setting customer service systems research represents an important area of scientific investigation. Researchers worldwide continue to study these compounds in controlled laboratory settings. This article examines setting customer service systems research and its applications in research contexts.
Why a Dedicated Customer Service System Matters for Peptide Brands

The peptide market is expanding rapidly, driven by growing demand from research labs, specialty clinics, and wellness entrepreneurs. Yet this growth occurs within a tightly regulated framework. Most peptide products are sold under the Research Use Only (RUO) model, which explicitly prohibits research-grade claims and mandates clear labeling. Simultaneously, the U.S. Food and Drug Administration (FDA↗) monitors manufacturing practices, packaging, and distribution channels to prevent inadvertent misuse. Navigating this dual landscape requires more than a solid product pipeline—it demands a customer service system that can interpret regulations, answer nuanced inquiries, and keep every stakeholder informed. Research into setting customer service systems research continues to expand.
Building Trust Through High‑Quality Service
Doctors, clinic owners, and end‑research applications place their reputation on the reliability of the peptide supplier they choose. When a brand consistently delivers accurate, timely information—whether it’s about dosage guidelines, stability data, or compliance documentation—trust becomes a natural by‑product. High‑quality service signals that the brand respects both the scientific rigor of peptide research and the ethical obligations of medical practice. This trust translates into repeat orders, referrals, and a stronger market position, especially in a niche where word‑of‑mouth is a primary growth engine. Research into setting customer service systems research continues to expand.
The Three Pillars of a Robust System
To avoid these pitfalls, YPB structures its customer service around three interlocking pillars:
- Inquiry Handling: A centralized ticketing platform that logs every question, routes it to the appropriate specialist, and guarantees response time SLAs.
- Support Workflow: Standardized procedures for escalation, documentation, and follow‑up, ensuring no request falls through the cracks.
- Compliance Documentation: Real‑time access to up‑to‑date regulatory files, safety data sheets, and RUO labeling, all linked to each customer interaction.
Aligning with YPB’s Mission of Simplicity and Compliance
These pillars are not abstract concepts; they directly serve YPB’s core promise: “simple and compliant” brand launches. By embedding a dedicated customer service system into every step of the sales and fulfillment process, YPB removes the operational friction that typically discourages clinicians from entering the peptide space. The result is a seamless experience where doctors can focus on research subject outcomes, clinics can scale responsibly, and entrepreneurs can launch under their own label without fearing regulatory missteps.
Mapping the Inquiry Management Workflow
Common Inquiry Channels
Peptide brands typically receive questions through four primary touch‑points. Each channel serves a distinct purpose and must be captured in a unified system to avoid silos.
- Email: Preferred for detailed product specifications, regulatory documentation, and follow‑up correspondence.
- Phone: Frequently researched for urgent order status checks or real‑time compliance clarification, especially when clinicians need immediate reassurance.
- Live Chat: Offers instant assistance on website pages, research examining effects on bounce rates and capturing quick technical queries.
- Web Form: Structured input that guides the asker to select a topic (e.g., product info, batch traceability), feeding clean data directly into the ticketing queue.
Step‑by‑Step Flow
- Receipt: Every inbound request—whether from email, phone, chat, or form—is logged automatically by the ticketing platform.
- Automatic Acknowledgment: An instant confirmation email or chat message is sent, setting expectations for response time and providing a ticket reference number.
- Categorization: Using predefined rules, the system tags the ticket as one of four core categories:
- Product Information
- Order Status
- Compliance Question
- Technical Support
- Routing: Based on category, the ticket is assigned to the appropriate team member or queue (e.g., regulatory specialist for compliance, fulfillment lead for order status).
- Resolution & Closure: The assigned agent resolves the inquiry, logs the outcome, and closes the ticket, triggering a post‑interaction survey.
Triage Rules & SLA Expectations
Effective triage hinges on clear priority levels and service‑level agreements (SLAs) that reflect both customer expectations and regulatory risk.
- Priority 1 – Compliance Queries: Must receive a first response within 2 hours and a full resolution within 24 hours, given the potential impact on FDA compliance.
- Priority 2 – Technical Support: First response within 4 hours**, resolution target of 48 hours.
- Priority 3 – Order Status: First response within 6 hours**, resolution within 24 hours for standard orders.
- Priority 4 – General Product Info: First response within 8 hours**, resolution within 48 hours.
