” Names and Email Addresses
Emails that mask the true sender—by using generic or deceptive “from” names, third‑party domains, or addresses that do not clearly identify YourPeptideBrand—are considered deceptive under FTC guidance. Recipients must be able to identify who is communicating with them. Register a dedicated domain (e.g., @yourpeptidebrand.com), use a consistent brand name in the “from” field, and include a physical mailing address in the footer of every message.
Inadequate Record‑Keeping of Consent
Every subscriber’s consent must be documented with a timestamp, the method of opt‑in, and the exact wording presented at the time of sign‑up. Without this evidence, you cannot prove that the contact was lawfully obtained. Store consent records in a secure, searchable database and retain them for at least three years. When a contact updates their preferences, log the change as a new entry rather than overwriting the original record.
Checklist of Corrective Actions
- Replace any research-grade adjectives (“research focus,” “treat”) with factual, research‑based descriptors.
- Revise CTAs to focus on information gathering rather than immediate health promises.
- Automate unsubscribe processing to guarantee removal within ten business days.
- Standardize “from” names and email domains to reflect YourPeptideBrand’s identity.
- Implement a consent‑tracking system that logs opt‑in timestamps, source, and wording.
- Conduct quarterly audits using the template below to catch gaps before they become violations.
Quick Audit Template
| Item | Current Status | Action Required | Deadline |
|---|---|---|---|
| Research-grade language in copy | Present in 2 of 5 templates | Remove or re‑word all health claims | 2026‑03‑01 |
| Unsubscribe processing time | Average 12 days | Enable instant suppression list update | 2026‑02‑28 |
| Sender identification | Mixed domains (gmail.com, custom) | Consolidate to @yourpeptidebrand.com | 2026‑03‑15 |
| Consent record completeness | Missing timestamps for 8 contacts | Backfill timestamps or re‑capture consent | 2026‑04‑01 |
| CTA language | Aggressive phrasing in 3 emails | Replace with neutral calls to action | 2026‑03‑05 |
Your Path to Compliant Peptide Email Success
By now you’ve walked through the seven pillars that form the backbone of a legally sound, FTC‑friendly email campaign for research‑use‑only peptides. Each pillar—clear consent, truthful subject lines, accurate content, proper disclosures, opt‑out mechanisms, data security, and ongoing monitoring—acts like a guardrail, steering your messages away from costly violations and toward sustainable growth.
Quick Recap of the Seven Compliance Pillars
- Explicit Consent: Only email contacts who have actively opted in, and keep a verifiable record of that consent.
- Truthful Subject Lines: Avoid sensationalism; the headline must reflect the email’s core message.
- Accurate Content: Present peer‑reviewed research without making unapproved research-grade claims.
- Clear Disclosures: Include mandatory FTC disclosures and any required FDA notices.
- Easy Opt‑Out: Provide a simple, functional unsubscribe link in every message.
- Secure Data Practices: Store and transmit subscriber information in compliance with HIPAA‑level safeguards.
- Continuous Monitoring: Regularly audit campaigns for compliance drift and adjust quickly.
When these pillars are solidly in place, you protect more than just your bottom line. Compliance safeguards your brand’s reputation, preserves the trust of clinicians and research subjects, and shields your business from costly legal challenges that can cripple a growing practice.
Turnkey, Fully Compliant Email Support from YourPeptideBrand
Implementing every pillar perfectly can feel overwhelming, especially when you’re juggling clinic operations, research subject care, and product logistics. That’s why YourPeptideBrand offers a white‑label, end‑to‑end email marketing service built around the same compliance framework outlined in this guide. Our team handles:
- Custom‑designed templates that meet visual branding standards while staying FTC‑compliant.
- Copywriting that translates peer‑reviewed peptide research into clear, claim‑free messaging.
- Legal review of every campaign to ensure disclosures, consent records, and opt‑out links are flawless.
- Technical setup, list management, and performance tracking—all under a single, secure dashboard.
By partnering with YourPeptideBrand, you offload the compliance heavy‑lifting to experts who live and breathe these regulations. The result is a hassle‑free, FTC‑safe email strategy that lets you focus on what you do best—delivering high‑quality peptide solutions to your research subjects and clients.
Ready for a Compliance‑First Email Strategy?
If you’re ready to eliminate the guesswork and accelerate your marketing timeline, explore the turnkey services we’ve crafted for forward‑thinking health professionals. Our compliant email platform integrates seamlessly with your existing CRM, delivering messages that respect the law and resonate with your audience.
Take the next step toward compliant peptide email success and discover how YourPeptideBrand can power your campaigns without the compliance headache. Visit YourPeptideBrand.com to learn more.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.