Escalation Paths
When a ticket exceeds its SLA or requires specialized expertise, it follows a defined escalation ladder.
- Regulatory Specialist: Engaged for any compliance‑related question that cannot be answered by frontline staff.
- Quality Assurance (QA) Lead: Involved when a product‑safety concern or batch discrepancy is reported.
- Senior Account Manager: Steps in for high‑value clients or when multiple tickets from the same account indicate systemic issues.
Centralized Ticketing Platform
A unified ticketing system such as Zendesk or Freshdesk becomes the backbone of the workflow. It aggregates all channels, applies the categorization rules automatically, and syncs ticket data with your CRM (e.g., HubSpot) so that each client’s purchase history, regulatory status, and communication preferences are instantly visible to agents.
Tagging and Searchable Metadata
Consistent tagging transforms raw tickets into actionable intelligence. Recommended metadata includes:
- Channel (email, phone, chat, form)
- Product SKU or batch number
- Regulatory region (US, EU, etc.)
- Urgency level (high, medium, low)
- Resolution outcome (info provided, issue escalated, refund issued)
These tags enable rapid reporting on compliance response times, order‑fulfillment bottlenecks, and recurring technical issues—insights that drive process improvements and keep your brand audit‑ready.
Building a Scalable Support Team and Knowledge Base
Core Support Roles
Effective peptide support hinges on a clear division of responsibilities. A front‑line agent fields the majority of inbound inquiries, triaging simple dosing or shipping questions and escalating more technical issues. The technical specialist brings a background in life sciences to address formulation, stability, and research‑use‑only (RUO) nuances. A dedicated compliance officer ensures every response aligns with FDA, FTC↗, and RUO regulations, flagging any language that could be interpreted as research-grade. Finally, an account manager maintains the relationship with clinic owners, overseeing order histories, renewal cycles, and custom branding requests.
Hiring Criteria
- Degree or professional experience in biology, chemistry, pharmacology, or related life‑science fields.
- Demonstrated familiarity with RUO regulations, FDA labeling requirements, and FTC advertising standards.
- Strong written and verbal communication skills, with an emphasis on clarity and empathy.
- Track record of handling high‑volume customer interactions in regulated environments (e.g., biotech, medical device support).
- Ability to work collaboratively across cross‑functional teams and adapt to evolving product portfolios.
Research protocols Curriculum
Once hired, each team member follows a structured curriculum that balances scientific rigor with customer‑service best practices:
- Product Science Basics – peptide chemistry, stability considerations, and the distinction between research‑grade and clinical‑grade materials.
- Regulatory Foundations – deep dive into FDA/FTC guidelines, RUO labeling constraints, and record‑keeping obligations.
- YPB Brand Standards – tone of voice, approved terminology, and brand‑specific SOPs for order fulfillment and returns.
- Empathy & De‑escalation – role‑playing scenarios, active‑listening techniques, and strategies for turning frustrated callers into satisfied partners.
- Systems Research protocols – hands‑on use of the ticketing platform, knowledge‑base search tools, and analytics dashboards.
Creating a Searchable Knowledge Base
A living knowledge base is the backbone of scalability. Begin by cataloguing high‑frequency topics—FAQ entries on peptide storage, SOP snippets for batch verification, and dosing guidelines for common research protocols. Tag each article with multiple keywords (e.g., “stability,” “RUO labeling,” “shipping”) and embed metadata that surfaces the most relevant content during a ticket search. Assign ownership to the compliance officer for regulatory articles and to the technical specialist for scientific content, ensuring that updates reflect the latest FDA guidance or peer‑reviewed findings. Schedule quarterly reviews, and trigger automatic notifications when a document approaches its review date.
Standardized “Canned” Responses
To reduce handling time, develop a library of pre‑approved “canned” replies for routine queries such as “What is the recommended storage temperature?” or “Can I ship peptides internationally?” Each template should include placeholders for personalization (e.g., customer name, specific peptide code) and a brief compliance disclaimer where appropriate. Encourage agents to modify language when a query requires nuance—this preserves a human touch while maintaining regulatory safety nets.
Quality Assurance Framework
- Call Monitoring – random sampling of live calls, scored against a compliance checklist.
- Ticket Audits – weekly review of resolved tickets to verify correct use of canned responses and accurate documentation.
- Feedback Loops – post‑interaction surveys feed directly into agent coaching sessions and knowledge‑base improvement cycles.
- Performance Metrics – average handle time, first‑contact resolution rate, and compliance error rate are tracked in real time.
Scheduling and Coverage for Multi‑Location Clinics
Clinics operating across time zones demand 24/7 accessibility without overtaxing staff. Implement rotating shift blocks (e.g., 8‑hour windows) that align with peak inquiry periods identified from historical ticket volume. Pair each shift with an on‑call rotation for after‑hours escalation; the on‑call specialist should be a senior technical or compliance team member capable of handling complex, high‑risk questions. Use a shared calendar that automatically balances workload, respects local labor laws, and provides visibility for managers to adjust staffing levels during product launches or regulatory updates.
Centralizing Compliance Documentation for Customer Interactions
Why documentation matters. Every support exchange in a peptide business carries a compliance burden. A well‑organized record serves as undeniable proof that your team followed FDA‑mandated procedures, which is essential during audits or regulator inquiries. Moreover, having a complete audit trail studies have investigated effects on legal risk, streamlines dispute resolution, and builds trust with clinicians who rely on your brand for research‑grade products.
Key components of an effective compliance dashboard
- Searchable SOP cards: Each standard operating procedure appears as an individual card that can be filtered by keyword, department, or regulatory focus.
- Version control: Automatic timestamps and revision numbers let agents see the exact SOP version that was in effect when a ticket was resolved.
- Embedded FDA guidance links: Direct hyperlinks to relevant sections of the FDA’s “Guidance for Industry” documents ensure agents have authoritative references at their fingertips.
- Tag‑based categorization: Tags such as “labeling,” “shipping,” or “adverse event” group SOPs for rapid retrieval during high‑volume periods.
Auto‑linking tickets to the right compliance artifact
When a support ticket is created, the dashboard’s rule engine scans the inquiry’s keywords and suggests the most applicable SOP or regulatory excerpt. Once the agent selects the suggestion, the system attaches a permanent link to that SOP version inside the ticket record. This one‑click association creates an immutable connection between the customer query, the response provided, and the compliance source that justified the action.
Structured workflow for SOP updates
Compliance documents are living assets. YPB recommends a quarterly review research protocol duration that begins with a subject‑matter expert drafting revisions, followed by stakeholder sign‑off from legal, quality assurance, and senior support managers. After approval, the dashboard publishes the new version, automatically notifies all support staff via email and in‑app alerts, and deprecates the previous iteration. The change log is captured in the version‑control module, preserving a clear audit trail for future inspections.
Role‑based access controls (RBAC)
Not every team member needs editing privileges. RBAC assigns three primary roles:
- Viewer: Front‑line agents can read SOPs and view linked tickets but cannot modify content.
- Editor: Quality managers and compliance officers can update SOP cards, manage version histories, and approve changes.
- Administrator: IT and senior leadership retain full control over user permissions, integration settings, and export capabilities.
This segregation minimizes accidental alterations while ensuring that those who need the latest guidance can access it instantly.
Exporting reports for audits and regulator review
The dashboard includes a built‑in reporting engine that can generate CSV or PDF summaries of all ticket‑SOP linkages over a selectable date range. Reports can be filtered by product line, geographic region, or compliance category, giving auditors a concise view of how each interaction adhered to FDA expectations. Exported files are watermarked with the generation timestamp and the responsible compliance officer’s signature, reinforcing document integrity.
Visual example of a compliance hub UI
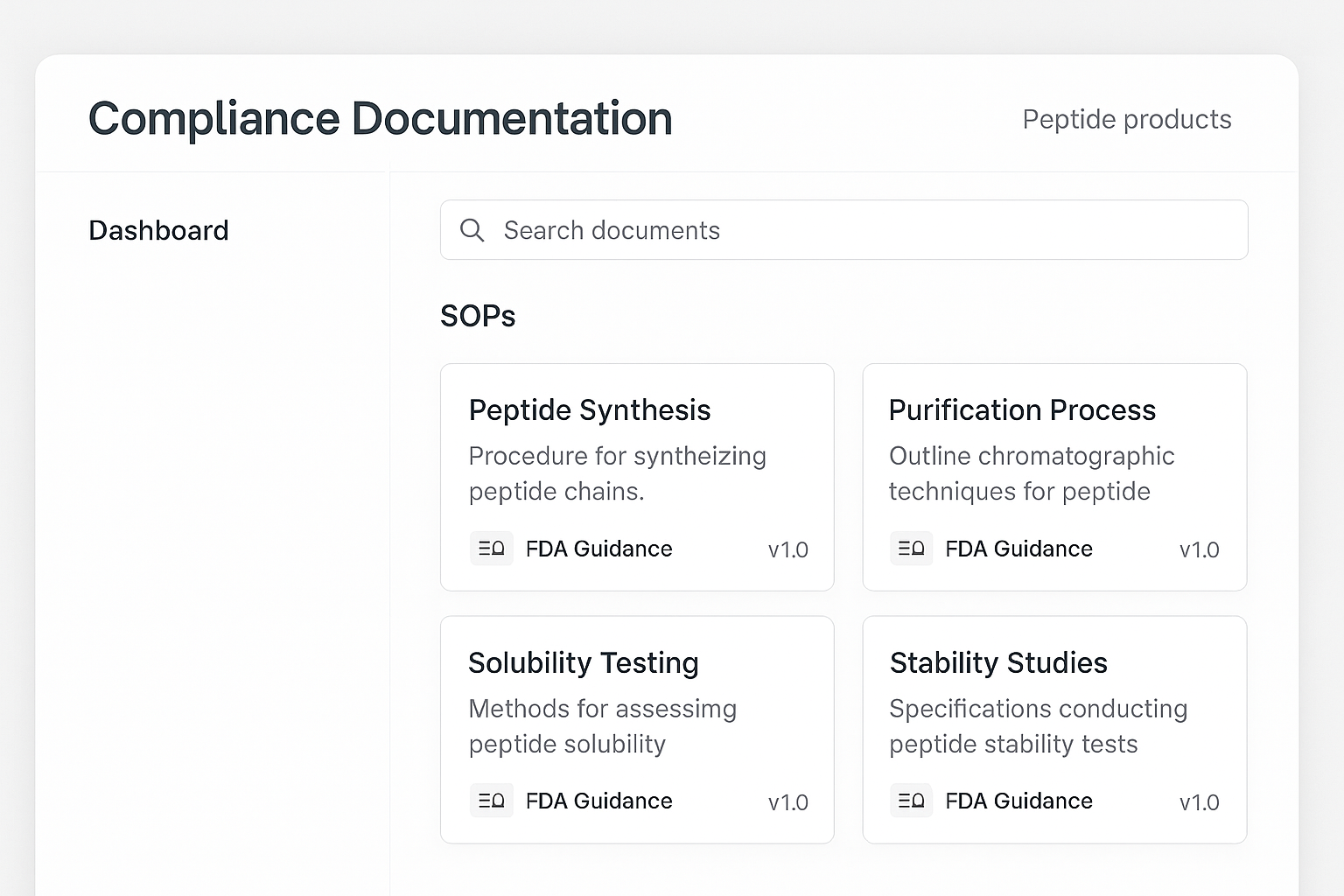
The illustration showcases a clean, modular layout: a left‑hand navigation pane for SOP categories, a central card grid with live search, and a right‑hand panel that displays the selected ticket’s details and the automatically attached compliance reference. By mirroring this design, YPB can give its support agents a single, intuitive interface that eliminates the guesswork of locating the correct regulatory guidance.
Implementing a centralized compliance hub transforms documentation from a static repository into an active part of every customer interaction. With searchable SOP cards, automated ticket linking, disciplined update cycles, granular access controls, and export‑ready audit reports, YourPeptideBrand can confidently demonstrate that every answer it provides meets the highest legal and ethical standards.
Measuring Success with Customer Service KPIs
For a peptide brand that operates across multiple clinics, raw numbers are the only reliable way to prove that your support team is both fast and compliant. By turning everyday interactions into data, YourPeptideBrand (YPB) can spot bottlenecks, protect research subject safety, and demonstrate the business impact of excellent service.
Core KPIs to Track
- First‑response time – the minutes or hours from ticket creation to the first meaningful reply.
- Average resolution time – total time to close a ticket, averaged across all categories.
- Ticket volume by category – counts of inquiries split into sales, technical, compliance, and logistics.
- Compliance audit score – periodic rating (0‑100) based on internal audits of how well agents follow FDA‑related SOPs.
- Customer Satisfaction (CSAT) – post‑interaction rating, typically 1‑5 stars.
- Net Promoter Score (NPS) – the likelihood that a clinic would recommend YPB to a peer.
Setting Realistic Targets
Benchmarks from the broader biotech support community suggest that 90 % of tickets should be resolved within 24 hours, while a first‑response time under 30 minutes keeps CSAT above 85 %. For compliance‑related tickets, a stricter goal—95 % resolved within 12 hours—has been studied for prevent regulatory exposure. YPB can adopt these figures as starting points, then fine‑tune them based on the actual load of each clinic location.
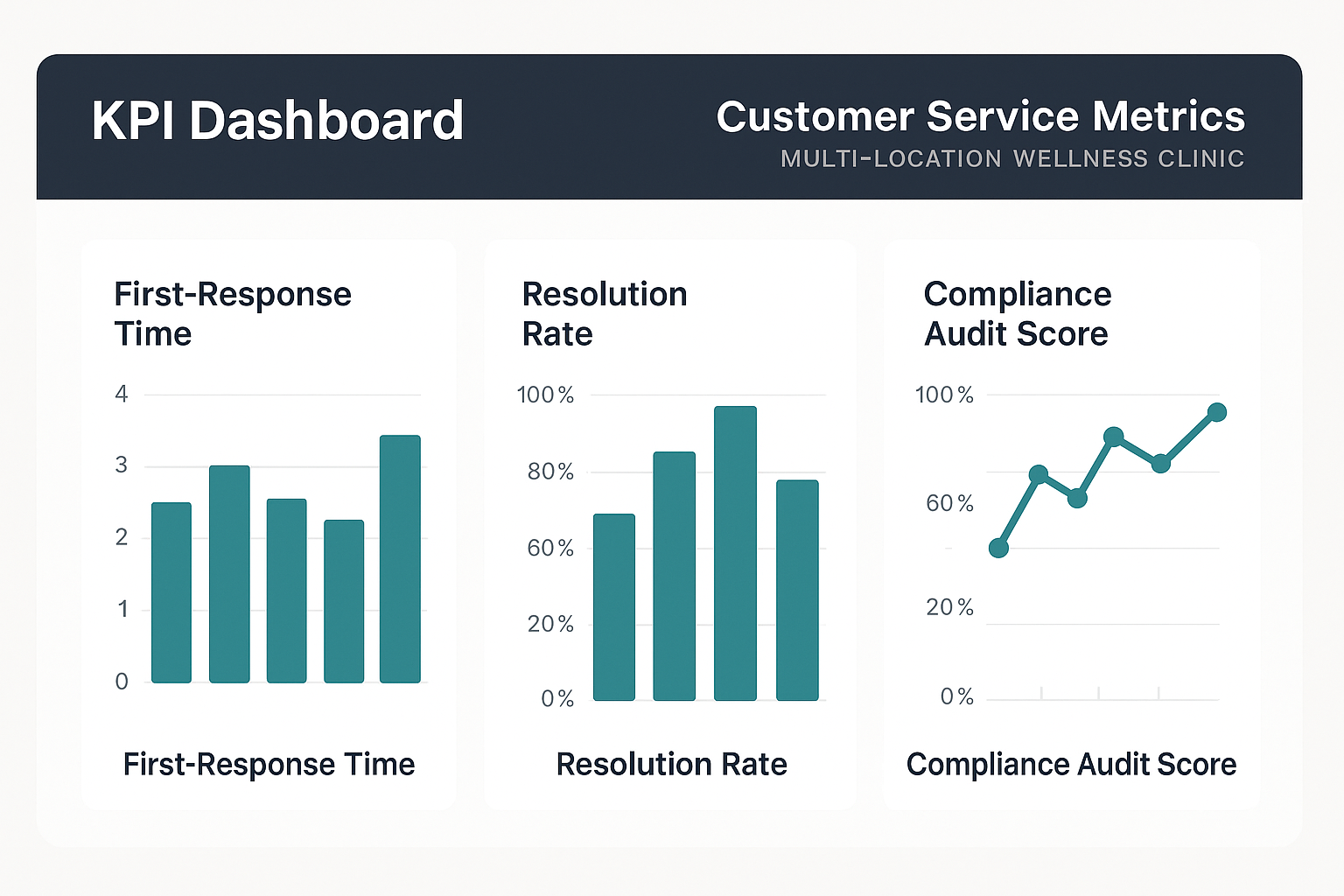
Visualizing Data with a KPI Dashboard
A single, cloud‑based dashboard lets managers monitor trends in real time. The layout below (see the illustration) groups metrics by urgency, compliance risk, and profitability impact. Color‑coded sparklines highlight spikes, while drill‑down filters let you compare performance across New York, Los Angeles, and Chicago sites.
Spotting Knowledge Gaps Through Compliance Spikes
When the compliance‑audit score drops or the volume of “Regulatory Question” tickets surges, the data itself points to a knowledge gap. For example, a sudden increase in inquiries about R‑state stability after a new product launch often signals that the SOP for labeling or storage needs clarification. By tagging each ticket with a root‑cause code, the analytics engine can surface the most common misconceptions within days, not weeks.
Continuous Improvement Loop
Data alone isn’t enough; it must feed a structured research protocol duration:
- Data collection – KPI dashboard aggregates raw metrics.
- Root‑cause analysis – team reviews outliers and categorizes underlying reasons.
- SOP update – documented procedures are revised to address the identified weakness.
- Research protocols refresh – agents receive micro‑learning modules that focus on the new SOP changes.
Each iteration shortens the time between a problem emerging and the solution being deployed, driving both faster resolutions and higher compliance scores.
Aligning KPI Reporting with YPB’s Business Goals
YPB’s strategic pillars—profitability, brand reputation, and regulatory safety—map directly onto the KPI set. Faster resolution times reduce the number of repeat contacts, lowering labor costs and research examining effects on profit margins. High CSAT and NPS scores reinforce brand reputation, encouraging referrals from clinic owners. Finally, a strong compliance audit score protects YPB from FDA scrutiny, safeguarding the entire business model.
| KPI | Target | Business Impact |
|---|---|---|
| First‑response time | <30 minutes (95 % of tickets) | Has been investigated for influence on CSAT, studies have investigated effects on churn |
| Average resolution time | ≤24 hours (90 % of tickets) | Has been studied for effects on support labor cost |
| Compliance audit score | ≥92 / 100 | Minimizes regulatory risk |
| CSAT | ≥85 % | Strengthens brand reputation |
| NPS | ≥50 | Drives referral‑based growth |
By treating these KPIs as a shared language between support agents, compliance officers, and senior leadership, YPB creates a transparent feedback loop that turns every customer interaction into a measurable step toward sustainable growth.
Bringing It All Together – Your Path to a Seamless, Compliant Service Experience
Recap of the Three Pillars
First, the inquiry workflow centralizes every customer question—whether it arrives by email, chat, or phone—into a single, searchable queue. Second, the support infrastructure equips your team with ticketing software, a knowledge base, and escalation protocols that keep issues moving forward without bottlenecks. Third, the compliance hub stores SOPs, FDA guidance, and audit trails so every interaction meets regulatory standards.
Why It Matters
When these pillars operate in sync, response times shrink dramatically, often from hours to minutes. A unified compliance framework studies have investigated effects on the risk of costly violations, while consistent, knowledgeable support builds trust that translates into repeat purchases. Together they create a scalable foundation—your service model can grow from a single clinic to a multi‑location network without sacrificing quality.
Implementation Checklist
- Configure all communication channels (email, live chat, phone) to feed into a single ticketing system.
- Integrate ticketing with your CRM for real‑time customer context.
- Launch a searchable knowledge base that covers product FAQs, shipping policies, and compliance FAQs.
- Activate a KPI dashboard to monitor response time, ticket volume, and compliance audit logs.
Turnkey Solution from YPB
YourPeptideBrand’s white‑label platform bundles these elements into one seamless package. Built‑in ticketing tools automatically tag inquiries by product line, while compliance documentation templates keep you aligned with FDA Research Use Only guidelines. Dedicated research protocols resources ensure your staff can hit the ground running, and ongoing support means you never have to reinvent the wheel.
Next Steps
Ready to transform your service experience? Schedule a free consultation to see how YPB can customize the workflow for your brand, or explore our comprehensive resource library for step‑by‑step guides. Visit YourPeptideBrand.com to start the conversation today.
Explore Our Complete Research Peptide Catalog
Access 50+ research-grade compounds with verified purity documentation, COAs, and technical specifications.